Keywords
5-aminopyrazoles
chemoselectivity
heterocyclization
itaconimides
pyrazolo[3,4-b]pyridines
recyclization
Abstract
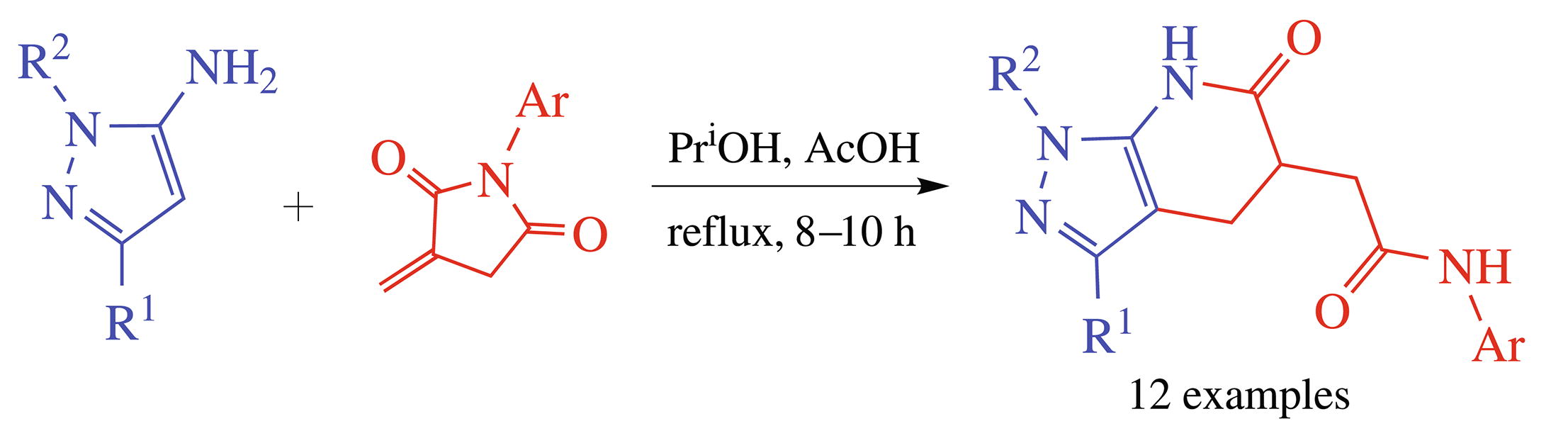
New polyfunctional hydrogenated pyrazolo[3,4-b]pyridines were obtained by the chemoselective reaction between N-arylitaconimides and 5-aminopyrazoles. The transformation is the cascade reaction including Michael addition at the active double bond of the itaconimide followed by the intramolecular transamidation and recyclization.
References
1.
![Synthesis and 3D QSAR of New Pyrazolo[3,4-b]pyridines: Potent and Selective Inhibitors of A1 Adenosine Receptors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Manetti F., Schenone S., Bondavalli F., Brullo C., Bruno O., Ranise A., Mosti L., Menozzi G., Fossa P., Trincavelli M.L., Martini C., Martinelli A., Tintori C., Botta M.
Journal of Medicinal Chemistry,
2005
2.
![Design, synthesis and biological activity of acyl substituted 3-amino-5-methyl-1,4,5,7-tetrahydropyrazolo[3,4-b]pyridin-6-ones as potential hypnotic drugs](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Falcó J.L., Lloveras M., Buira I., Teixidó J., Borrell J.I., Méndez E., Terencio J., Palomer A., Guglietta A.
European Journal of Medicinal Chemistry,
2005
3.
![5-Aryl-pyrazolo[3,4-b]pyridines: potent inhibitors of glycogen synthase kinase-3 (GSK-3)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Witherington J., Bordas V., Garland S.L., Hickey D.M., Ife R.J., Liddle J., Saunders M., Smith D.G., Ward R.W.
Bioorganic and Medicinal Chemistry Letters,
2003
4.
![Advances in the Synthesis of Pyrazolo[3,4-b]Pyridines](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
K. Dodiya D., R. Trivedi A., B. Kataria V., H. Shah V.
Current Organic Chemistry,
2012
5.
![Synthesis and biological evaluation of novel alkyl amide functionalized trifluoromethyl substituted pyrazolo[3,4-b]pyridine derivatives as potential anticancer agents.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chavva K., Pillalamarri S., Banda V., Gautham S., Gaddamedi J., Yedla P., Kumar C.G., Banda N.
Bioorganic and Medicinal Chemistry Letters,
2013
6.
![Facile, novel and efficient synthesis of new pyrazolo[3,4-b]pyridine products from condensation of pyrazole-5-amine derivatives and activated carbonyl groups](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ghaedi A., Bardajee G.R., Mirshokrayi A., Mahdavi M., Shafiee A., Akbarzadeh T.
RSC Advances,
2015
7.

Hamblin J.N., Angell T.D., Ballantine S.P., Cook C.M., Cooper A.W., Dawson J., Delves C.J., Jones P.S., Lindvall M., Lucas F.S., Mitchell C.J., Neu M.Y., Ranshaw L.E., Solanke Y.E., Somers D.O., et. al.
Bioorganic and Medicinal Chemistry Letters,
2008
8.
10.1016/j.mencom.2022.09.041_b0035
Tucker
Patent W0 2009117278 A2,
2009
9.
![Synthesis and anti-inflammatory evaluation of some pyrazolo[3,4-b]pyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sharma P.K., Singh K., Kumar S., Kumar P., Dhawan S.N., Lal S., Ulbrich H., Dannhardt G.
Medicinal Chemistry Research,
2010
10.

Meyers J., Carter M., Mok N.Y., Brown N.
Future Medicinal Chemistry,
2016
11.

Lovering F., Bikker J., Humblet C.
Journal of Medicinal Chemistry,
2009
12.

Morley A.D., Pugliese A., Birchall K., Bower J., Brennan P., Brown N., Chapman T., Drysdale M., Gilbert I.H., Hoelder S., Jordan A., Ley S.V., Merritt A., Miller D., Swarbrick M.E., et. al.
Drug Discovery Today,
2013
13.

Paterson I., Anderson E.A.
Science,
2005
14.

Cuenda A., Sanz-Ezquerro J.J.
Trends in Biochemical Sciences,
2017
15.

Over B., Wetzel S., Grütter C., Nakai Y., Renner S., Rauh D., Waldmann H.
Nature Chemistry,
2012
16.

17.
10.1016/j.mencom.2022.09.041_b0080
Al-Mousawi
J. Chem. Res., Synop.,
1997
18.

Chebanov V.A., Sakhno Y.I., Desenko S.M., Chernenko V.N., Musatov V.I., Shishkina S.V., Shishkin O.V., Kappe C.O.
Tetrahedron,
2007
19.
![Synthesis of pyrazolo[1,5‐a]pyrimidines in the reaction of 5‐amino‐3‐arylpyrazoles with methoxymethylene meldrum's acid derivatives and thermolysis of their pyrazolylaminomethylene derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Quiroga J., Hormaza A., Insuasty B., Saitz C., Cañete A., Jullian C.
Journal of Heterocyclic Chemistry,
1998
20.
![Reaction of 5-amino-1-aryl-3-methylpyrazoles with benzylidene derivatives of meldrum's acid: Synthesis and characterization of pyrazolo[3,4-b]pyridinones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Quiroga J., Hormaza A., Insuasty B., Márquez M.
Journal of Heterocyclic Chemistry,
1998
21.
![SYNTHESIS, MOLECULAR STRUCTURE AND TAUTOMERISM OF 1(2)H-DIHYDROPYRAZOLO[3,4-b]PYRIDIN-6-ONES](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Quiroga J., Insuasty B., Hormaza A., Cabildo P., Claramunt R.M., Elguero J.
Heterocyclic Communications,
1999
22.
![Synthesis of pyrazolo[3,4-b]pyridines under microwave irradiation in multi-component reactions and their antitumor and antimicrobial activities – Part 1](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
El-borai M.A., Rizk H.F., Abd-Aal M.F., El-Deeb I.Y.
European Journal of Medicinal Chemistry,
2012
23.

Vasantha V.A., Jana S., Parthiban A., Vancso J.G.
Chemical Communications,
2014
24.
![Design, synthesis and biological evaluation of 1H-pyrrolo[2,3-b]pyridine and 1H-pyrazolo[3,4-b]pyridine derivatives as c-Met inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Liu N., Wang Y., Huang G., Ji C., Fan W., Li H., Cheng Y., Tian H.
Bioorganic Chemistry,
2016
25.
![Design, Synthesis, and Biological Evaluation of AT1 Angiotensin II Receptor Antagonists Based on the Pyrazolo[3,4-b]pyridine and Related Heteroaromatic Bicyclic Systems](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cappelli A., Nannicini C., Gallelli A., Giuliani G., Valenti S., Mohr G.L., Anzini M., Mennuni L., Ferrari F., Caselli G., Giordani A., Peris W., Makovec F., Giorgi G., Vomero S., et. al.
Journal of Medicinal Chemistry,
2008
26.
10.1016/j.mencom.2022.09.041_b0115
Fathy
J. Chem. Pharm. Res.,
2015
27.
![Synthesis and functionalisation of 1H-pyrazolo[3,4-b]pyridines involving copper and palladium-promoted coupling reactions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Lavecchia G., Berteina-Raboin S., Guillaumet G.
Tetrahedron Letters,
2004
28.
10.1016/j.mencom.2022.09.041_b0125
Panchal
Int. J. Appl. Eng. Res.,
2019
29.
![A novel method for the synthesis of pyrimido[1,2-a]benzimidazoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Kokonova A.V., Potapov A.Y., Kolpakova M.G., Sabynin A.L., Zubkov F.I.
Chemistry of Heterocyclic Compounds,
2016
30.
![A novel synthetic approach to hydroimidazo[1,5-b]pyridazines by the recyclization of itaconimides and HPLC–HRMS monitoring of the reaction pathway](/storage/images/resized/ex6KJoZujZOZFZh7jGfeHauiftuB3CI7iwJVFRDg_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Potapov A.Y., Krysin M.Y., Zubkov F.I., Sapronova L.V.
Beilstein Journal of Organic Chemistry,
2017
31.

Kovygin Y.A., Shikhaliev K.S., Krysin M.Y., Potapov A.Y., Ledenyova I.V., Kosheleva Y.A., Vandyshev D.Y.
Chemistry of Heterocyclic Compounds,
2019
32.

33.

Ferris R.G., Hazen R.J., Roberts G.B., St. Clair M.H., Chan J.H., Romines K.R., Freeman G.A., Tidwell J.H., Schaller L.T., Cowan J.R., Short S.A., Weaver K.L., Selleseth D.W., Moniri K.R., Boone L.R., et. al.
Antimicrobial Agents and Chemotherapy,
2005
34.

Gu S., Zhang X., He Q., Yang L., Ma X., Zheng Y., Yang S., Chen F.
Bioorganic and Medicinal Chemistry,
2011