Keywords
1,2,4-triazolin-5-ylidene ligands
anticancer activities
NMR probe
palladium-NHC
palladium(II) complexes
Abstract
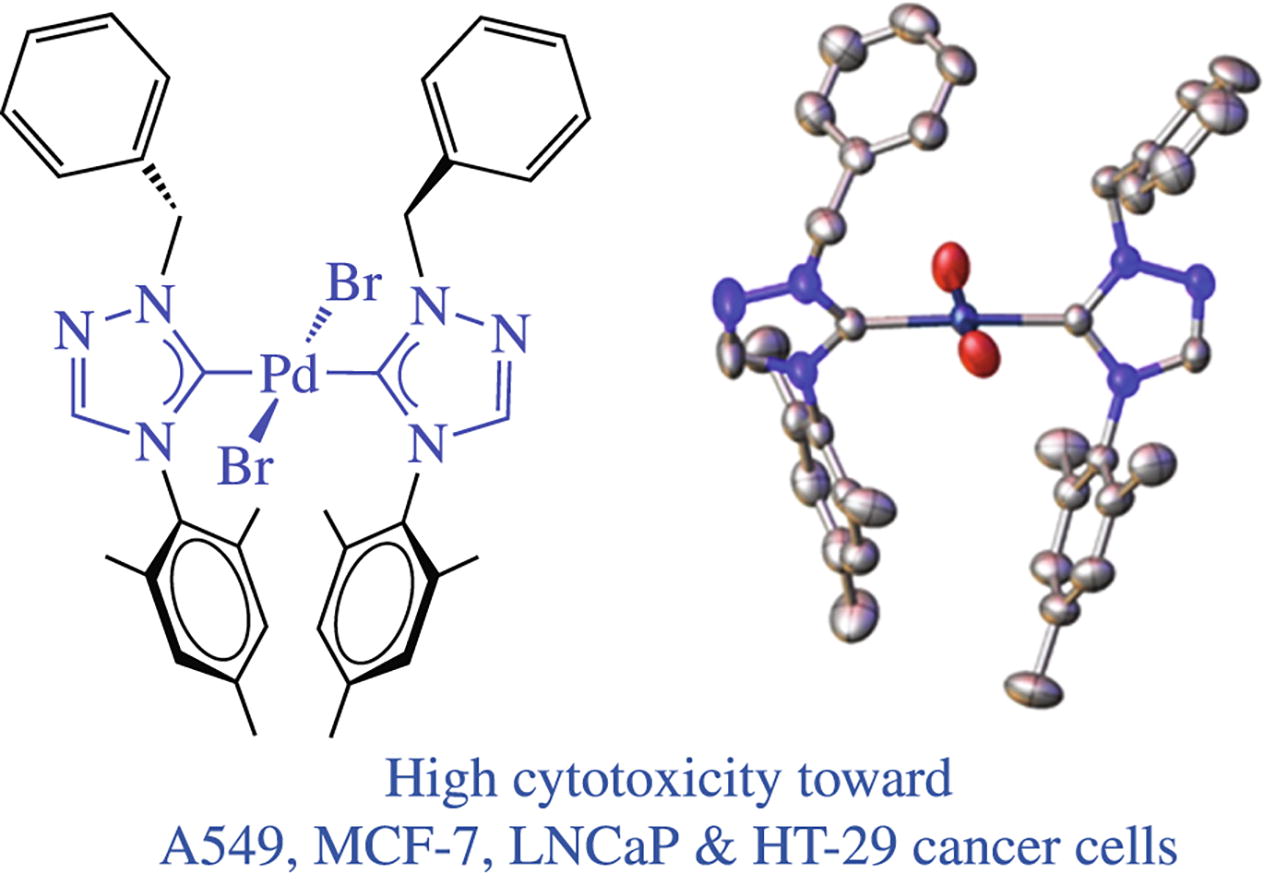
Two novel palladium(ii) complexes, trans-[PdBr2(tz-Mes)2] and trans-[PdBr2(tz-Dipp)2] featuring less explored 1,2,4-triazole-derived N-heterocyclic carbene (tz) have been synthesized. In solution, they exist both as trans-syn and trans-anti rotamers while their syn/anti ratios can be determined by 1H NMR harnessing the built-in aromatic rings as NMR probes. Complex trans-[PdBr2(tz-Mes)2] is highly cytotoxic toward A549, MCF-7, LNCaP and HT-29 cancerous cells.
References
1.

Bourissou D., Guerret O., Gabbaï F.P., Bertrand G.
Chemical Reviews,
1999
2.

Peris E.
Chemical Reviews,
2017
3.

Hopkinson M.N., Richter C., Schedler M., Glorius F.
Nature,
2014
4.

Smith C.A., Narouz M.R., Lummis P.A., Singh I., Nazemi A., Li C., Crudden C.M.
Chemical Reviews,
2019
5.

Arduengo A.J., Harlow R.L., Kline M.
Journal of the American Chemical Society,
1991
6.

Flanigan D.M., Romanov-Michailidis F., White N.A., Rovis T.
Chemical Reviews,
2015
7.

Huynh H.V.
Chemical Reviews,
2018
8.
10.1016/j.mencom.2022.09.008_b0040
Lappert
J. Chem. Soc., Dalton Trans.,
1977
9.

Dorta R., Stevens E.D., Hoff C.D., Nolan S.P.
Journal of the American Chemical Society,
2003
10.

Kantchev E., O'Brien C., Organ M.
Angewandte Chemie - International Edition,
2007
11.

Fortman G.C., Nolan S.P.
Chemical Society Reviews,
2011
12.
Rzhevskiy S.A., Topchiy M.A., Bogachev V.N., Ageshina A.A., Minaeva L.I., Sterligov G.K., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2021
13.
Rzhevskiy S.A., Topchiy M.A., Golenko Y.D., Gribanov P.S., Sterligov G.K., Kirilenko N.Y., Ageshina A.A., Bermeshev M.V., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2020
14.

Hussaini S.Y., Haque R.A., Razali M.R.
Journal of Organometallic Chemistry,
2019
15.
10.1016/j.mencom.2022.09.008_b0075
Scattolin
Cell Rep. Phys. Sci.,
2021
16.

Kumar A., Naaz A., Prakasham A.P., Gangwar M.K., Butcher R.J., Panda D., Ghosh P.
ACS Omega,
2017
17.

Canovese L., Santo C., Scattolin T., Visentin F., Bertolasi V.
Journal of Organometallic Chemistry,
2015
18.

Fong T.T., Lok C., Chung C.Y., Fung Y.E., Chow P., Wan P., Che C.
Angewandte Chemie - International Edition,
2016
19.

Bernd M.A., Bauer E.B., Oberkofler J., Bauer A., Reich R.M., Kühn F.E.
Dalton Transactions,
2020
20.

Nguyen V.H., Ibrahim M.B., Mansour W.W., El Ali B.M., Huynh H.V.
Organometallics,
2017
21.

Nguyen V.H., Dang T.T., Nguyen H.H., Huynh H.V.
Organometallics,
2020
22.
10.1016/j.mencom.2022.09.008_b0110
Barr
PLoS ONE,
2013
23.

Wawruszak A., Luszczki J.J., Grabarska A., Gumbarewicz E., Dmoszynska-Graniczka M., Polberg K., Stepulak A.
PLoS ONE,
2015
24.
10.1016/j.mencom.2022.09.008_b0120
Štarha
PLoS ONE,
2015
25.

Dhar S., Gu F.X., Langer R., Farokhzad O.C., Lippard S.J.
Proceedings of the National Academy of Sciences of the United States of America,
2008
26.
10.1016/j.mencom.2022.09.008_b0130
Leong
PLoS ONE,
2016
27.

Ratzon E., Najajreh Y., Salem R., Khamaisie H., Ruthardt M., Mahajna J.
BMC Cancer,
2016