Keywords
in vitro antibacterial screening
N-acylhydrazones
1,3,4-oxadiazoles
benzohydrazide
chloramine trihydrate
intramolecular cyclization
multicomponent reactions
oxidative cyclization
SwissADME prediction study
tandem reactions
Abstract
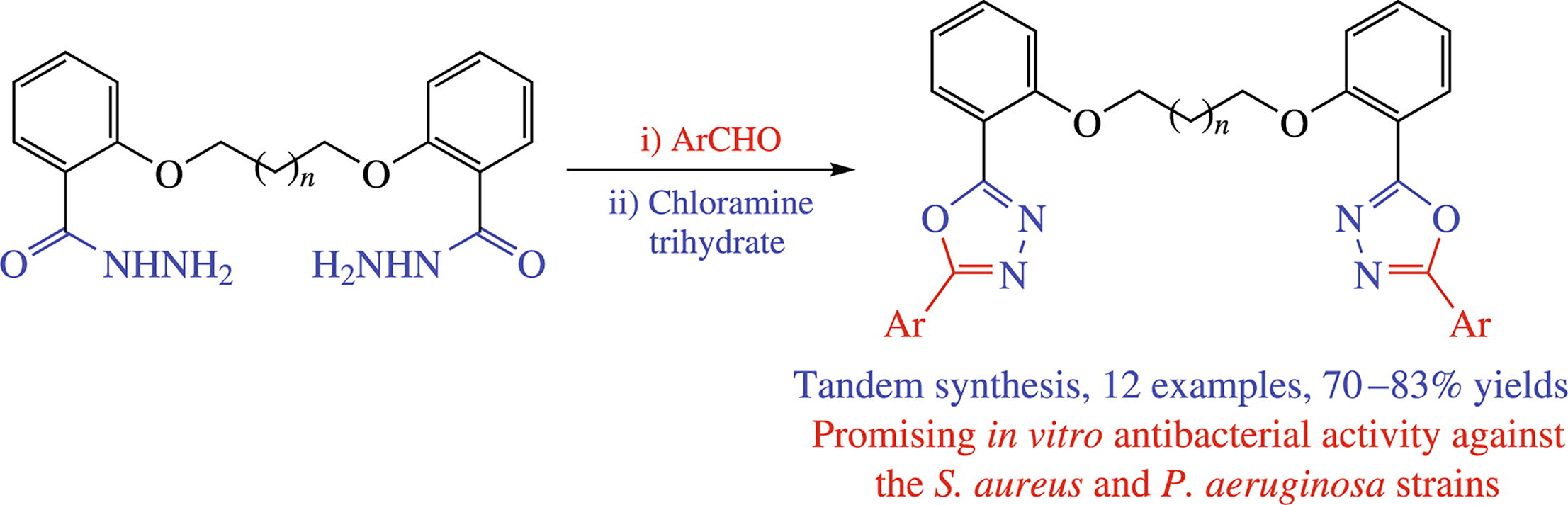
A three-component tandem protocol involving the reactions of bis(benzohydrazides), aromatic aldehydes and chloramine trihydrate yielded a new series of bis(1,3,4-oxadiazoles). The target hybrids were formed by an initial bis(N-benzoyl-hydrazones) formation, followed by chloramine trihydrate- mediated oxidative cyclization. The 4-methoxyphenyl-containing compounds demonstrated promising antibacterial activity against the Staphylococcus aureus and Pseudomonas aeruginosa strains with MIC value of 1.6 µm.
References
1.

Laxminarayan R., Duse A., Wattal C., Zaidi A.K., Wertheim H.F., Sumpradit N., Vlieghe E., Hara G.L., Gould I.M., Goossens H., Greko C., So A.D., Bigdeli M., Tomson G., Woodhouse W., et. al.
The Lancet Infectious Diseases,
2013
2.

Mekky A.E., Sanad S.M.
Bioorganic Chemistry,
2020
3.

Annunziato
International Journal of Molecular Sciences,
2019
4.
![New pyrido[3′,2′:4,5]thieno[3,2-d]pyrimidin-4(3H)-one hybrids linked to arene units: synthesis of potential MRSA, VRE, and COX-2 inhibitors](/storage/images/resized/qZfu8vVyaDIo81Au73p6SNE8AGGzf73yfiTC33bu_small_thumb.webp)
Sanad S.M., Mekky A.E.
Canadian Journal of Chemistry,
2021
5.
![Synthesis, cytotoxicity, in-vitro antibacterial screening and in-silico study of novel thieno[2,3-b]pyridines as potential pim-1 inhibitors](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Mekky A.E., Sanad S.M., Said A.Y., Elneairy M.A.
Synthetic Communications,
2020
6.

Xu W., Silverman M.H., Yu X.Y., Wright G., Brown N.
Bioorganic and Medicinal Chemistry,
2019
7.

Guimarães C.R., Boger D.L., Jorgensen W.L.
Journal of the American Chemical Society,
2005
8.

Nazar S., Siddiqui N., Alam O.
Archiv der Pharmazie,
2020
9.

Ahsan M.J., Choupra A., Sharma R.K., Jadav S.S., Padmaja P., Hassan M.Z., Al-Tamimi A.B., Geesi M.H., Bakht M.A.
Anti-Cancer Agents in Medicinal Chemistry,
2017
10.

Gan X., Hu D., Li P., Wu J., Chen X., Xue W., Song B.
Pest Management Science,
2015
11.
![Design, Synthesis and Biological Evaluation of 2 (((5-aryl-1,2,4-oxadiazol-3-yl)methyl)thio)benzo[d]oxazoles: New Antiinflammatory and Antioxidant Agents](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Yatam S., Jadav S.S., Gundla R., Gundla K.P., Reddy G.M., Ahsan M.J., Chimakurthy J.
ChemistrySelect,
2018
12.

Antimicrobial and antiprotozoal activity of 3-acetyl-2,5-disubstituted-1,3,4-oxadiazolines: a review
Paruch K., Popiołek Ł., Wujec M.
Medicinal Chemistry Research,
2019
13.

Poonam, Bhasin G., Srivastava R., Singh R.
Journal of the Iranian Chemical Society,
2021
14.

Siwach A., Verma P.K.
BMC Chemistry,
2020
15.

Zheng Z., Liu Q., Kim W., Tharmalingam N., Fuchs B.B., Mylonakis E.
Future Medicinal Chemistry,
2018
16.

Chauhan J., Ravva M.K., Sen S.
Organic Letters,
2019
17.

Abdildinova A., Gong Y.
ACS Combinatorial Science,
2018
18.

Wang S., Wang K., Kong X., Zhang S., Jiang G., Ji F.
Advanced Synthesis and Catalysis,
2019
19.

Liu L., Feng S.
Organic and Biomolecular Chemistry,
2017
20.
![Utility of Pyridine‐2(1 H )‐thiones in the Synthesis of Novel Bis‐Thieno[2,3‐ b ]pyridines and Their Fused Azines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sanad S.M., Abdel‐Fattah A.M., Attaby F.A., Elneairy M.A.
Journal of Heterocyclic Chemistry,
2019
21.

Zhao Q., Ren L., Hou J., Yu W., Chang J.
Organic Letters,
2018
22.
![3‐Aminopyrazolo[3,4‐ b ]pyridine: Effective Precursor for Barium Hydroxide‐Mediated Three Components Synthesis of New Mono‐ and Bis(pyrimidines) with Potential Cytotoxic Activity](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sanad S.M., Mekky A.E.
Chemistry and Biodiversity,
2021
23.

Sanad S.M., Mekky A.E.
Journal of the Iranian Chemical Society,
2020
24.

Benković T., Kontrec D., Kazazić S., Chiş V., Miljanić S., Galić N.
Molecular Diversity,
2019
25.

26.

Grover J., Bhatt N., Kumar V., Patel N.K., Gondaliya B.J., Elizabeth Sobhia M., Bhutani K.K., Jachak S.M.
RSC Advances,
2015
27.

Yu W., Huang G., Zhang Y., Liu H., Dong L., Yu X., Li Y., Chang J.
Journal of Organic Chemistry,
2013
28.

Musad E.A., Mohamed R., Ali Saeed B., Vishwanath B.S., Lokanatha Rai K.M.
Bioorganic and Medicinal Chemistry Letters,
2011
29.

Cin G.T., Verep G., Topel S.D., Ciger V.
Chemistry of Heterocyclic Compounds,
2013
30.
Sanad S.M., Mekky A.E., Said A.Y., Elneairy M.A.
Mendeleev Communications,
2021
31.

Mohammad H., Reddy P.V., Monteleone D., Mayhoub A.S., Cushman M., Seleem M.N.
European Journal of Medicinal Chemistry,
2015
32.
10.1016/j.mencom.2022.09.014_b0160
Navin
C: J. Biosci.,
2017
33.

Rezki N., Al-Yahyawi A., Bardaweel S., Al-Blewi F., Aouad M.
Molecules,
2015
34.

Hofny H.A., Mohamed M.F., Gomaa H.A., Abdel-Aziz S.A., Youssif B.G., El-koussi N.A., Aboraia A.S.
Bioorganic Chemistry,
2021
35.

Omar F.A., Abelrasoul M., Sheha M.M., Hassan H.Y., Ibrahiem Y.M.
ChemistrySelect,
2018
36.

Shingare R.M., Patil Y.S., Sangshetti J.N., Patil R.B., Rajani D.P., Madje B.R.
Medicinal Chemistry Research,
2018
37.

Daina A., Michielin O., Zoete V.
Scientific Reports,
2017
38.

Lipinski C.A.
Drug Discovery Today: Technologies,
2004