Keywords
ball milling
cascade reactions
gram-scale synthesis
indoles
mechanochemical reactions
multi-substituted 3-(1H-pyrrol-2-yl)-1H-indoles
multicomponent reactions
Abstract
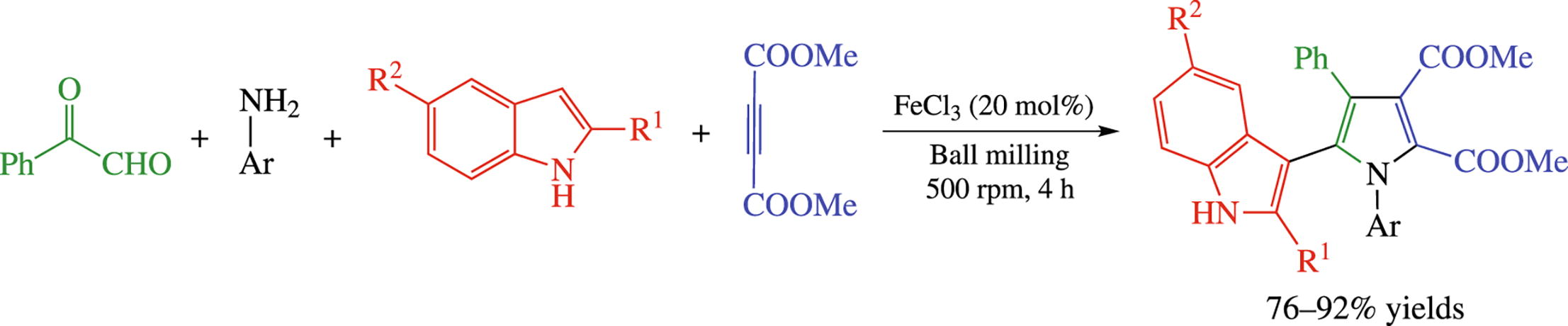
Iron(iii) chloride has been found to serve as an efficient catalyst for a mechanochemical (ball milling) one-pot four- component cascade reaction of phenyl glyoxal, anilines, indoles and activated alkyne affording highly-substituted 3-(1H-pyrrol-2-yl)-1H-indoles. The procedure is beneficial because of mild conditions, easily available starting materials, cheap catalyst and possibility for scaling.
References
1.

Anastas P.T., Kirchhoff M.M.
Accounts of Chemical Research,
2002
2.

Sheldon R.A.
Green Chemistry,
2005
3.

Sarkar A., Santra S., Kundu S.K., Hajra A., Zyryanov G.V., Chupakhin O.N., Charushin V.N., Majee A.
Green Chemistry,
2016
4.

Li J., Nagamani C., Moore J.S.
Accounts of Chemical Research,
2015
5.

Leonardi M., Villacampa M., Menéndez J.C.
Chemical Science,
2018
6.

Bolm C., Hernández J.G.
Angewandte Chemie - International Edition,
2019
7.

Zhu S., Li F., Wang G.
Chemical Society Reviews,
2013
8.

Egorov I.N., Santra S., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Majee A., Ranu B.C., Rusinov V.L., Chupakhin O.N.
Green Chemistry,
2020
9.

Cozzi P., Mongelli N.
Current Pharmaceutical Design,
1970
10.
10.1016/j.mencom.2022.09.018_b0050
Cirrincione
Chemistry of Heterocyclic Compounds,
1992
11.
10.1016/j.mencom.2022.09.018_b0055
Comprehensive Heterocyclic Chemistry II,
1996
12.

Bhardwaj V., Gumber D., Abbot V., Dhiman S., Sharma P.
RSC Advances,
2015
13.

Chatterjee T., Srinivasan A., Ravikanth M., Chandrashekar T.K.
Chemical Reviews,
2016
14.

Wilkerson W.W., Galbraith W., Gans-Brangs K., Grubb M., Hewes W.E., Jaffee B., Kenney J.P., Kerr J., Wong N.
Journal of Medicinal Chemistry,
1994
15.
Adamovich S.N., Sadykov E.K., Ushakov I.A., Oborina E.N., Belovezhets L.A.
Mendeleev Communications,
2021
16.

Curran D., Grimshaw J., Perera S.D.
Chemical Society Reviews,
1991
17.
New benzodithiophene-pyrrolopyrroledione-thienopyrazine random terpolymers for organic photovoltaics
Keshtov M.L., Kuklin S.A., Konstantinov I.O., Zou Y., Sharma G.D.
Mendeleev Communications,
2021
18.
19.

Balakrishna A., Aguiar A., Sobral P.J., Wani M.Y., Almeida e Silva J., Sobral A.J.
Catalysis Reviews - Science and Engineering,
2018
20.

Lei T., Liu W., Li J., Huang M., Yang B., Meng Q., Chen B., Tung C., Wu L.
Organic Letters,
2016
21.

Quinonero O., Jean M., Vanthuyne N., Roussel C., Bonne D., Constantieux T., Bressy C., Bugaut X., Rodriguez J.
Angewandte Chemie - International Edition,
2015
22.

Estévez V., Villacampa M., Menéndez J.C.
Chemical Society Reviews,
2014
23.

Declerck V., Ribière P., Martinez J., Lamaty F.
Journal of Organic Chemistry,
2004
24.

Reddy B.V., Reddy M.R., Rao Y.G., Yadav J.S., Sridhar B.
Organic Letters,
2013
25.
10.1016/j.mencom.2022.09.018_b0125
2010
26.
27.

Dömling A.
Chemical Reviews,
2005
28.

Przhevalskii N.M., Laypanov R.K., Tokmakov G.P., Lukina I.V., Vershinkin D.A., Tafeenko V.A.
Russian Chemical Bulletin,
2021
29.

Bolm C., Legros J., Le Paih J., Zani L.
Chemical Reviews,
2004
30.

Sherry B.D., Fürstner A.
Accounts of Chemical Research,
2008
31.

Mahato S., Mukherjee A., Santra S., Zyryanov G.V., Majee A.
Organic and Biomolecular Chemistry,
2019
32.

Samanta S., Mondal S., Santra S., Kibriya G., Hajra A.
Journal of Organic Chemistry,
2016
33.
![Iron(III)-catalyzed three-component domino strategy for the synthesis of imidazo[1,2-a]pyridines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Santra S., Mitra S., Bagdi A.K., Majee A., Hajra A.
Tetrahedron Letters,
2014
34.
![Iron(III)-Catalyzed Cascade Reaction between Nitroolefins and 2-Aminopyridines: Synthesis of Imidazo[1,2-a]pyridines and Easy Access towards Zolimidine](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Santra S., Bagdi A.K., Majee A., Hajra A.
Advanced Synthesis and Catalysis,
2013
35.

Chatterjee S., Harden I., Bistoni G., Castillo R.G., Chabbra S., van Gastel M., Schnegg A., Bill E., Birrell J.A., Morandi B., Neese F., DeBeer S.
Journal of the American Chemical Society,
2022
36.

Tomifuji R., Maeda K., Takahashi T., Kurahashi T., Matsubara S.
Organic Letters,
2018
37.

Yang L., Lei C., Wang D., Huang Z., Wang M.
Organic Letters,
2010