Keywords
2-fluoroallyl amines
allylic amination
chiral palladium complexes
enantioselective catalysis
fluoroalkenes
Abstract
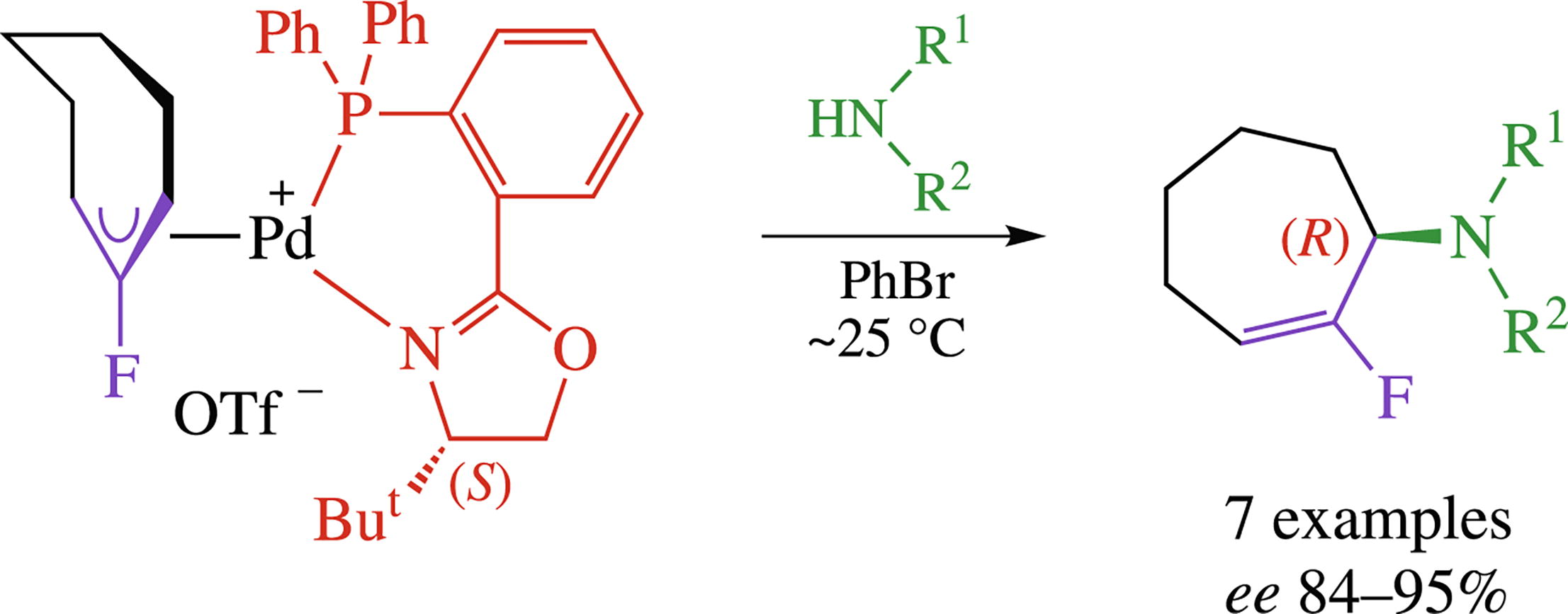
The key enantioselectivity-determining step in Pd-catalyzed asymmetric amination of 2-fluoroallylic substrates was optimized using model reaction of η3-(2-fluorocycloheptenyl)-palladium complexes bearing chiral P,P- and P,N-ligands with various amines. (S)-ButPHOX was found to be the most effective ligand allowing the formation of 2-fluoroallyl amines and anilines with high enantioselectivity.
References
1.

Meanwell N.A.
Journal of Medicinal Chemistry,
2018
2.

Johnson B.M., Shu Y., Zhuo X., Meanwell N.A.
Journal of Medicinal Chemistry,
2020
3.

Edmondson S.D., Wei L., Xu J., Shang J., Xu S., Pang J., Chaudhary A., Dean D.C., He H., Leiting B., Lyons K.A., Patel R.A., Patel S.B., Scapin G., Wu J.K., et. al.
Bioorganic and Medicinal Chemistry Letters,
2008
4.
10.1016/j.mencom.2022.09.016_b0020
Drouin
ChemBioChem,
1817
5.

Drouin M., Wadhwani P., Grage S.L., Bürck J., Reichert J., Tremblay S., Mayer M.S., Diel C., Staub A., Paquin J., Ulrich A.S.
Chemistry - A European Journal,
2020
6.
Afanasyev O.I., Cherkashchenko I., Kuznetsov A., Kliuev F., Semenov S., Chusova O., Denisov G., Chusov D.
Mendeleev Communications,
2020
7.
Runikhina S., Chusov D.
Mendeleev Communications,
2021
8.

Drouin M., Paquin J.
Beilstein Journal of Organic Chemistry,
2017
9.
10.1016/j.mencom.2022.09.016_b0045
Zhang
Eur. J. Med. Chem.,
2019
10.

Zhang B., Shan G., Zheng Y., Yu X., Ruan Z., Li Y., Lei X.
Marine Drugs,
2019
11.

Wei B., Gunzner-Toste J., Yao H., Wang T., Wang J., Xu Z., Chen J., Wai J., Nonomiya J., Tsai S.P., Chuh J., Kozak K.R., Liu Y., Yu S., Lau J., et. al.
Journal of Medicinal Chemistry,
2017
12.

Lamy C., Hofmann J., Parrot-Lopez H., Goekjian P.
Tetrahedron Letters,
2007
13.

Dutheuil G., Bailly L., Couve-Bonnaire S., Pannecoucke X.
Journal of Fluorine Chemistry,
2007
14.

Jacobsen C.B., Nielsen M., Worgull D., Zweifel T., Fisker E., Jørgensen K.A.
Journal of the American Chemical Society,
2011
15.

Lu Y., Goldstein E.L., Stoltz B.M.
Organic Letters,
2018
16.

Schwarz K.J., Pearson C.M., Cintron‐Rosado G.A., Liu P., Snaddon T.N.
Angewandte Chemie,
2018
17.

Zhang X., Cheng Y., Zhao X., Cao Z., Xiao X., Xu Y.
Organic Chemistry Frontiers,
2021
18.

Drouin M., Paquin J.
Tetrahedron,
2018
19.

Pàmies O., Margalef J., Cañellas S., James J., Judge E., Guiry P.J., Moberg C., Bäckvall J., Pfaltz A., Pericàs M.A., Diéguez M.
Chemical Reviews,
2021
20.

Evans P., Grange R., Clizbe E.
Synthesis,
2016
21.

Bobrova A.Y., Novikov M.A., Mezentsev I.A., Tomilov Y.V.
Journal of Fluorine Chemistry,
2020
22.

Bobrova A.Y., Novikov M.A., Tomilov Y.V.
Organic and Biomolecular Chemistry,
2021