Keywords
13-diketone
3-hydroxyindolin-2-one
aldol reaction
isatin
noncatalytic
on water
Abstract
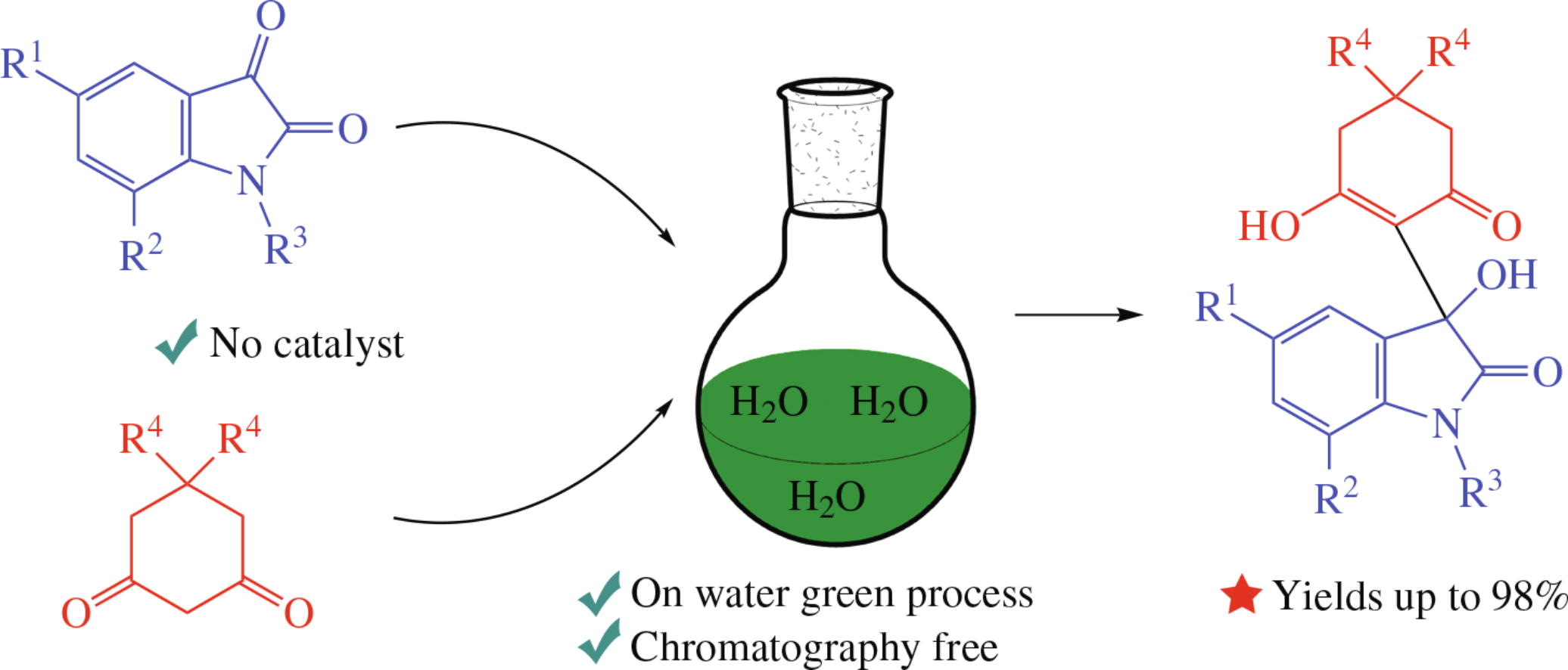
Noncatalytic on water aldol transformation of isatins and cyclic 1,3-diketones results in substituted 3-hydroxy-3-(2-hydroxy-6-oxocyclohex-1-en-1-yl)indolin-2-ones in 87–98% yields. Optimized conditions have been found and a mechanistic rationale for the reaction has been deduced. The new efficient and facile process represents a convenient way to compounds with the 3-hydroxyindolin-2-one and 3-hydroxycyclohex-2-en-1-one moieties.
References
1.

Cortes-Clerget M., Yu J., Kincaid J.R., Walde P., Gallou F., Lipshutz B.H.
Chemical Science,
2021
2.

Simon M., Li C.
Chemical Society Reviews,
2012
3.
Organic Reactions in Water: Principles, Strategies and Applications, ed. U. M. Lindström, Blackwell Publishing, Oxford, 2007.
4.

Narayan S., Muldoon J., Finn M.G., Fokin V.V., Kolb H.C., Sharpless K.B.
Angewandte Chemie - International Edition,
2005
5.

Butler R.N., Coyne A.G., Cunningham W.J., Moloney E.M.
Journal of Organic Chemistry,
2013
6.

Butler R.N., Cunningham W.J., Coyne A.G., Burke L.A.
Journal of the American Chemical Society,
2004
7.

Butler R.N., Coyne A.G.
Organic and Biomolecular Chemistry,
2016
8.

9.

10.

Wender P.A.
Natural Product Reports,
2014
11.

Nicolaou K.C., Montagnon T., Vassilikogiannakis G., Mathison C.J.
Journal of the American Chemical Society,
2005
12.

Tsukano C., Siegel D., Danishefsky S.
Angewandte Chemie,
2007
13.

Rohr K., Mahrwald R.
Advanced Synthesis and Catalysis,
2008
14.

Mato R., Reyes E., Carrillo L., Uria U., Prieto L., Manzano R., Vicario J.L.
Chemical Communications,
2020
15.
10.1016/j.mencom.2022.07.036_b0075
Elinson
Chem. Rec.,
1950
16.
10.1016/j.mencom.2022.07.036_b0080
Henry
Seances Acad. Sci.,
1895
17.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Barba F., Batanero B.
Tetrahedron,
2008
18.

Feroci M., Elinson M.N., Rossi L., Inesi A.
Electrochemistry Communications,
2009
19.
10.1016/j.mencom.2022.07.036_b0095
Iyer
J. Indian Inst. Sci.,
1931
20.

Villemin D.
Journal of the Chemical Society Chemical Communications,
1983
21.

Elinson M.N., Merkulova V.M., Ilovaisky A.I., Chizhov A.O., Belyakov P.A., Barba F., Batanero B.
Electrochimica Acta,
2010
22.

Tang Y., Sattler I., Thiericke R., Grabley S., Feng X.
European Journal of Organic Chemistry,
2001
23.

Peddibhotla S.
Current Bioactive Compounds,
2009
24.
10.1016/j.mencom.2022.07.036_b0120
Badillo
Curr. Opin. Drug Discovery Dev.,
2010
25.

Zhou F., Liu Y., Zhou J.
Advanced Synthesis and Catalysis,
2010
26.

Maharvi G.M., Ali S., Riaz N., Afza N., Malik A., Ashraf M., Iqbal L., Lateef M.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2008
27.

Khan K.M., Maharvi G.M., Khan M.T., Jabbar Shaikh A., Perveen S., Begum S., Choudhary M.I.
Bioorganic and Medicinal Chemistry,
2006
28.
10.1016/j.mencom.2022.07.036_b0140
Iqbal
J. Chem. Soc. Pak.,
2007
29.

Nie D., Honn K.V.
Cellular and Molecular Life Sciences,
2002
30.

Nikam S., Kornberg B.
Current Medicinal Chemistry,
2001
31.
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2012
32.

Elinson M.N., Ilovaisky A.I., Merkulova V.M., Belyakov P.A., Barba F., Batanero B.
Tetrahedron,
2012
33.

Elinson M.N., Nasybullin R.F., Sokolova O.O., Zaimovskaya T.A., Egorov M.P.
Monatshefte fur Chemie,
2015
34.
Elinson M.N., Ryzhkov F.V., Zaimovskaya T.A., Egorov M.P.
Mendeleev Communications,
2015
35.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
36.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Leonova N.A., Egorov M.P.
Mendeleev Communications,
2020
37.
10.1016/j.mencom.2022.07.036_b0185
Sheldon
Green Chemistry and Catalysis,
2007
38.

Nenajdenko V.G.
Russian Chemical Reviews,
2020
39.

40.
Elinson M.N., Medvedev M.G., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2013
41.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Egorov M.P.
Comptes Rendus Chimie,
2013
42.

Yao W., Cui Y., Wang P., Mao Y.
Letters in Organic Chemistry,
2016
43.

Zhao J., Liu X., Zhang Y., Xue Y.
Journal of Physical Chemistry B,
2021
44.

Yadav L.D., Singh S., Rai V.K.
Green Chemistry,
2009