Keywords
cyclohexanone
Ketones
levoglucosenone
Michael adduct
regioselective reduction
Abstract
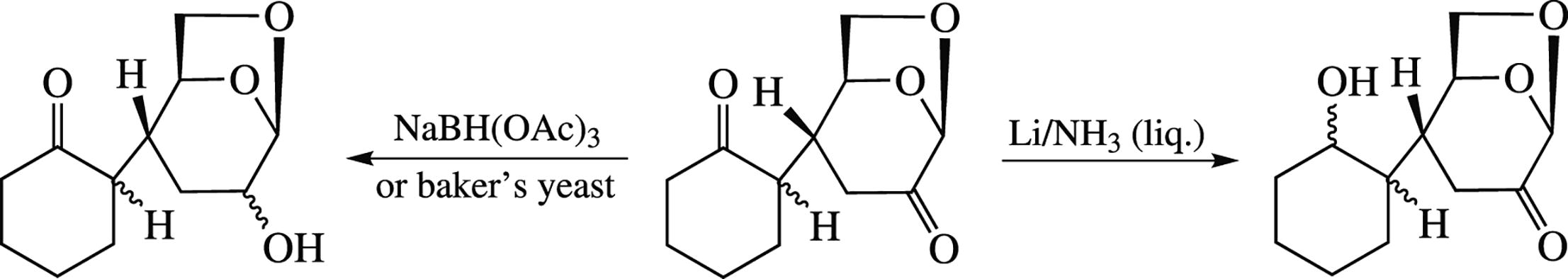
Conditions for the highly regioselective reduction of the specified keto groups in the Michael adduct of levoglucosenone and cyclohexanone have been developed. Selective reduction of the keto group in the cyclohexanone moiety was performed under the action of lithium metal in NH3/THF system. Reduction of the keto group in the carbohydrate residue was accomplished microbiologically or by refluxing with NaBH(OAc)3 in benzene.
References
1.
10.1016/j.mencom.2022.09.021_h0005
Khalilova
Russ. J. Org. Chem.,
2014
2.
Faizullina L.K., Khalilova Y.A., Valeev F.A.
Mendeleev Communications,
2021
3.

Galimova Y.S., Tagirov A.R., Faizullina L.K., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2017
4.

Stork G., Darling S.D.
Journal of the American Chemical Society,
1964
5.

Stork G., Rosen P., Goldman N., Coombs R.V., Tsuji J.
Journal of the American Chemical Society,
1965
6.
10.1016/j.mencom.2022.09.021_h0030
Caine
Organic Reactions,
1976
7.
10.1016/j.mencom.2022.09.021_h0035
Dryden
Organic Reactions in Steroid Chemistry,
1972
8.

Ves’kina N.A., Afon’kina S.R., Odinokov V.N.
Russian Journal of Organic Chemistry,
2011
9.
10.1016/j.mencom.2022.09.021_h0045
Gribble
J. Chem. Soc., Chem. Commun.,
1975
10.
![Recent studies on veratrum alkaloids: a new reaction of sodium triacetoxyborohydride [NaBH(OAc)3]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Saksena A.K., Mangiaracina P.
Tetrahedron Letters,
1983
11.

Turnbull M.D., Hatter G., Ledgerwood D.E.
Tetrahedron Letters,
1984
12.
Sharipov B.T., Davydova A.N., Faizullina L.K., Valeev F.A.
Mendeleev Communications,
2019