Keywords
1,2-diamino-4-phenylimidazole
arylidenecyanoacetates
cascade processes
diastereomers
imidazo[1,5-b]pyridazines
quantum chemical calculations
regioselectivity
Abstract
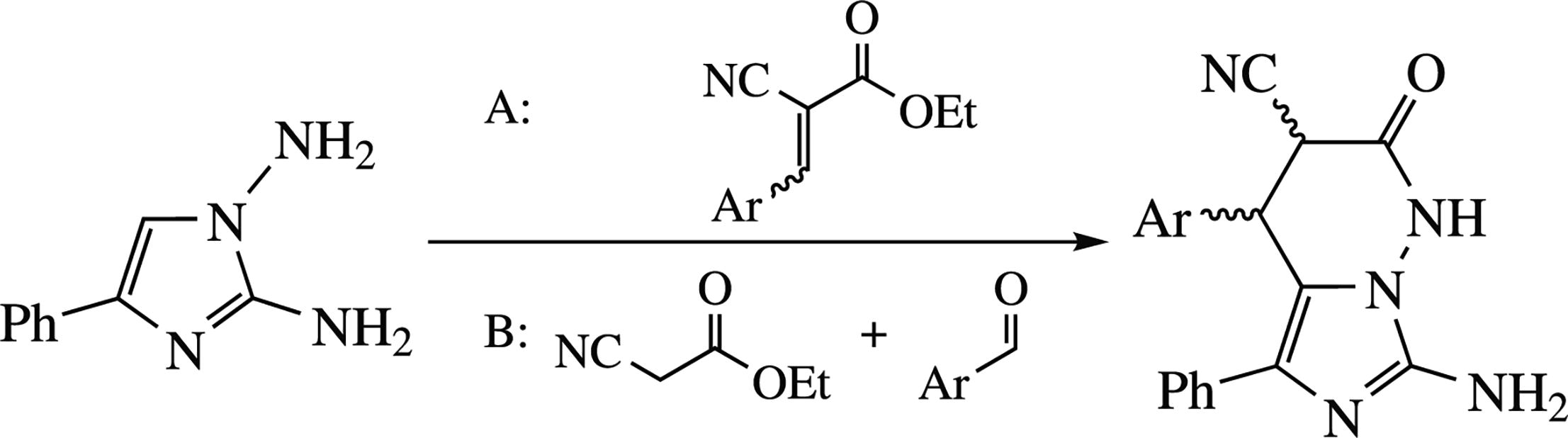
The cascade heterocyclization of 1,2-diamino-4-phenyl-imidazole with ethyl 2-arylidene-2-cyanoacetates affords 1,2,3,4-tetrahydroimidazo[1,5-b]pyridazine-3-carbonitrile derivatives as mixtures of diastereomers. The experimental data and quantum chemical calculations were used to propose processes. The three-component processing with above-mentioned diamine, ethyl cyanoacetate and aromatic aldehydes leads to the same products in generally lower yields.
References
1.

Murray C.W., Rees D.C.
Angewandte Chemie - International Edition,
2015
2.

Palmer N., Peakman T.M., Norton D., Rees D.C.
Organic and Biomolecular Chemistry,
2016
3.

Luise N., Wyatt P.G.
Chemistry - A European Journal,
2018
4.

Twigg D.G., Kondo N., Mitchell S.L., Galloway W.R., Sore H.F., Madin A., Spring D.R.
Angewandte Chemie - International Edition,
2016
5.

Hassan H., Marsden S.P., Nelson A.
Bioorganic and Medicinal Chemistry,
2018
6.

Lüthy M., Wheldon M.C., Haji-Cheteh C., Atobe M., Bond P.S., O’Brien P., Hubbard R.E., Fairlamb I.J.
Bioorganic and Medicinal Chemistry,
2015
7.

Campbell P.S., Jamieson C., Simpson I., Watson A.J.
Chemical Communications,
2018
8.

Lovering F., Bikker J., Humblet C.
Journal of Medicinal Chemistry,
2009
9.

Ritchie T.J., Macdonald S.J., Young R.J., Pickett S.D.
Drug Discovery Today,
2011
10.

Hann M.M., Leach A.R., Harper G.
Journal of Chemical Information and Computer Sciences,
2001
11.

Foulks J.M., Carpenter K.J., Luo B., Xu Y., Senina A., Nix R., Chan A., Clifford A., Wilkes M., Vollmer D., Brenning B., Merx S., Lai S., McCullar M.V., Ho K., et. al.
Neoplasia,
2014
12.
![T-type channel blocking properties and antiabsence activity of two imidazo[1,2-b]pyridazine derivatives structurally related to indomethacin](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Rimoli M.G., Russo E., Cataldi M., Citraro R., Ambrosino P., Melisi D., Curcio A., De Lucia S., Patrignani P., De Sarro G., Abignente E.
Neuropharmacology,
2009
13.

Chapman T.M., Osborne S.A., Bouloc N., Large J.M., Wallace C., Birchall K., Ansell K.H., Jones H.M., Taylor D., Clough B., Green J.L., Holder A.A.
Bioorganic and Medicinal Chemistry Letters,
2013
14.
J. Kehler, L. K. Rasmussen, M. Langgård, M. Jessing, P.J.V. Vital, K. Juhl, M. Marigo, Patent WO 2019/121840 A1, 2019.
15.
R.T. Skerlj, E.M.J. Bourque, W.J. Greenlee, P.T. Lansbury, Patent WO 2017/192930, 2017.
16.
C. Hirth-Dietrich, P. Sandner, J.-P. Stasch, M. Hahn, M. Follmann, Vakalopoulos, Patent WO 2016/177660 A1, 2016.
17.

Follmann M., Ackerstaff J., Redlich G., Wunder F., Lang D., Kern A., Fey P., Griebenow N., Kroh W., Becker-Pelster E., Kretschmer A., Geiss V., Li V., Straub A., Mittendorf J., et. al.
Journal of Medicinal Chemistry,
2017
18.
![Structure-based design, synthesis, and biological evaluation of imidazo[1,2-b]pyridazine-based p38 MAP kinase inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kaieda A., Takahashi M., Takai T., Goto M., Miyazaki T., Hori Y., Unno S., Kawamoto T., Tanaka T., Itono S., Takagi T., Hamada T., Shirasaki M., Okada K., Snell G., et. al.
Bioorganic and Medicinal Chemistry,
2018
19.
Lipson V.V., Svetlichnaya N.V., Shishkina S.V., Shishkin O.V.
Mendeleev Communications,
2008
20.
![A novel synthetic approach to hydroimidazo[1,5-b]pyridazines by the recyclization of itaconimides and HPLC–HRMS monitoring of the reaction pathway](/storage/images/resized/ex6KJoZujZOZFZh7jGfeHauiftuB3CI7iwJVFRDg_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Potapov A.Y., Krysin M.Y., Zubkov F.I., Sapronova L.V.
Beilstein Journal of Organic Chemistry,
2017
21.
![Condensation of 1,2-diamino-4-phenylimidazole and N-arylmaleimides with the formation of new tetrahydroimidazo[1,5-b]pyridazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Potapov A.Y., Krysin M.Y.
Chemistry of Heterocyclic Compounds,
2015
22.
10.1016/j.mencom.2022.09.023_b0110
El-Gazzar
Egypt. J. Chem.,
2003
23.
![Reactions with heterocyclic amidines: synthesis of several new pyrazolo[1,5-a]pyrimidines and pyrazolo[1,5-c]-as-triazines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sadek K.U., Selim M.A., El-Maghraby M.A.
Journal of Chemical & Engineering Data,
1985
24.
Mohareb R.M., Megally Abdo N.Y., Al-farouk F.O.
Acta Chimica Slovenica,
2017
25.

Chavan S.S., Supekar M.V., Burate P.A., Rupanwar B.D., Shelke A.M., Suryavanshi G.
Organic and Biomolecular Chemistry,
2017
26.

Zahedifar M., Sheibani H.
Research on Chemical Intermediates,
2013
27.
![Three-component synthesis of some 2-amino-5-hydroxy-[1,2,4]triazolo[1,5-a]pyrimidine-6-carbonitriles and 2-(cyanoamino)-4-hydroxypyrimidine-5-carbonitriles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ranjbar-Karimi R., Beiki-Shoraki K., Amiri A.
Monatshefte fur Chemie,
2010
28.
![Heterocyclic synthesis with nitriles: Syntheses of some novel pyrazolo[1,5-a]pyrimidine and Pyrrolo[2?,3?:3,4]pyrazolo[1,5-a]pyrimidine Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Abdelrazek F.M.
Journal für praktische Chemie,
1989
29.
Yu. Vandyshev D., Kosheleva E.A., Polikarchuk V.A., Mangusheva D.A., Denisov G.L., Shikhaliev K.S.
Mendeleev Communications,
2021
30.

Iwasaki S.
Helvetica Chimica Acta,
1976
31.

Gong P., Xu F., Cheng L., Gong X., Zhang J., Gu W., Han W.
Chemical Communications,
2021
32.

Bonfio C., Caumes C., Duffy C.D., Patel B.H., Percivalle C., Tsanakopoulou M., Sutherland J.D.
Journal of the American Chemical Society,
2019
33.
![Regioselective Synthesis of Imidazo[1,5‐ b ]pyridazines by Cascade Cyclizations of 1,2‐Diamino‐4H‐phenylimidazole with 1,3‐Diketones, Acetoacetic Ester and Their Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Kruzhilin A.A., Kosheleva E.A., Shikhaliev K.S., Denisov G.L., Vandyshev D.Y.
ChemistrySelect,
2021
34.

Ibrahim H.M., Ahmed Arafa W.A., Behbehani H.
RSC Advances,
2018