Keywords
aryl azides
aryl isatin imines
aza-Wittig reaction
imines
isatins
stereoselectivity
Abstract
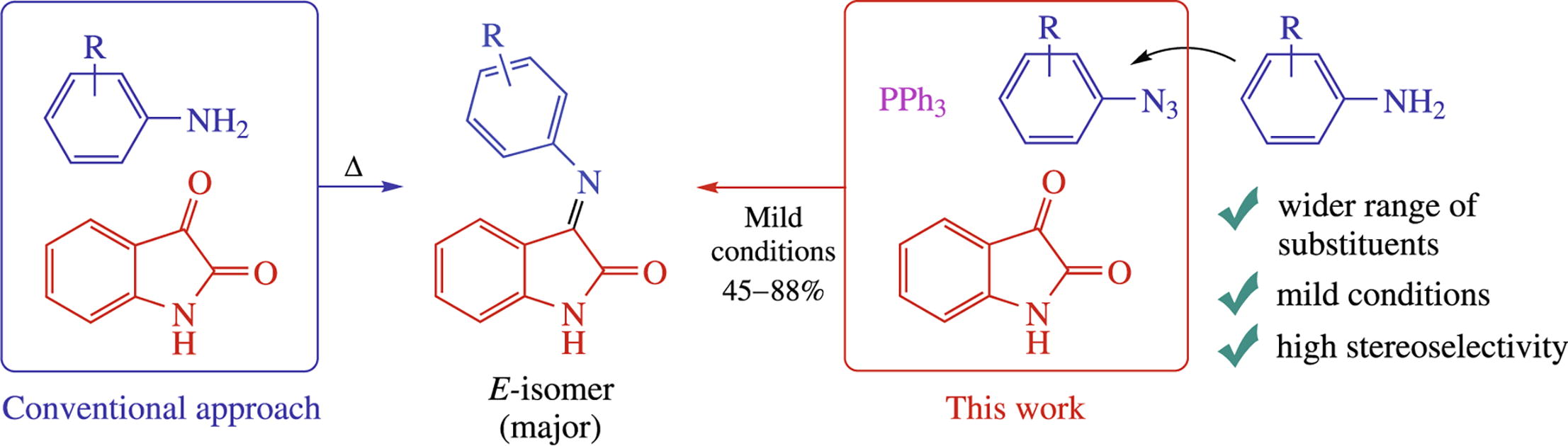
A general synthesis of NH-isatin N′-arylimines involves the aza-Wittig reaction of isatin and aryl azides, providing higher E-diastereoselectivity as compared to previously described methods. The procedure is applicable even to anilines which do not directly react with isatins.
References
1.

Ding K., Lu Y., Nikolovska-Coleska Z., Qiu S., Ding Y., Gao W., Stuckey J., Krajewski K., Roller P.P., Tomita Y., Parrish D.A., Deschamps J.R., Wang S.
Journal of the American Chemical Society,
2005
2.

Chinnasamy R., Sundararajan R., Govindaraj S.
Journal of Advanced Pharmaceutical Technology and Research,
2010
3.

Bogdanov A.V., Voloshina A.D., Khamatgalimov A.R., Terekhova N.V., Mironov V.F.
Doklady Chemistry,
2020
4.

Liu S., Mao Q., Zhan G., Qin R., Chen B., Xue J., Luo M., Zhao Q., Han B.
Organic and Biomolecular Chemistry,
2021
5.

Sai Allaka B., Basavoju S., Rama Krishna G.
Advanced Synthesis and Catalysis,
2021
6.
Zlotin S.G., Banina O.A., Sudarikov D.V., Nigmatov A.G., Frolova L.L., Kutchin A.V.
Mendeleev Communications,
2020
7.
10.1016/j.mencom.2022.09.022_b0035
Luczywo
Nat. Prod. Res.,
2021
8.

Chen T., Cai C.
Organic and Biomolecular Chemistry,
2016
9.

Ribeiro C.J., Nunes R.C., Amaral J.D., Gonçalves L.M., Rodrigues C.M., Moreira R., Santos M.M.
European Journal of Medicinal Chemistry,
2017
10.
![Stereocontrolled [3+2] Cycloaddition of Donor-Acceptor Cyclopropanes to Iminooxindoles: Access to Spiro[oxindole-3,2′-pyrrolidines]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Akaev A.A., Bezzubov S.I., Desyatkin V.G., Vorobyeva N.S., Majouga A.G., Melnikov M.Y., Budynina E.M.
Journal of Organic Chemistry,
2019
11.

Sun C., Lin X., Weinreb S.M.
Journal of Organic Chemistry,
2006
12.

Jarrahpour A., Zarei M.
Tetrahedron,
2009
13.
![Rh2(OAc)4 catalyzed substrate selective [4+2]/[2+2] cycloaddition of acylketenes: a highly chemo- and regioselective synthesis of spiro(oxindolyl)oxazinones and β-lactams](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Subba Reddy B.V., Karthik G., Rajasekaran T., Antony A., Sridhar B.
Tetrahedron Letters,
2012
14.

Bashiri M., Jarrahpour A., Nabavizadeh S.M., Karimian S., Rastegari B., Haddadi E., Turos E.
Medicinal Chemistry Research,
2020
15.
![Synthesis of 1,3-diaryl-spiro[azetidine-2,3′-indoline]-2′,4-diones: Via the Staudinger reaction: Cis - Or trans -diastereoselectivity with different addition modes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Filatov V., Kukushkin M., Kuznetsova J., Skvortsov D., Tafeenko V., Zyk N., Majouga A., Beloglazkina E.
RSC Advances,
2020
16.

Filatov V.E., Kuznetsova J., Petrovskaya L., Yuzabchuk D., Tafeenko V.A., Zyk N.V., Beloglazkina E.K.
ACS Omega,
2021
17.
10.1016/j.mencom.2022.09.022_b0085
Verma
Acta Pharm.,
2004
18.
10.1016/j.mencom.2022.09.022_b0090
Banerji
Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem.,
2005
19.

Azizian J., Mohammadi M.K., Firuzi O., Razzaghi-asl N., Miri R.
Medicinal Chemistry Research,
2011
20.

Mio S., Kumagawa Y., Sugai S.
Tetrahedron,
1991
21.

22.

23.

Üngören Ş.H., Kani İ., Günay A.
Tetrahedron Letters,
2012
24.

Wang L., Wang Y., Chen M., Ding M.
Advanced Synthesis and Catalysis,
2014
25.

Malamiri F., Khaksar S., Badri R., Tahanpesar E.
Combinatorial Chemistry and High Throughput Screening,
2019
26.

Yan W., Wang D., Feng J., Li P., Zhao D., Wang R.
Organic Letters,
2012
27.

Nakamura S., Hyodo K., Nakamura M., Nakane D., Masuda H.
Chemistry - A European Journal,
2013
28.

Holmquist M., Blay G., Pedro J.R.
Chemical Communications,
2014
29.

Zhao H., Li B., Pang H., Tian T., Chen X., Song X., Meng W., Yang Z., Zhao Y., Liu Y.
Organic Letters,
2016
30.

da Silva C.D., Katla R., dos Santos B.F., Tavares Junior J.M., Albuquerque T.B., Kupfer V.L., Rinaldi A.W., Domingues N.L.
Synthesis,
2019
31.

Nakamura S., Matsuzaka K., Hatanaka T., Funahashi Y.
Organic Letters,
2020
32.

Brønsted Base-Catalyzed Reductive Cyclization of Alkynyl α-Iminoesters through Auto-Tandem Catalysis
Kondoh A., Terada M.
Organic Letters,
2018
33.

Yavari I., Adib M., Hojabri L.
Tetrahedron,
2002
34.

El Khatib F., Bellan J., Koenig M.
Phosphorus, Sulfur and Silicon and the Related Elements,
1998
35.

Simmons J.T., Allen J.R., Morris D.R., Clark R.J., Levenson C.W., Davidson M.W., Zhu L.
Inorganic Chemistry,
2013