Keywords
o-quinones
alkylation
electrochemistry
oxidation
phenol
selenium dioxide
tetralines
X-ray study
Abstract
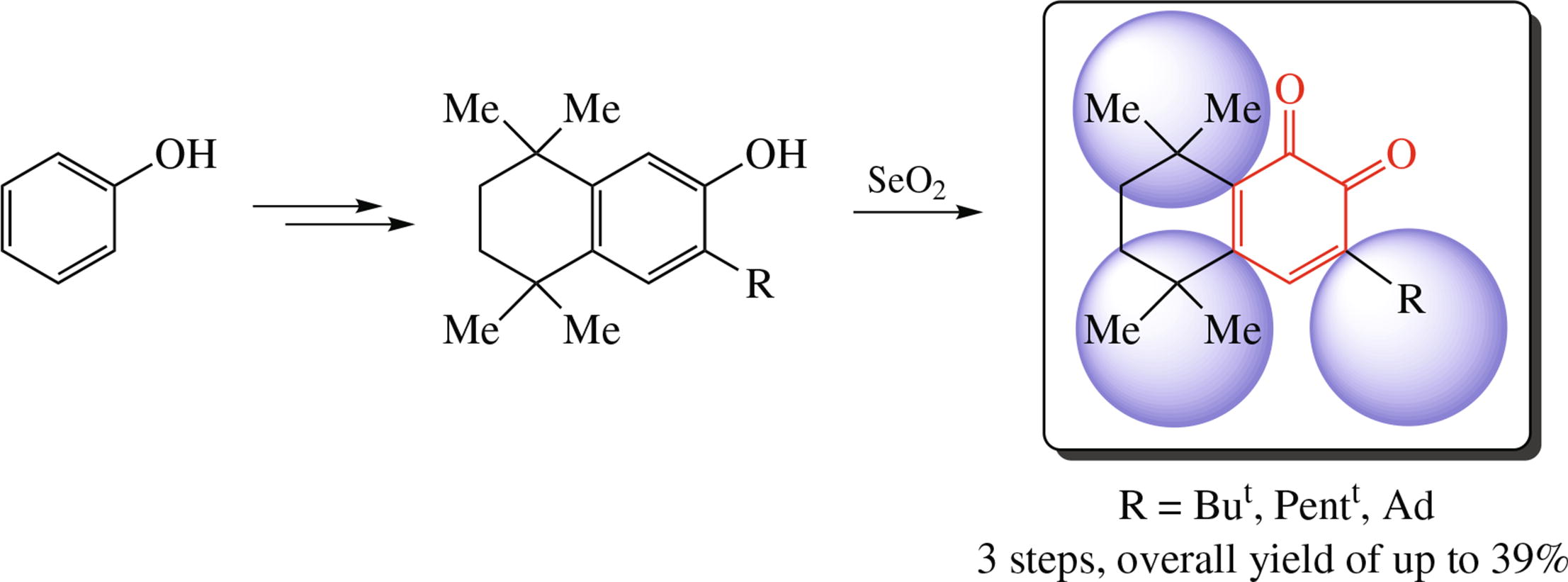
New tri-tert-alkyl substituted o-quinones of 3-tert-alkyl-5,5,8,8-tetramethyl-5,6,7,8-tetrahydronaphthalene-1,2-dione chemotype were obtained from phenol and 2,5-dichloro-2,5-dimethylhexane, with the SeO2 oxidation of the sterically hindered phenol moiety having been performed at the final step. The electrochemical reduction of these quinones proceeds in two stages: the first reduction wave (E1/2 = –0.60 ÷ –0.62 V) is reversible, while the second stage is irreversible.
References
1.
Riley P.A., Ramsden C.A., Land E.J.
2011
2.

Ito S., Sugumaran M., Wakamatsu K.
International Journal of Molecular Sciences,
2020
3.

Wendlandt A.E., Stahl S.S.
Angewandte Chemie - International Edition,
2015
4.

Burmistrova D., Smolyaninov I., Berberova N., Eremenko I.
ChemistrySelect,
2020
5.
Stremski Y., Ahmedova A., Dołęga A., Statkova-Abeghe S., Kirkova D.
Mendeleev Communications,
2021
6.

Ershova I.V., Piskunov A.V., Cherkasov V.K.
Russian Chemical Reviews,
2020
7.

Kieser J.M., Jones L.O., Lin N.J., Zeller M., Schatz G.C., Bart S.C.
Inorganic Chemistry,
2021
8.
10.1016/j.mencom.2022.07.035_b0040
Zhang
Eur. J. Med. Chem.,
2021
9.

Greb L., Ebner F., Ginzburg Y., Sigmund L.M.
European Journal of Inorganic Chemistry,
2020
10.

Martyanov K., Kuropatov V.
Inorganics,
2018
11.

Shurygina M.P., Zakharina M.Y., Baten'kin M.A., Konev A.N., Shavyrin A.S., Chelnokov E.A., Shushunova N.Y., Arsenyev M.V., Chesnokov S.A., Abakumov G.A.
European Polymer Journal,
2020
12.

Mensov S.N., Abakumov G.A., Arsenyev M.V., Baten'kin M.A., Chesnokov S.A., Konev A.N., Polushtaytsev Y.V., Shurygina M.P., Zakharina M.Y.
Journal of Applied Polymer Science,
2020
13.

Abakumov G.A., Cherkasov V.K., Abakumova L.G., Nevodchikov V.I., Druzhkov N.O., Makarenko N.P., Kursky J.A.
Journal of Organometallic Chemistry,
1995
14.

Poddel'sky A.I., Arsenyev M.V., Astaf'eva T.V., Chesnokov S.A., Fukin G.K., Abakumov G.A.
Journal of Organometallic Chemistry,
2017
15.

Zherebtsov M.A., Arsenyev M.V., Chesnokov S.A., Cherkasov V.K.
Russian Journal of Organic Chemistry,
2020
16.

Cherkasov V.K., Abakumov G.A., Shavyrin A.S., Kuz'michev V.V., Baranov E.V., Smolyaninov I.V., Kuropatov V.A.
Asian Journal of Organic Chemistry,
2015
17.

Kocherova T.N., Druzhkov N.O., Martyanov K.A., Shavyrin A.S., Arsenyev M.V., Kulikova T.I., Baranov E.V., Kuropatov V.A., Cherkasov V.K.
Russian Chemical Bulletin,
2020
18.

Kocherova T.N., Druzhkov N.O., Shavyrin A.S., Arsenyev M.V., Baranov E.V., Kuropatov V.A., Cherkasov V.K.
Russian Chemical Bulletin,
2021
19.

El Had M.A., Guardia J.J., Ramos J.M., Taourirte M., Chahboun R., Alvarez-Manzaneda E.
Organic Letters,
2018
20.

Yoo J.W., Lee C.W., Park S., Ko J.
Applied Catalysis A: General,
1999
21.

Sokolenko Y.A., Kuznetsova L.N., Orlovskaya N.F.
Russian Chemical Bulletin,
1996
22.
10.1016/j.mencom.2022.07.035_b0110
Comprehensive Organic Synthesis,
2014
23.

Brock C.P., Duncan L.L.
Chemistry of Materials,
1994
24.

René A., Evans D.H.
Journal of Physical Chemistry C,
2012
25.
10.1016/j.mencom.2022.07.035_b0125
Smolyaninov
Monatsh. Chem.,
1813
26.

Ershova I.V., Meshcheryakova I.N., Trofimova O.Y., Pashanova K.I., Arsenyeva K.V., Khamaletdinova N.M., Smolyaninov I.V., Arsenyev M.V., Cherkasov A.V., Piskunov A.V.
Inorganic Chemistry,
2021
27.

Broere D.L., Plessius R., van der Vlugt J.I.
Chemical Society Reviews,
2015
28.
10.1016/j.mencom.2022.07.035_b0140
Symons
Curr. Opin. Electrochem.,
2021
29.
APEX3, Bruker Molecular Analysis Research Tool, v. 2018.7-2, Bruker, Madison, WI, 2018.
30.
SAINT, Data Reduction and Correction Program, v. 8.38A, Bruker, Madison, WI, 2017.
31.
SADABS, Bruker/Siements Area Detector Absorption Correction Program, v. 2016/2, Bruker, Madison, WI, 2016.
32.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
33.
10.1016/j.mencom.2022.07.035_b0165
Sheldrick
Acta Crystallorg.,
2015
34.
10.1016/j.mencom.2022.07.035_b0170
Sheldrick
Structure Determination Software Suite,
2003