Keywords
acetonitrile
amides
antiviral agents
caryophyllene
humulene
influenza
molecular modeling
Ritter reaction
Abstract
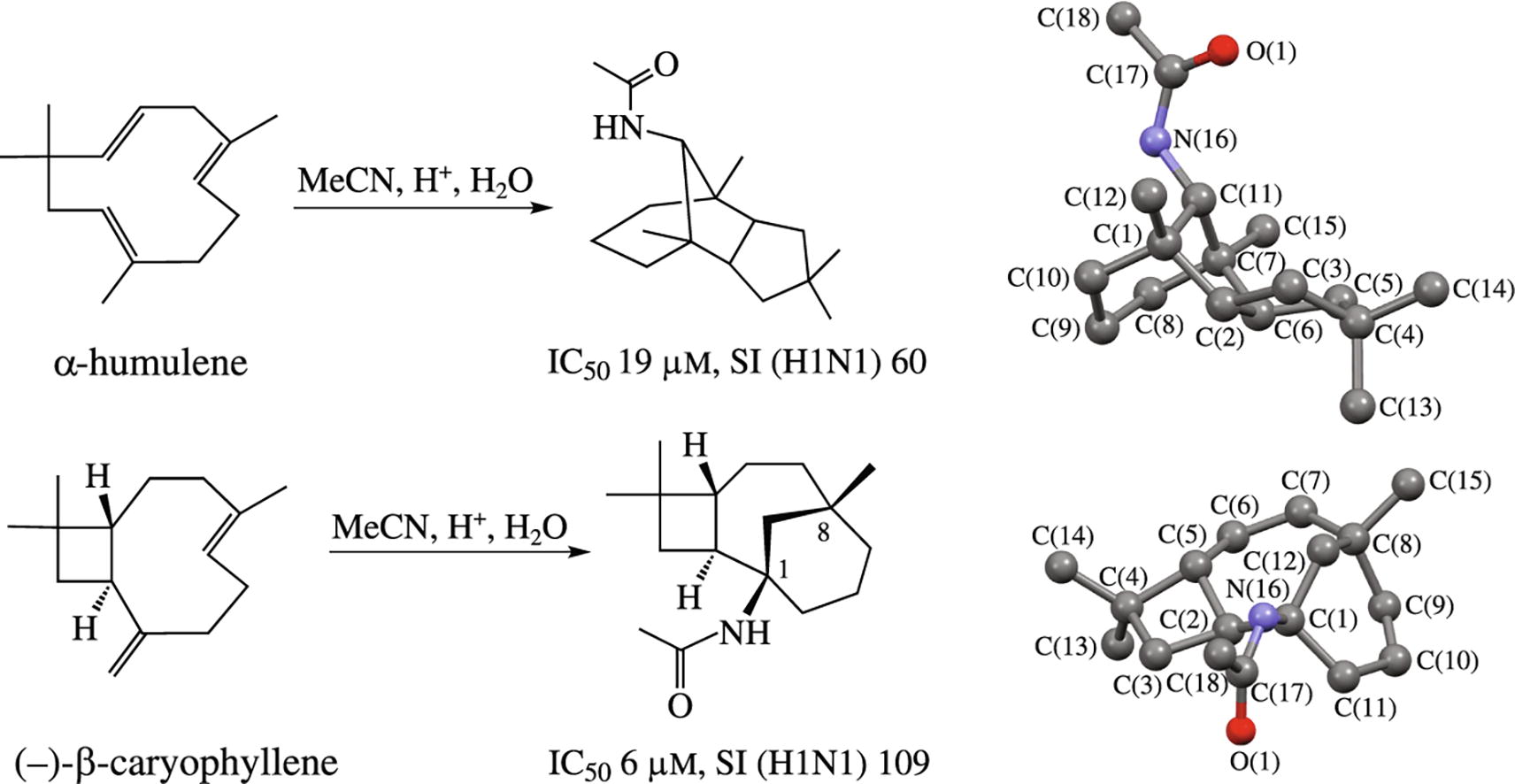
The Ritter reaction of humulene with acetonitrile occurs as the biomimetic process to afford the amide having the skeleton of a natural alcohol. The structures of the amides obtained from humulene and caryophyllene were confirmed by XRD data. The activity of some cage compounds against influenza virus allowed one to suggest the mechanism of antiviral action based on interfering with membrane fusion activity of viral hemagglutinin.
References
1.

Wani A.R., Yadav K., Khursheed A., Rather M.A.
Microbial Pathogenesis,
2021
2.

Yarovaya O.I., Salakhutdinov N.F.
Russian Chemical Reviews,
2021
3.

Fernandes E.S., Passos G.F., Medeiros R., da Cunha F.M., Ferreira J., Campos M.M., Pianowski L.F., Calixto J.B.
European Journal of Pharmacology,
2007
4.

Legault Jean, Dahl Wivecke, Debiton Eric, Pichette André, Madelmont Jean-Claude
Planta Medica,
2003
5.

Zheng G., Kenney P.M., Lam L.K.
Journal of Natural Products,
1992
6.

Sylvestre M., Legault J., Dufour D., Pichette A.
Phytomedicine,
2005
7.

Sylvestre M., Pichette A., Lavoie S., Longtin A., Legault J.
Phytotherapy Research,
2007
8.

Goren A.C., Piozzi F., Akcicek E., Kılıç T., Çarıkçı S., Mozioğlu E., Setzer W.N.
Phytochemistry Letters,
2011
9.

Yarovaya O.I., Korchagina D.V., Rybalova T.V., Gatilov Y.V., Polovinka M.P., Barkhash V.A.
Russian Journal of Organic Chemistry,
2004
10.

Chen M., Chen X., Hu Y., Ye R., Lv J., Li B., Zhang F.
Organic Chemistry Frontiers,
2021
11.
![An alkaloid-like 3-azabicyclo[3.3.1]non-3-ene library obtained from the bridged Ritter reaction](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Williams S.G., Bhadbhade M., Bishop R., Ung A.T.
Tetrahedron,
2017
12.

Nickon A., Iwadare T., McGuire F.J., Mahajan J.R., Narang S.A., Umezawa B.
Journal of the American Chemical Society,
1970
13.

Polovinka M.P., Korchagina D.V., Gatilov Y.V., Bagrianskaya I.Y., Barkhash V.A., Perutskii V.B., Ungur N.D., Vlad P.F., Shcherbukhin V.V., Zefirov N.S.
Journal of Organic Chemistry,
1994
14.

Lamare V., Archelas A., Faure R., Cesario M., Pascard C., Furstoss R.
Tetrahedron,
1989
15.

Racero J.C., Macías-Sánchez A.J., Hernández-Galán R., Hitchcock P.B., Hanson J.R., Collado I.G.
Journal of Organic Chemistry,
2000
16.

Volobueva A.S., Yarovaya O.I., Kireeva M.V., Borisevich S.S., Kovaleva K.S., Mainagashev I.Y., Gatilov Y.V., Ilyina M.G., Zarubaev V.V., Salakhutdinov N.F.
Molecules,
2021
17.
Paramonova M.P., Snoeck R., Andrei G., Khandazhinskaya A.L., Novikov M.S.
Mendeleev Communications,
2020
18.

Shcherbakov D., Baev D., Kalinin M., Dalinger A., Chirkova V., Belenkaya S., Khvostov A., Krut’ko D., Medved’ko A., Volosnikova E., Sharlaeva E., Shanshin D., Tolstikova T., Yarovaya O., Maksyutov R., et. al.
ACS Medicinal Chemistry Letters,
2021
19.

Abdelwhab E.M., Veits J., Mettenleiter T.C.
Virus Research,
2017
20.

Zarubaev V.V., Pushkina E.A., Borisevich S.S., Galochkina A.V., Garshinina A.V., Shtro A.A., Egorova A.A., Sokolova A.S., Khursan S.L., Yarovaya O.I., Salakhutdinov N.F.
Virology,
2018
21.
10.1016/j.mencom.2022.09.013_b0105
Borisevich
J. Biomol. Struct. Dyn.,
2021
22.
10.1016/j.mencom.2022.09.013_b0110
Gamblin
Science,
1838
23.
G. M. Sheldrick, SADABS, Program for Area Detector Adsorption Correction, University of Göttingen, Göttingen, 1996
24.
G. M. Sheldrick, SHELX-97, Programs for Crystal Structure Analysis, University of Göttingen, Göttingen, 199