Keywords
ammonium acetate
C–H acids
multicomponent reactions
stereoselectivity
trifluoromethylpiperidines
Abstract
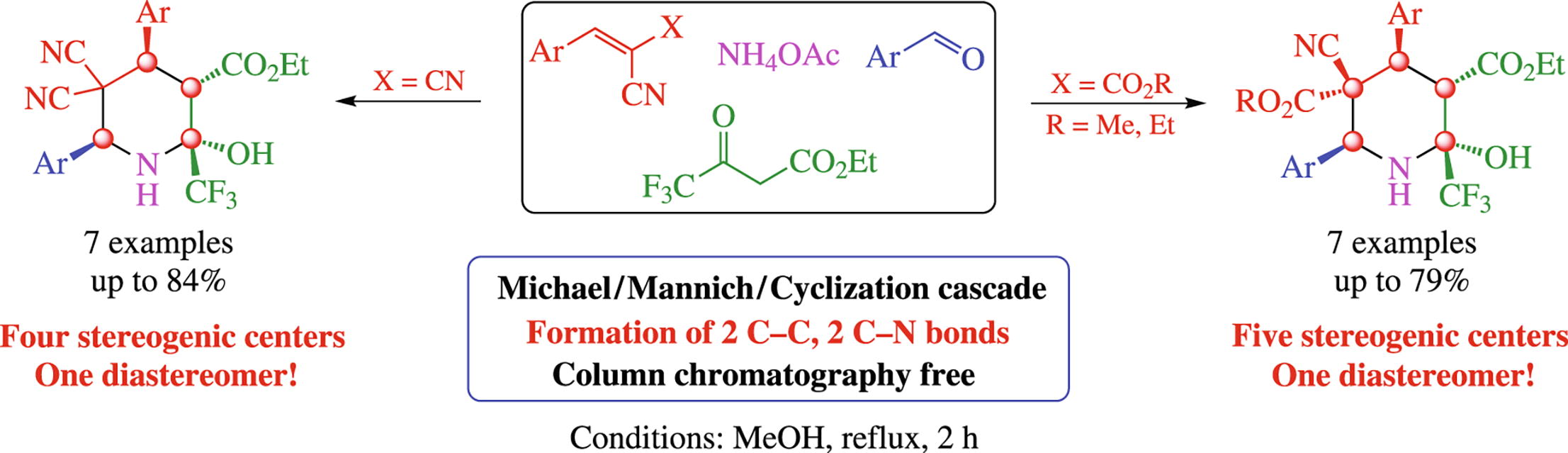
The Michael–Mannich cascade cyclization of cyano olefins, ethyl 4,4,4-trifluoro-3-oxobutanoate, aromatic aldehydes and ammonium acetate provides convenient stereoselective formation of ethyl 5,5-dicyano-4,6-diaryl-2-hydroxy-2-(trifluoromethyl)piperidine-3-carboxylates with fourstereogenic centers and dialkyl 4,6-diaryl-5-cyano-2-hydroxy-2-(trifluoromethyl)piperidine-3,5-dicarboxylates with five stereogenic centers. Ammonium acetate plays dual role, acting as a base and as a nitrogen source.
References
1.

von Itzstein M., Wu W., Kok G.B., Pegg M.S., Dyason J.C., Jin B., Van Phan T., Smythe M.L., White H.F., Oliver S.W., Colman P.M., Varghese J.N., Ryan D.M., Woods J.M., Bethell R.C., et. al.
Nature,
1993
2.

Kim C.U., Lew W., Williams M.A., Liu H., Zhang L., Swaminathan S., Bischofberger N., Chen M.S., Mendel D.B., Tai C.Y., Laver W.G., Stevens R.C.
Journal of the American Chemical Society,
1997
3.

Borza I., Domany G.
Current Topics in Medicinal Chemistry,
2006
4.

den Ouden H., Pellis L., Rutten G.E., Geerars-van Vonderen I.K., Rubingh C.M., van Ommen B., van Erk M.J., Beulens J.W.
Metabolomics,
2016
5.
10.1016/j.mencom.2022.09.020_b0025
Organofluorine Chemistry: Principles and Commercial Application,
1994
6.
10.1016/j.mencom.2022.09.020_b0030
Organic Chemistry in Medicinal Chemistry and Biochemical Applications,
1993
7.

Iwai N., Sakai R., Tsuchida S., Kitazume M., Kitazume T.
Journal of Fluorine Chemistry,
2009
8.

Buscemi S., Pace A., Palumbo Piccionello A., Macaluso G., Vivona N., Spinelli D., Giorgi G.
Journal of Organic Chemistry,
2005
9.

Wu J., Zhang R., Dai A., Guo S., He F.
Chinese Journal of Organic Chemistry,
2021
10.

Guo S., Zhao W., Wang Y., Zhang W., Chen S., Wei P., Wu J.
Journal of Agricultural and Food Chemistry,
2021
11.

Zhao L., Hu J., Wang Z., Yin M., Zou Y., Gao S., Fu Y., Ye F.
Pesticide Biochemistry and Physiology,
2020
12.
10.1016/j.mencom.2022.09.020_b0060
Multicomponent Reactions in Organic Synthesis,
2015
13.

Vereshchagin A.N.
Russian Chemical Bulletin,
2017
14.

Dömling A.
Chemical Reviews,
2005
15.
Vereshchagin A.N., Karpenko K.A., Elinson M.N., Dorofeeva E.O., Goloveshkin A.S., Egorov M.P.
Mendeleev Communications,
2018
16.

Vereshchagin A.N., Karpenko K.A., Elinson M.N., Goloveshkin A.S., Ushakov I.E., Egorov M.P.
Research on Chemical Intermediates,
2018
17.

Vereshchagin A.N., Karpenko K.A., Elinson M.N., Goloveshkin A.S., Dorofeeva E.O., Egorov M.P.
Research on Chemical Intermediates,
2019
18.

Elinson M.N., Feducovich S.K., Zaimovskaya T.A., Vereshchagin A.N., Gorbunov S.V., Nikishin G.I.
Russian Chemical Bulletin,
2005
19.
Vereshchagin A.N., Elinson M.N., Stepanov N.O., Nikishin G.I.
Mendeleev Communications,
2009
20.

Elinson M.N., Fedukovich S.K., Zaimovskaya T.A., Vereshchagin A.N., Nikishin G.I.
Russian Chemical Bulletin,
2003
21.

Elinson M.N., Vereshchagin A.N., Ryzkov F.V.
Current Organic Chemistry,
2017
22.

Vereshchagin A.N., Iliyasov T.M., Karpenko K.A., Smirnov V.A., Ushakov I.E., Elinson M.N.
Chemistry of Heterocyclic Compounds,
2021
23.

Zhou L., Yuan F., Zhou Y., Duan W., Zhang M., Deng H., Song L.
Tetrahedron,
2018
24.

Song L., Shi W., Wang Y., Zhu Y., Zhang M., Deng H.
Synthesis,
2016
25.
![Iodine catalyzed one-pot multi-component reaction to CF3-containing spiro[indene-2,3′-piperidine] derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dai B., Duan Y., Liu X., Song L., Zhang M., Cao W., Zhu S., Deng H., Shao M.
Journal of Fluorine Chemistry,
2012