Keywords
[3+2] cycloaddition
GPCR privileged ligands
photoaffinity labeling
silver(t) catalysis
tetrazoles
α-diazo acetamides
Abstract
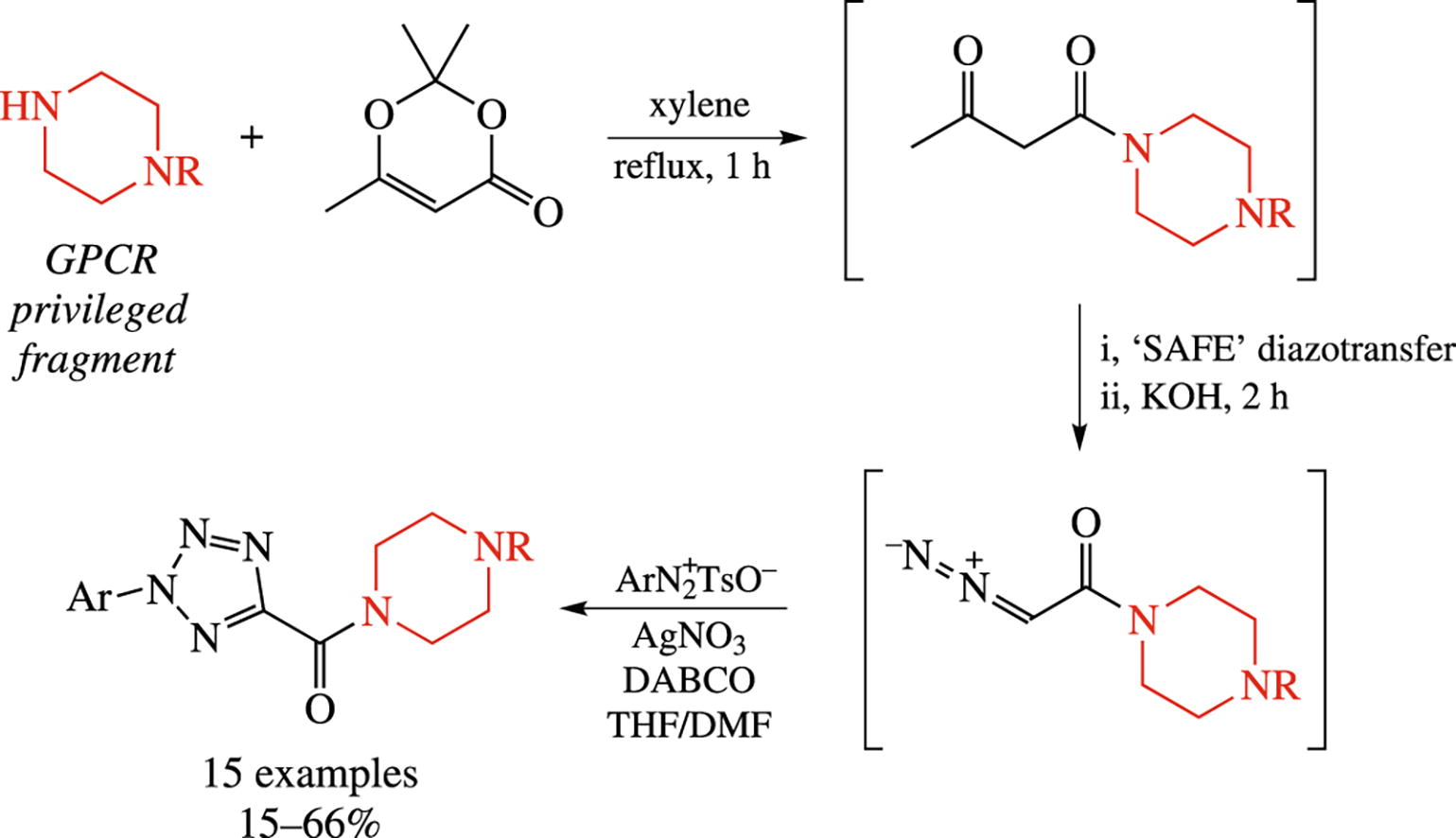
A four-step approach to 2-aryl-2H-tetrazole-5-carboxamides bearing GPCR-focused N-substituted piperazine residues involves ‘SAFE’ diazotransfer onto 1-(piperazin-1-yl)butane-1,3-diones followed by [3+2] cycloaddition of arenediazonium cations at the intermediate ααα-diazo acetamides. The compounds prepared are intended for photoaffinity labeling of aminergic G-protein coupled receptors.
References
1.

Bunnage M.E., Gilbert A.M., Jones L.H., Hett E.C.
Nature Chemical Biology,
2015
2.

Ruoho A.E., Kiefer H., Roeder P.E., Singer S.J.
Proceedings of the National Academy of Sciences of the United States of America,
1973
3.

Smith E., Collins I.
Future Medicinal Chemistry,
2015
4.

Park H., Koo J.Y., Srikanth Y.V., Oh S., Lee J., Park J., Park S.B.
Chemical Communications,
2016
5.

Tamura T., Tsukiji S., Hamachi I.
Journal of the American Chemical Society,
2012
6.

Burton N.R., Kim P., Backus K.M.
Organic and Biomolecular Chemistry,
2021
7.

FLEET G.W., PORTER R.R., KNOWLES J.R.
Nature,
1969
8.

Smith R.A., Knowles J.R.
Journal of the American Chemical Society,
1973
9.

Galardy R.E., Craig L.C., Jamieson J.D., Printz M.P.
Journal of Biological Chemistry,
1974
10.

Zhong Y., Yasuda N., Li H., McLaughlin M., Tschaen D.
Topics in Current Chemistry,
2016
11.

Evaluation of α-Pyrones and Pyrimidones as Photoaffinity Probes for Affinity-Based Protein Profiling
Battenberg O.A., Nodwell M.B., Sieber S.A.
Journal of Organic Chemistry,
2011
12.

Herner A., Marjanovic J., Lewandowski T.M., Marin V., Patterson M., Miesbauer L., Ready D., Williams J., Vasudevan A., Lin Q.
Journal of the American Chemical Society,
2016
13.
![[3+2]-Cycloaddition of α-Diazocarbonyl Compounds with Arenediazonium Salts Catalyzed by Silver Nitrate Delivers 2,5-Disubstituted Tetrazoles](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Chuprun S., Dar’in D., Kantin G., Krasavin M.
Synthesis,
2019
14.
10.1016/j.mencom.2022.09.011_b0070
Levashova
Eur. J. Org. Chem.,
2020
15.
Sapegin A.V., Peshkov A.A., Kanov E.V., Gainetdinov R.R., Duszyńska B., Bojarski A.J., Krasavin M.
Mendeleev Communications,
2021
16.
Levashova E., Firsov A., Bakulina O., Peshkov A., Kanov E., Gainetdinov R.R., Krasavin M.
Mendeleev Communications,
2021
17.

Bondensgaard K., Ankersen M., Thøgersen H., Hansen B.S., Wulff B.S., Bywater R.P.
Journal of Medicinal Chemistry,
2004
18.

Briner K., Collado I., Fisher M.J., García-Paredes C., Husain S., Kuklish S.L., Mateo A.I., O’Brien T.P., Ornstein P.L., Zgombick J., de Frutos Ó.
Bioorganic and Medicinal Chemistry Letters,
2006
19.

Christopher J.A., Brown J., Doré A.S., Errey J.C., Koglin M., Marshall F.H., Myszka D.G., Rich R.L., Tate C.G., Tehan B., Warne T., Congreve M.
Journal of Medicinal Chemistry,
2013
20.
21.

Dar’in D., Kantin G., Krasavin M.
Chemical Communications,
2019
22.
Shevalev R.M., Zhmurov P.A., Dar’in D.V., Krasavin M.
Mendeleev Communications,
2020