Keywords
13-dipolar cycloaddition
2-azabicyclo[2.2.1]heptanes
hydroxyimoyl halides
isoxazolines
nitrile oxides
norbornene
Abstract
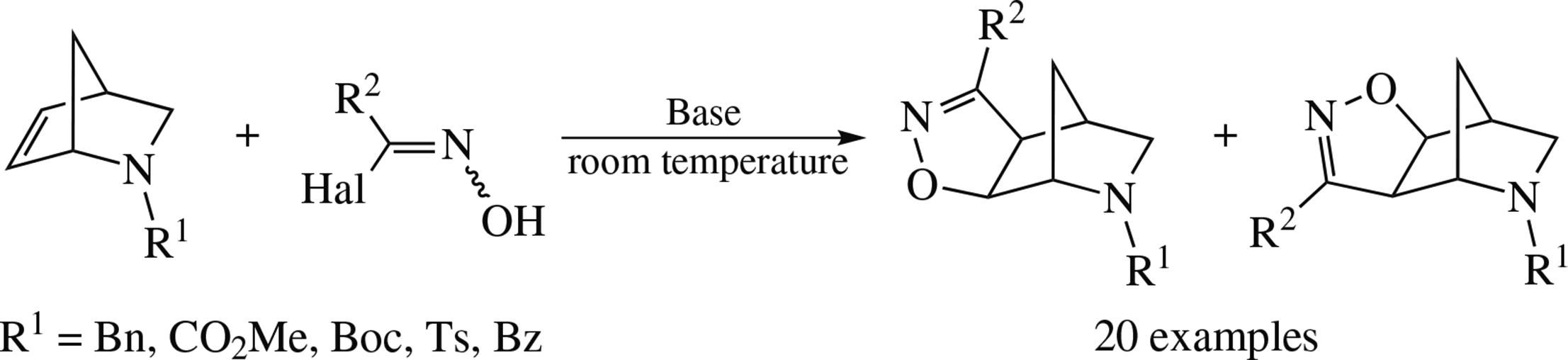
The reaction of 2-azanorbornene derivatives with nitrile oxides (generated in situ by dehydrohalogenation of N-hydroxyimoyl halides) affords two regioisomers with the exo-arrangement of the isoxazoline ring.
References
1.

Wojaczyńska E., Wojaczyński J., Kleniewska K., Dorsz M., Olszewski T.K.
Organic and Biomolecular Chemistry,
2015
2.

Kozikowski A.P.
Accounts of Chemical Research,
1984
3.

Litvinovskaya R.P., Khripach V.A.
Russian Chemical Reviews,
2001
4.

Huisgen R., Christl M.
Angewandte Chemie,
1967
5.

Huisgen R., Christl M.
Chemische Berichte,
1973
6.

De Sarlo(rk) F., Brandi A., Guarna A., Goti A., Corezzi S.
Tetrahedron Letters,
1983
7.

de Sarlo F., Guarna A., Brandi A., Goti A.
Tetrahedron,
1985
8.

Caldirola P., Ciancaglione M., De Amici M., De Micheli C.
Tetrahedron Letters,
1986
9.
![Exceptional reactivity of the bicyclo[2.2.1]heptene double bond](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Huisgen R., Ooms P.H., Mingin M., Allinger N.L.
Journal of the American Chemical Society,
1980
10.

Lee G.A.
Synthesis,
1982
11.

Cunico R.F., Bedell L.
Journal of Organic Chemistry,
1983
12.
10.1016/j.mencom.2022.07.038_b0060
Cecchi
Synlett,
2007
13.

Cecchi L., De Sarlo F., Machetti F.
Chemistry - A European Journal,
2008
14.

Pigou P.E., Stirling C.J.
Journal of the Chemical Society Perkin Transactions 2,
1988
15.

Muri D., Bode J.W., Carreira E.M.
Organic Letters,
2000
16.
![[2 + 2 + 1] Cycloaddition of N-tosylhydrazones, tert-butyl nitrite and alkenes: a general and practical access to isoxazolines](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Ma L., Jin F., Cheng X., Tao S., Jiang G., Li X., Yang J., Bao X., Wan X.
Chemical Science,
2021
17.

Mendelsohn B.A., Lee S., Kim S., Teyssier F., Aulakh V.S., Ciufolini M.A.
Organic Letters,
2009
18.

Liang H., Ciufolini M.A.
Tetrahedron,
2010
19.

Jen T., Mendelsohn B.A., Ciufolini M.A.
Journal of Organic Chemistry,
2010
20.

Zlatopolskiy B.D., Kandler R., Kobus D., Mottaghy F.M., Neumaier B.
Chemical Communications,
2012
21.

Xiang C., Li T., Yan J.
Synthetic Communications,
2013
22.

Molteni G., Del Buttero P.
Tetrahedron,
2011
23.

Tranmer G.K., Tam W.
Organic Letters,
2002
24.

Mayo P., Hecnar T., Tam W.
Tetrahedron,
2001
25.
10.1016/j.mencom.2022.07.038_b0125
Gucma
J. Braz. Chem. Soc.,
2016
26.

Pinto A., Tamborini L., Pennacchietti E., Coluccia A., Silvestri R., Cullia G., De Micheli C., Conti P., De Biase D.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2015
27.

P. Curran D., A. Gothe S., Choi S.
Heterocycles,
1993
28.

Chiu D., Lin T., Huang W., Cheng T., Tsai K., Fang J.
Organic and Biomolecular Chemistry,
2017
29.

Chen C., Chiu T., Chen Y., Fang J.
Tetrahedron,
2019
30.

Quadrelli P., Piccanello A., Martinez N.V., Bovio B., Mella M., Caramella P.
Tetrahedron,
2006
31.

Quadrelli P., Memeo M., Mantione D., Bovio B.
Synthesis,
2011
32.
10.1016/j.mencom.2022.07.038_b0160
Khmelnitsky
Khimiya furoksanov: stroenie i svoistva (Chemistry of Furoxans: Structure and Properties),
1996
33.
10.1016/j.mencom.2022.07.038_b0165
Gordon
The Chemist’s Companion,
1972
34.
O. A. Reutov, A. L. Kurts and K. P. Butin, Organicheskaya khimiya (Organic Chemistry), Binom. Laboratoriya znanii, Moscow, 2004, vol. 3 (in Russian).