Keywords
allyl bromide
cyrene
ginkgolides
intramolecular cationic cyclization
trichothecene sesquiterpenoids
Abstract
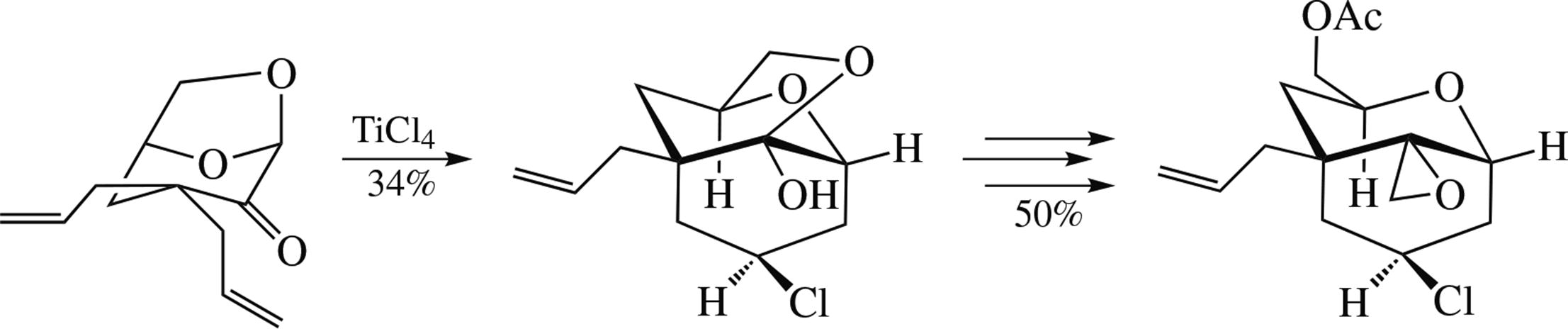
The reaction of cyrene with allyl bromide in THF in the presence of ButOK gave the α,α-diallyl derivative of cyrene in 50% yield. This derivative smoothly undergoes iodo- cyclization on the keto group that is converted into a ketal center to give annulated bis-tetrahydrofuran; intramolecular cationic cyclization in the presence of SnCl4 or TiCl4 results in 2-oxabicyclo[3.3.1]nonan-9-ones. The prospects of using these reactions towards the synthesis of trichothecene sesquiterpenoids modified in the central moiety were assessed.
References
1.
(a) M. B. Comba, Y. Tsai, A. M. Sarotti, M. I. Mangione, A. G. Suárez and R. A. Spanevello, Eur. J. Org. Chem., 2018, 590; (b) D. D. Ward and F. Shafizadeh, Carbohydr. Res., 1981, 95, 155; (c) F. Shafizadeh, M. G. Essig and D. D. Ward, Carbohydr. Res., 1983, 114, 71; (d) P. Bhate and D. Horton, Carbohydr. Res., 1983, 122, 189; (e) I. Minoru, F. Naoki and T. Goto, Chem. Lett., 1985, 14, 71; (f) M. Isobe, N. Fukami, T. Nishikava and T. Goto, Heterocycles, 1987, 25, 521; (g) M. S. Miftakhov, I. N. Gaisina and F. A. Valeev, Russ. Chem. Bull., 1996, 45, 1942 (Izv. Akad. Nauk, Ser. Khim., 1996, 2047); (h) A. M. Sarotti, A. G. Suárez and R. A. Spanevello, Tetrahedron Lett., 2011, 52, 3116; (i) F. A. Valeev, I. N. Gaisina and M. S. Miftakhov, Russ. Chem. Bull., 1996, 45, 2453 (Izv. Akad. Nauk, Ser. Khim., 1996, 2584); (j) M. A. Zurita, A. Avila, R. A. Spanevello, A. G. Suárez and A. M. Sarotti, Carbohydr. Res., 2015, 402, 67; (k) I. M. Biktagirov, L. Kh. Faizullina, S. M. Salikhov, F. Z. Galin and F. A. Valeev, Mendeleev Commun., 2017, 27, 237; (l) B. T. Sharipov, A. A. Pershin and F. A. Valeev, Mendeleev Commun., 2017, 27, 119; (m) L. Kh. Faizullina, A. R. Tagirov, S. M. Salikhov and F. A. Valeev, Mendeleev Commun., 2022, 32, 100.
2.
10.1016/j.mencom.2022.07.037_b0010
Sharipov
Butlerovskie Soobshcheniya,
2011
3.

Sharipov B.T., Krasnoslobodtseva O.Y., Spirikhin L.V., Valeev F.A.
Russian Journal of Organic Chemistry,
2010
4.

Biktagirov I.M., Faizullina L.K., Salikhov S.M., Iskakova M.M., Safarov M.G., Galin F.Z., Valeev F.A.
Russian Journal of Organic Chemistry,
2015
5.

Ledingham E.T., Stockton K.P., Greatrex B.W.
Australian Journal of Chemistry,
2017
6.
7.

Structure of bilobalide, a rare tert-butyl containing sesquiterpenoid related to the C20-ginkgolides
Nakanishi K., Habaguchi K., Nakadaira Y., Woods M.C., Maruyama M., Major R.T., Alauddin M., Patel A.R., Weinges K., Baehr W.
Journal of the American Chemical Society,
1971
8.

Corey E.J., Kang M.C.
Journal of the American Chemical Society,
1984
9.
Weinges K., Hepp M., Huber-Patz U., Rodewald H., Irngartinger H.
1986
10.

Snider B.B., Rodini D.J., Karras M., Kirk T.C., Deutsch E.A., Cordova R., Price R.T.
Tetrahedron,
1981
11.

Snider B.B., Rodini D.J., Kirk T.C., Cordova R.
Journal of the American Chemical Society,
1982
12.

Zanardi M.M., Suárez A.G.
Tetrahedron Letters,
2009
13.

Kraus G.A., Thurston J.
Tetrahedron Letters,
1987
14.

Tagirov A.R., Galimova Y.S., Faizullina L.K., Spirikhin L.V., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2017
15.

Hanessian S., Banoub J.
Carbohydrate Research,
1977
16.
(a) A. T. McPhail and G. A. Sim, J. Chem. Soc. C, 1966, 1394; (b) W. O. Godtfredsen, J. F. Grove and C. H. Tamm, Helv. Chim. Acta, 1967, 50, 1666; (c) P. Traxler, W. Zürcher and C. Tamm, Helv. Chim. Acta, 1970, 53, 2071; (d) J. F. Grove, J. Chem. Soc. C, 1970, 378; (e) E. W. Colvin, R. A. Raphael and J. S. Roberts, J. Chem. Soc. D, 1971, 858; (f) E. W. Colvin, S. Malchenko, R. A. Raphael and J. S. Roberts, J. Chem. Soc., Perkin Trans. 1, 1973, 1989; (g) A. J. Pearson and C. W. Ong, J. Am. Chem. Soc., 1981, 103, 6686; (h) G. A. Kraus, B. Roth, K. Frazier and M. Shimagaki, J. Am. Chem. Soc., 1982, 104, 1114; (i) R. H. Schlessinger and R. A. Nugent, J. Am. Chem. Soc., 1982, 104, 1116; (j) B. M. Trost and P. G. McDougal, J. Am. Chem. Soc., 1982, 104, 6110.