Keywords
Abstract
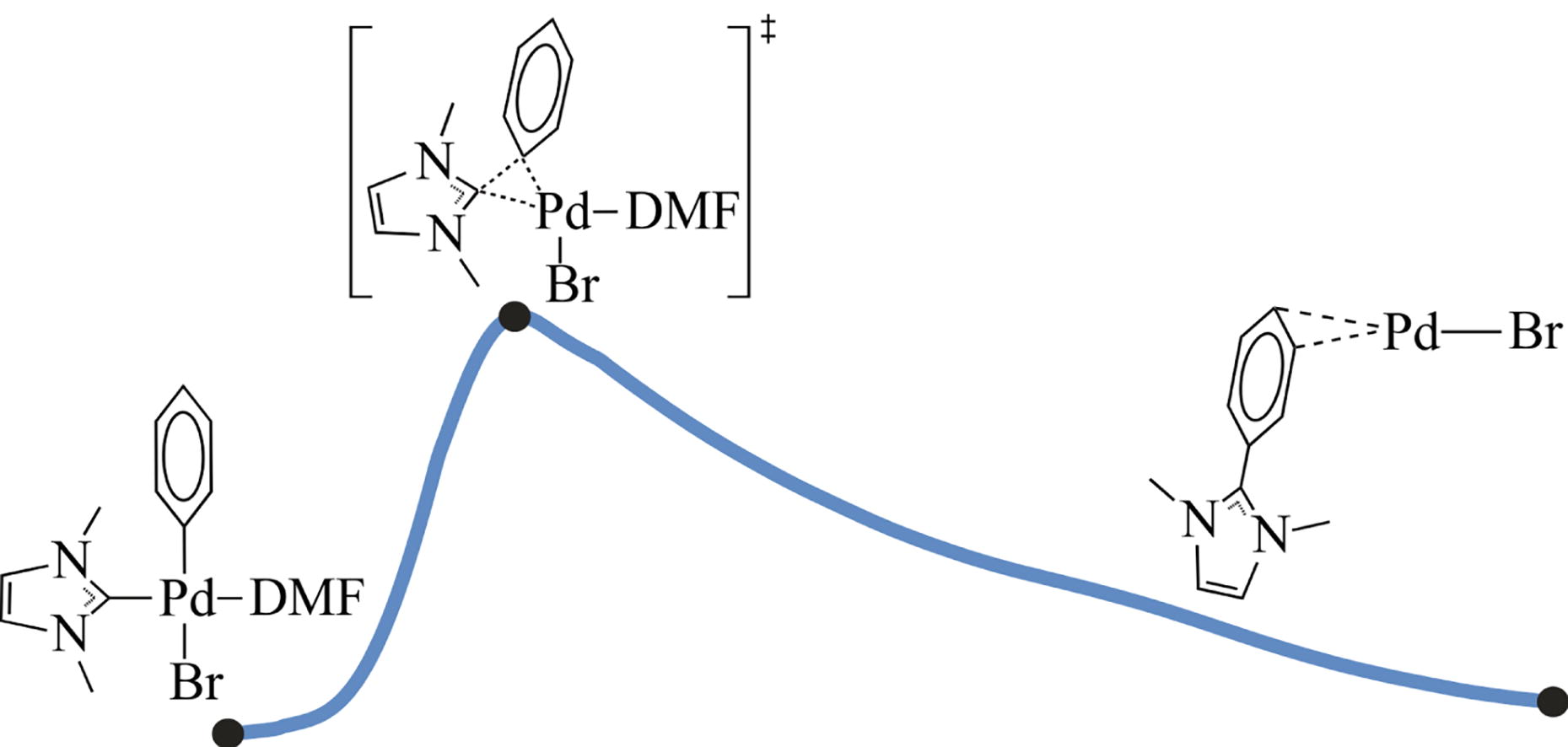
C–C coupling reactions are of great importance in metal-catalyzed synthetic transformations. Reductive elimination of two carbon centers is the key stage, which takes place in the metal coordination sphere. In the present study, we provide a detailed analysis of nonclassical R–NHC coupling in the model (NHC)Pdii(Ph)(X)(Solv) complex, which is a representative intermediate of the Mizoroki–Heck and cross-coupling reactions. This C–C bond formation stage proceeds as Ph ligand movement and insertion into the Pd–NHC bond, rather than classical C–C coupling. Based on the analysis by the quantum theory of atoms in molecules (QTAIM) of the reaction path structures, the atomic rearrangements and alterations in the electronic system during the R–NHC coupling process were characterized in detail.
References