Keywords
alkylation
antibacterial agents
biocides
biological activity
pyridinium compounds
quaternary bis-ammonium compounds
Abstract
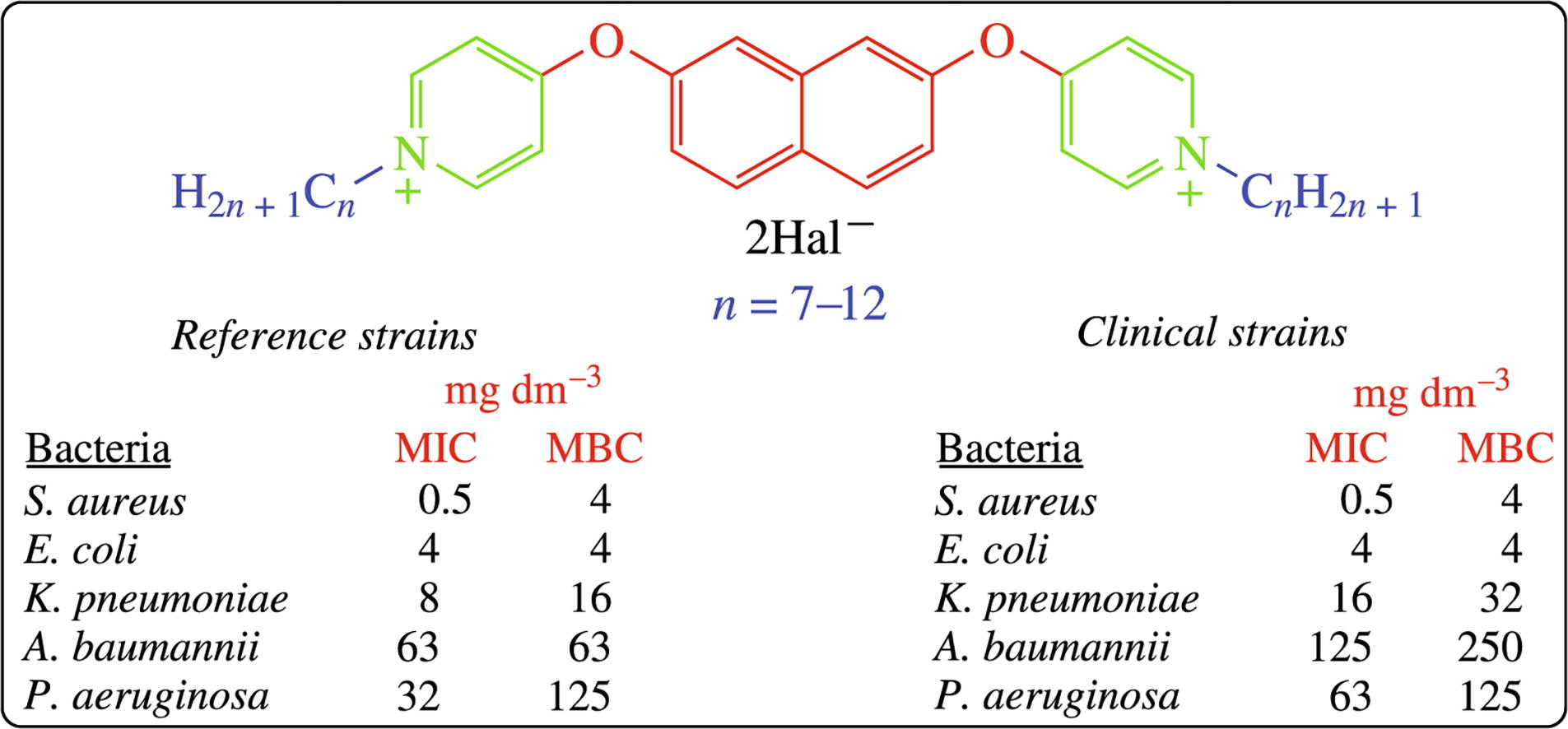
Novel para-substituted bis-pyridinium compounds containing 2,7-dioxynaphthalene spacer were synthesized in two simple steps from the corresponding dihydroxy-naphthalene. The microbiological study on five reference (E. coli ATCC 25922, K. pneumoniae ATCC 70060, S. aureus ATCC 43300, P. aeruginosa ATCC27853, and A. baumannii ATCC 15308) and five clinical (E. coli B-3421/19,K. pneumoniae B-2523/18, S. aureus B-8648, P. aeruginosa B-2099/18 and A. baumannii B-2926/18) bacterial strains showed promising range of antibacterial properties for these biocides, compared to modern sanitizers.
References
1.

Bailey D.M., DeGrazia C.G., Hoff S.J., Schulenberg P.L., O'Connor J.R., Paris D.A., Slee A.M.
Journal of Medicinal Chemistry,
1984
2.

Kourai H., Yabuhara T., Shirai A., Maeda T., Nagamune H.
European Journal of Medicinal Chemistry,
2006
3.

Grenier M.C., Davis R.W., Wilson-Henjum K.L., LaDow J.E., Black J.W., Caran K.L., Seifert K., Minbiole K.P.
Bioorganic and Medicinal Chemistry Letters,
2012
4.

Ator L.E., Jennings M.C., McGettigan A.R., Paul J.J., Wuest W.M., Minbiole K.P.
Bioorganic and Medicinal Chemistry Letters,
2014
5.

Murakami K., Yumoto H., Murakami A., Amoh T., Viducic D., Hirota K., Tabata A., Nagamune H., Kourai H., Matsuo T., Miyake Y.
Journal of Applied Microbiology,
2017
6.

Obando D., Koda Y., Pantarat N., Lev S., Zuo X., Bijosono Oei J., Widmer F., Djordjevic J.T., Sorrell T.C., Jolliffe K.A.
ChemMedChem,
2018
7.

Hübner N.-., Siebert J., Kramer A.
Skin Pharmacology and Physiology,
2010
8.

Amalaradjou M., Venkitanarayanan K.
Pathogens,
2014
9.

Krasowski G., Junka A., Paleczny J., Czajkowska J., Makomaska-Szaroszyk E., Chodaczek G., Majkowski M., Migdał P., Fijałkowski K., Kowalska-Krochmal B., Bartoszewicz M.
Membranes,
2021
10.

Loose M., Naber K.G., Purcell L., Wirth M.P., Wagenlehner F.M.
Urologia Internationalis,
2021
11.

Shepherd M.J., Moore G., Wand M.E., Sutton J.M., Bock L.J.
Journal of Hospital Infection,
2018
12.
10.1016/j.mencom.2022.09.012_b0060
Jennings
Dis.,
2015
13.

Sowmiah S., Esperança J.M., Rebelo L.P., Afonso C.A.
Organic Chemistry Frontiers,
2018
14.

Obłąk E., Piecuch A., Rewak-Soroczyńska J., Paluch E.
Applied Microbiology and Biotechnology,
2018
15.

Bureš F.
Topics in Current Chemistry,
2019
16.

Vereshchagin A.N., Frolov N.A., Egorova K.S., Seitkalieva M.M., Ananikov V.P.
International Journal of Molecular Sciences,
2021
17.

Vereshchagin A.N., Gordeeva A.M., Frolov N.A., Proshin P.I., Hansford K.A., Egorov M.P.
European Journal of Organic Chemistry,
2019
18.
Vereshchagin A.N., Frolov N.A., Konyuhova V.Y., Hansford K.A., Egorov M.P.
Mendeleev Communications,
2019
19.
Vereshchagin A.N., Frolov N.A., Konyuhova V.Y., Dorofeeva E.O., Hansford K.A., Egorov M.P.
Mendeleev Communications,
2020
20.
Vereshchagin A.N., Frolov N.A., Pakina A.S., Hansford K.A., Egorov M.P.
Mendeleev Communications,
2020
21.

Vereshchagin A.N., Frolov N.A., Konyuhova V.Y., Kapelistaya E.A., Hansford K.A., Egorov M.P.
RSC Advances,
2021
22.

Frolov N.A., Fedoseeva K.A., Hansford K.A., Vereshchagin A.N.
ChemMedChem,
2021
23.

Frolov N., Detusheva E., Fursova N., Ostashevskaya I., Vereshchagin A.
Pharmaceuticals,
2022
24.
10.1016/j.mencom.2022.09.012_b0120
Detusheva
Clin. Microbiol. Antimicrob. Chemother.,
2015