Keywords
coordination polymers
copper(I) iodide
photoluminescence
thermally activated delayed fluorescence
triphenylarsine
Abstract
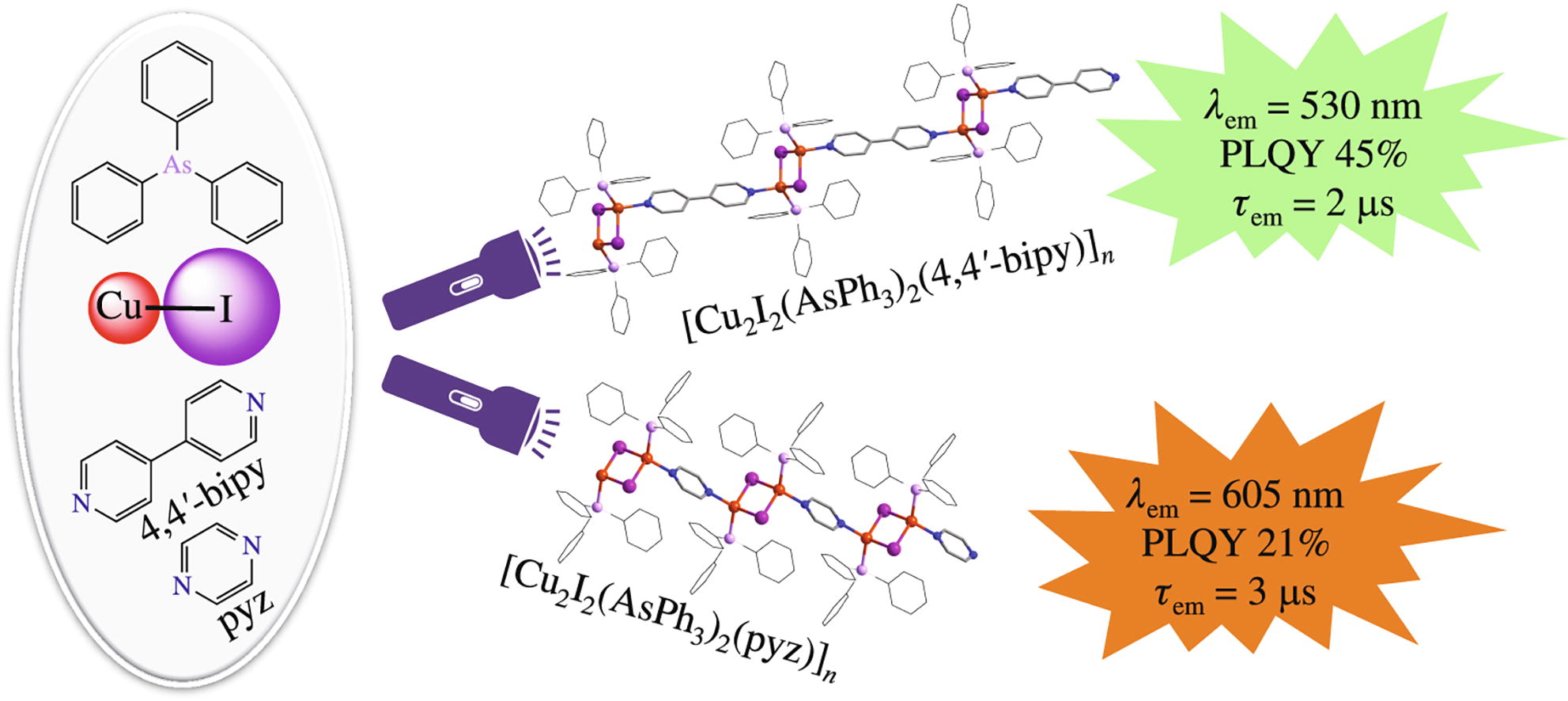
1D coordination polymers [Cu2I2(AsPh3)2(L)]n (L = 4,4'-bi-pyridine, pyrazine) were synthesized via the reaction of CuI with AsPh3 and N,N'-ditopic co-ligand s L. At ambient temperature, these polymers exhibit short-lived (2.3–3.2 μs) thermally activated delayed fluorescence (TADF) with max at 530 nm (L = 4,4'-bipyridine) and 605 nm (L = pyrazine) and quantum efficiency up to 45%.
References
1.

Kirst C., Tietze J., Mayer P., Böttcher H., Karaghiosoff K.
ChemistryOpen,
2022
2.

3.

Moutier F., Schiller J., Calvez G., Lescop C.
Organic Chemistry Frontiers,
2021
4.

Romanov A.S., Chotard F., Rashid J., Bochmann M.
Dalton Transactions,
2019
5.

Czerwieniec R., Leitl M.J., Homeier H.H., Yersin H.
Coordination Chemistry Reviews,
2016
6.

Yersin H., Czerwieniec R., Shafikov M.Z., Suleymanova A.F.
ChemPhysChem,
2017
7.

Huitorel B., Utrera-Melero R., Massuyeau F., Mevelec J., Baptiste B., Polian A., Gacoin T., Martineau-Corcos C., Perruchas S.
Dalton Transactions,
2019
8.

Cariati E., Lucenti E., Botta C., Giovanella U., Marinotto D., Righetto S.
Coordination Chemistry Reviews,
2016
9.

10.

Gou Y., Chen M., Li S., Deng J., Li J., Fang G., Yang F., Huang G.
Journal of Medicinal Chemistry,
2021
11.

Wallesch M., Volz D., Zink D.M., Schepers U., Nieger M., Baumann T., Bräse S.
Chemistry - A European Journal,
2014
12.

Galimova M.F., Zueva E.M., Dobrynin A.B., Samigullina A.I., Musin R.R., Musina E.I., Karasik A.A.
Dalton Transactions,
2020
13.

Kobayashi R., Inaba R., Imoto H., Naka K.
Bulletin of the Chemical Society of Japan,
2021
14.

Galimova M.F., Zueva E.M., Dobrynin A.B., Kolesnikov I.E., Musin R.R., Musina E.I., Karasik A.A.
Dalton Transactions,
2021
15.

Kobayashi R., Imoto H., Naka K.
European Journal of Inorganic Chemistry,
2020
16.

Kobayashi R., Kihara H., Kusukawa T., Imoto H., Naka K.
Chemistry Letters,
2021
17.

Artem'ev A.V., Demyanov Y.V., Rakhmanova M.I., Bagryanskaya I.Y.
Dalton Transactions,
2022
18.

Korenaga T., Ko A., Uotani K., Tanaka Y., Sakai T.
Angewandte Chemie - International Edition,
2011
19.

Brayton D.F., Beaumont P.R., Fukushima E.Y., Sartain H.T., Morales-Morales D., Jensen C.M.
Organometallics,
2014
20.

Chishiro A., Konishi M., Inaba R., Yumura T., Imoto H., Naka K.
Dalton Transactions,
2022
21.
Titov A.A., Filippov O.A., Smol’yakov A.F., Averin A.A., Shubina E.S.
Mendeleev Communications,
2021
22.

Green L.P., Steel T.R., Riisom M., Hanif M., Söhnel T., Jamieson S.M., Wright L.J., Crowley J.D., Hartinger C.G.
Frontiers in Chemistry,
2021
23.

Bratanovici B., Nicolescu A., Shova S., Dascălu I., Ardeleanu R., Lozan V., Roman G.
Research on Chemical Intermediates,
2019
24.

Kharisov B.I., Elizondo Martínez P., Jiménez-Pérez V.M., Kharissova O.V., Nájera Martínez B., Pérez N.
Journal of Coordination Chemistry,
2009
25.

Pettinari C., Tăbăcaru A., Boldog I., Domasevitch K.V., Galli S., Masciocchi N.
Inorganic Chemistry,
2012
26.
![[Zn2Ac2(μ-Ac)2(bpymb)]n (Ac, acetate; bpymb, 1,4-bis(4-pyridylmethyl)benzene), a 2D coordination polymer obtained with a flexible N,N’-ditopic bipyridine linker](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Portolés-Gil N., Vallcorba O., Domingo C., López-Periago A., Ayllón J.A.
Inorganica Chimica Acta,
2021
27.

Araki H., Tsuge K., Sasaki Y., Ishizaka S., Kitamura N.
Inorganic Chemistry,
2005
28.

Yang L., Powell D.R., Houser R.P.
Dalton Transactions,
2007
29.
CrysAlisPro 1.171.38.46, Data Collection, Reduction and Correction Program, Rigaku Oxford Diffraction, 2015.
30.
Bruker APEX3 software suite: APEX3, SADABS-2016/2 and SAINT, version 2018.7-2, Bruker AXS Inc., Madison, WI, 2017.
31.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
32.
10.1016/j.mencom.2022.09.027_b0160
Montalti
Handbook of Photochemistry,
2006