Keywords
albumin
fluorescence
hydroxy-substituted porphyrin
photooxidation
ROS
UV-VIS
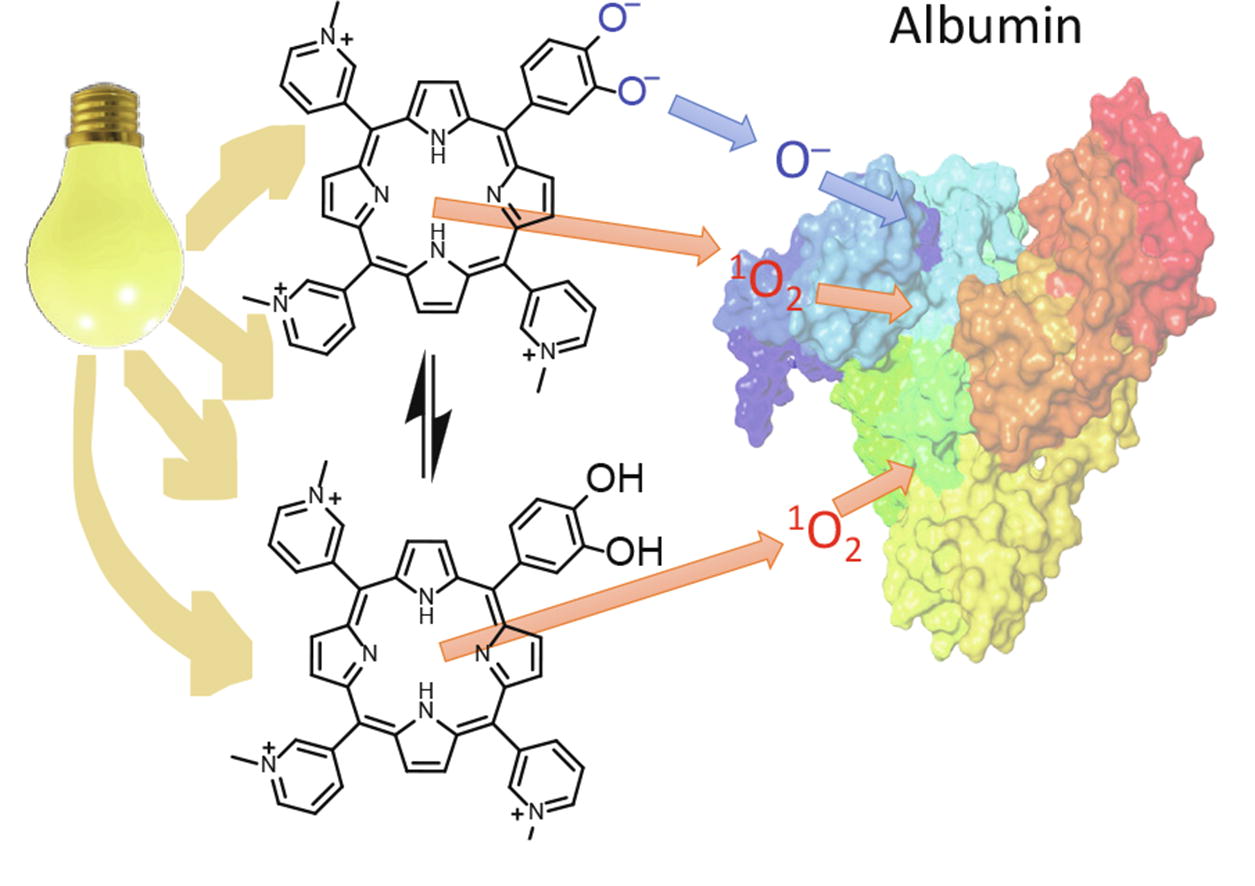
Abstract
The observed rate constants of the photoinduced oxidation of albumin increased by a factor of 1.5–2 upon deprotonation of the peripheral hydroxyl groups of 5-(3,4-dihydroxyphenyl)-10,15,20-tris(N-methylpyridinium-3-yl)porphyrin triiodide compared to its phenolic form.
References
1.

Ma L., Moan J., Berg K.
International Journal of Cancer,
1994
2.

Fakayode O.J., Kruger C.A., Songca S.P., Abrahamse H., Oluwafemi O.S.
Materials Science and Engineering C,
2018
3.

Ma L.W., Moan J., Berg K.
Lasers in Medical Science,
1994
4.

Moore C.M., Nathan T.R., Lees W.R., Mosse C.A., Freeman A., Emberton M., Bown S.G.
Lasers in Surgery and Medicine,
2006
5.

Zakharko M.A., Panchenko P.A., Zarezin D.P., Nenajdenko V.G., Pritmov D.A., Grin M.A., Mironov A.F., Fedorova O.A.
Russian Chemical Bulletin,
2020
6.

Lebedeva N.S., Popova T.E., Semeykin A.S.
Journal of Solution Chemistry,
2010
7.

Kolyada M.N., Osipova V.P., Berberova N.T., Shpakovsky D.B., Milaeva E.R.
Chemistry of Heterocyclic Compounds,
2021
8.

Guo H., Jiang J., Shi Y., Wang Y., Liu J., Dong S.
Journal of Physical Chemistry B,
2004
9.

Wang C., Wamser C.C.
Journal of Physical Chemistry A,
2014
10.

Otvagin V.F., Nyuchev A.V., Kuzmina N.S., Grishin I.D., Gavryushin A.E., Romanenko Y.V., Koifman O.I., Belykh D.V., Peskova N.N., Shilyagina N.Y., Balalaeva I.V., Fedorov A.Y.
European Journal of Medicinal Chemistry,
2018
11.

Cook M.J., Katritzky A.R., Linda P., Tack R.D.
Tetrahedron Letters,
1972
12.

Raczyńska E.D., Kosińska W., Ośmiałowski B., Gawinecki R.
Chemical Reviews,
2005
13.
Lebedeva N.S., Yurina E.S., Gubarev Y.A., Kiselev A.N., Syrbu S.A.
Mendeleev Communications,
2020
14.

Topal S.Z., Ongun M.Z., Önal E., Ertekin K., Hirel C.
Dyes and Pigments,
2017
15.

Conradie J., Wamser C.C., Ghosh A.
Journal of Physical Chemistry A,
2021
16.

Sasano Y., Yasuda N., Maeda H.
Dalton Transactions,
2017
17.

Guo H., Jiang J., Shi Y., Wang Y., Wang Y., Dong S.
Journal of Physical Chemistry B,
2005
18.

Spectroscopic characterization of free-base hydroxy(arylethynyl)porphyrins in acidic and basic media
Evens K.K., Splan K.E.
Journal of Porphyrins and Phthalocyanines,
2017
19.

Wu Y., Han Z., Wei L., Sun H., Wang T., Chen J., Zhang R., Lu X.
Analytical Chemistry,
2020
20.
10.1016/j.mencom.2022.07.040_b0100
Chaikovskaya
Russ. J. Phys. Chem.,
2000
21.

Venkatesan M., Mandal H., Chakali M., Bangal P.R.
Journal of Physical Chemistry C,
2019
22.
Lebedeva N.S., Yurina E.S., Gubarev Y.A., Guseinov S.S.
Mendeleev Communications,
2020
23.

Molecular mechanisms causing albumin aggregation. The main role of the porphyrins of the blood group
Lebedeva N.S., Yurina E.S., Gubarev Y.A., Koifman O.I.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2021
24.

Giovannetti R., Alibabaei L., Petetta L.
Journal of Photochemistry and Photobiology A: Chemistry,
2010
25.

Zurita A., Duran A., Ribó J.M., El-Hachemi Z., Crusats J.
RSC Advances,
2017
26.
10.1016/j.mencom.2022.07.040_b0130
Kopp
Am. Mineral.,
1967
27.

Carter D.C., Ho J.X.
Advances in Protein Chemistry,
1994
28.

Agazzi M.L., Spesia M.B., Gsponer N.S., Milanesio M.E., Durantini E.N.
Journal of Photochemistry and Photobiology A: Chemistry,
2015