Keywords
ambient heating
ethylene
low temperature reactions
nitro compounds
nitrogen tetroxide
pressure effect
Abstract
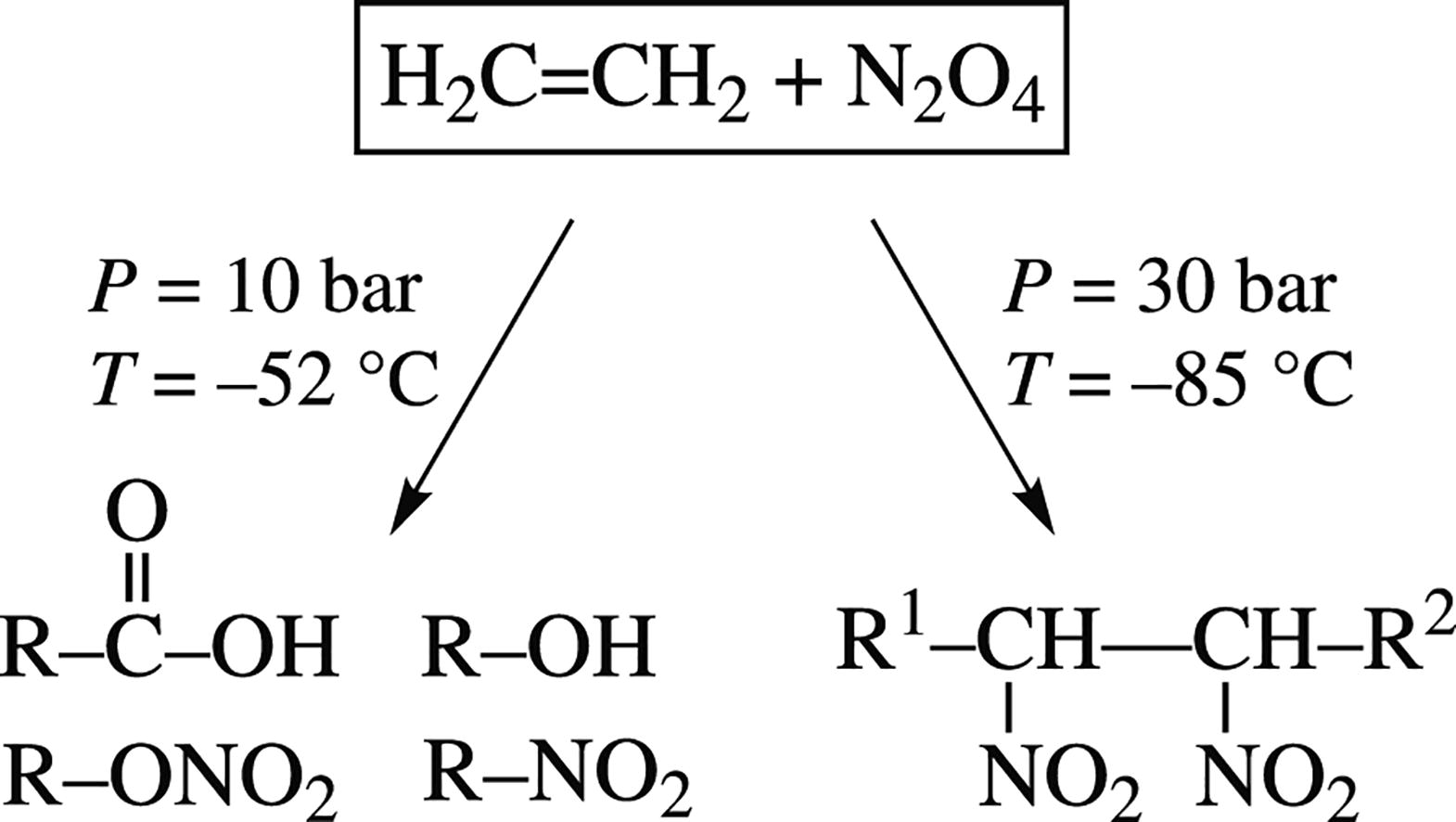
Calorimetric monitoring of the autoclave reaction N2O4 + C2H4 at –85 to +10 °C under argon pressure 10–30 bar revealed that the exothermic chemical reaction started at temperatures above –52 °C at 10 bar, whereas an intensive exothermic reaction started at –85 °C and pressure of 30 bar. IR study showed that oligo/polynitroethylene was formed at 30 bar, while carbonyl and hydroxy compound as well as nitrate R–ONO2 formation occurred upon processing at 10 bar.
References
1.
10.1016/j.mencom.2022.09.009_b0005
Higman
Gasification, Elsevier/Gulf Professional Publishing,
2003
2.
10.1016/j.mencom.2022.09.009_b0010
Zhu
J. Nat. Gas Chem.,
2004
3.
10.1016/j.mencom.2022.09.009_b0015
Fainshtein
Intermed. Eng.,
2008
4.
10.1016/j.mencom.2022.09.009_b0020
Schnobel
12AIChE – 2012 AIChE Spring Meeting and 8th Global Congress on Process Safety,
2012
5.
W. Hack, R. Copeland and D. Hines, 21st AIChE Ethylene Producers Conference, Tampa, 2009
6.

Riebsomer J.L.
Chemical Reviews,
1945
7.
10.1016/j.mencom.2022.09.009_b0035
Rozlovsky
Dokl. Akad. Nauk SSSR,
1970
8.
10.1016/j.mencom.2022.09.009_b0040
Rozlovsky
Dokl. Akad. Nauk SSSR,
1970
9.
10.1016/j.mencom.2022.09.009_b0045
Papisova
Dokl. Akad. Nauk SSSR,
1966
10.

Giménez-López J., Alzueta M.U., Rasmussen C.T., Marshall P., Glarborg P.
Proceedings of the Combustion Institute,
2011
11.

Stepanov A.V., Veselovsky V.V.
Russian Chemical Reviews,
2003
12.
10.1016/j.mencom.2022.09.009_b0060
Mikhaylovskaya
Dokl. Akad. Nauk SSSR,
1970
13.

Deng F., Zhang Y., Sun W., Huang W., Zhao Q., Qin X., Yang F., Huang Z.
Proceedings of the Combustion Institute,
2019
14.

Loison J.C., Dedonder-Lardeux C., Jouvet C., Solgadi D.
The Journal of Physical Chemistry,
1991
15.

Loison J.C., Dedonder-Lardeux C., Jouvet C., Solgadi D.
Faraday Discussions,
1994
16.

Liu J., Zhou W., Chambreau S.D., Vaghjiani G.L.
Journal of Physical Chemistry B,
2020
17.

Nitrate radicals and biogenic volatile organic compounds: oxidation, mechanisms, and organic aerosol
Ng N.L., Brown S.S., Archibald A.T., Atlas E., Cohen R.C., Crowley J.N., Day D.A., Donahue N.M., Fry J.L., Fuchs H., Griffin R.J., Guzman M.I., Herrmann H., Hodzic A., Iinuma Y., et. al.
Atmospheric Chemistry and Physics,
2017
18.

Nguyen T.L., Park J., Lee K., Song K., Barker J.R.
Journal of Physical Chemistry A,
2011
19.

Vereecken L., Francisco J.S.
Chemical Society Reviews,
2012
20.

Sarafian H.
American Journal of Computational Mathematics,
2021
21.

Gray P., Yoffe A.D.
Chemical Reviews,
1955
22.
10.1016/j.mencom.2022.09.009_b0110
Structure Determination of Organic Compounds: Tables of Spectral Data,
2000