Keywords
45-dihydroxyimidazolidin-2-ones
carboxylic acids
conglomerats
crystal structures
crystallization
glycolurils
racemates
ureido acids
Abstract
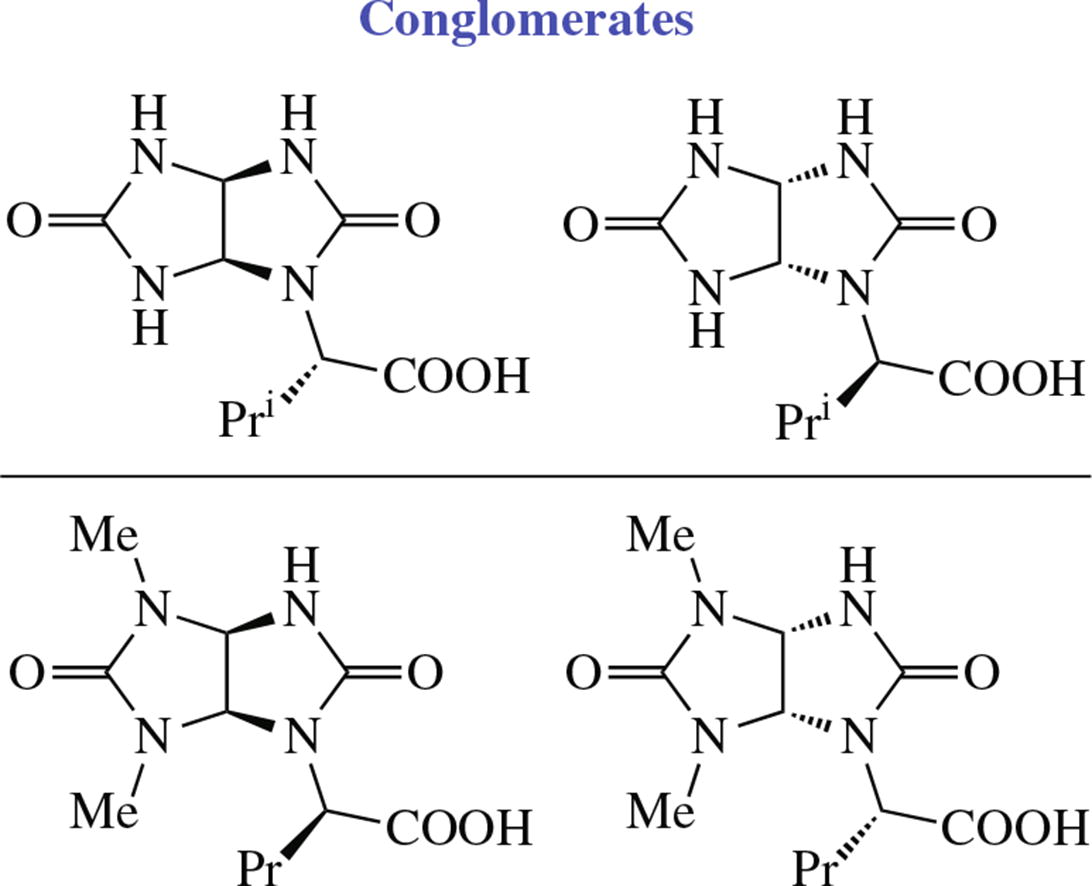
The two novel conglomerates were obtained by crystallization of racemic (2'S,3aS,6aR)/(2'R,3aR,6aS) (glycoluril-1-yl)-3-methylbutanoic acid and (2'R,3aR,6aR)/(2'S,3aS,6aS) (4,6-dimethylglycoluril-1-yl)pentanoic acid synthesized by highly diastereoselective condensation of 4,5-dihydroxy- imidazolidin-2-ones with racemic ureido acids. The differences in the molecular geometry of synthesized racemates were studied by X-ray diffraction that showed them to crystallize as conglomerates in non-centrosymmetric space groups Pna21 and P212121, respectively
References
1.
2.
10.1016/j.mencom.2022.07.034_b0010
Satyanarayana
Int. J. Chem. Tech. Res.,
2011
3.

Muller G.W., Konnecke W.E., Smith A.M., Khetani V.D.
Organic Process Research and Development,
1999
4.
10.1016/j.mencom.2022.07.034_b0020
Franks
Lancet,
1802
5.

Li Y., Coller J.K., Hutchinson M.R., Klein K., Zanger U.M., Stanley N.J., Abell A.D., Somogyi A.A.
Drug Metabolism and Disposition,
2013
6.

Fukumoto K., Toki H., Iijima M., Hashihayata T., Yamaguchi J., Hashimoto K., Chaki S.
Journal of Pharmacology and Experimental Therapeutics,
2017
7.

Ihmsen H.
Clinical Pharmacology and Therapeutics,
2001
8.

Zhang J., Li S., Hashimoto K.
Pharmacology Biochemistry and Behavior,
2014
9.

Hashimoto K.
Biochemical Pharmacology,
2020
10.

Chang L., Zhang K., Pu Y., Qu Y., Wang S., Xiong Z., Ren Q., Dong C., Fujita Y., Hashimoto K.
Pharmacology Biochemistry and Behavior,
2019
11.

Yang C., Shirayama Y., Zhang J., Ren Q., Yao W., Ma M., Dong C., Hashimoto K.
Translational Psychiatry,
2015
12.

Yokoyama R., Matsumoto S., Nomura S., Higaki T., Yokoyama T., Kiyooka S.
Tetrahedron,
2009
13.

Ishak R., Abbas O.
American Journal of Clinical Dermatology,
2013
14.
Anikina L.V., Vikharev Y.B., Baranov V.V., Malyshev O.R., Kravchenko A.N.
Mendeleev Communications,
2018
15.

Karnoukhova V.A., Baranov V.V., Vologzhanina A.V., Kravchenko A.N., Fedyanin I.V.
CrystEngComm,
2021
16.
Baranov V.V., Antonova M.M., Nelyubina Y.V., Kolotyrkina N.G., Zanin I.E., Kravchenko A.N., Makhova N.N.
Mendeleev Communications,
2014
17.

Baranov V.V., Baganova A.A., Chikunov I.E., Karnoukhova V.A., Kravchenko A.N.
Tetrahedron Letters,
2019
18.

Kravchenko A.N., Baranov V.V., Gazieva G.A., Chikunov I.E., Nelyubina Y.V.
Russian Chemical Bulletin,
2014
19.

New access to thioglycolurils by condensation of 4,5-dihydroxyimidazolidin-2-ones(thiones) with HSCN
Baranov V.V., Nelyubina Y.V., Kravchenko A.N., Kolotyrkina N.G., Biriukova K.A.
Tetrahedron Letters,
2015
20.

Nelyubina Y., Baranov V., Antonova M., Kolotyrkina N., Kravchenko A.
Synlett,
2017
21.
![Glycoluril derivatives form hydrogen bonded tapes rather than cucurbit[n]uril congeners](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wu A., Fettinger J.C., Isaacs L.
Tetrahedron,
2002
22.

Johnson D.W., Palmer L.C., Hof F., Iovine P.M., Rebek J.
Chemical Communications,
2002
23.
(b) N.-F. She, X.-G. Meng, M. Gao, A.-X. Wu and L Isaacs, Chem. Commun., 2008, 3133
24.
Wyler R., de Mendoza J., Rebek J.
1993
25.

Branda N., Wyler R., Rebek J.
Science,
1994
26.

Branda N., Grotzfeld R.M., Valdes C., Rebek J.J.
Journal of the American Chemical Society,
1995
27.

Grotzfeld R.M., Branda N., Rebek J.
Science,
1996
28.

Meissner R., Garcias X., Mecozzi S., Rebek J.
Journal of the American Chemical Society,
1997
29.

Tokunaga Y., Rebek J.
Journal of the American Chemical Society,
1998
30.

Rivera J.M., Martín T., Rebek J.
Journal of the American Chemical Society,
1998
31.

Kang J., Hilmersson G., Santamaría J., Rebek J.
Journal of the American Chemical Society,
1998
32.

Rebek J.
Accounts of Chemical Research,
1998
33.

Rowan A.E., Elemans J.A., Nolte R.J.
Accounts of Chemical Research,
1999
34.

Rivera J.M., Rebek J.
Journal of the American Chemical Society,
2000
35.

Rivera J.M., Martín T., Rebek J.
Journal of the American Chemical Society,
2001
36.

Elemans J.A., Rowan A.E., Nolte R.J.
Industrial & Engineering Chemistry Research,
2000
37.

Szabo T., O'Leary B.M., Rebek, Jr. J.
Angewandte Chemie - International Edition,
1998
38.

O'Leary B.M., Szabo T., Svenstrup N., Schalley C.A., Lützen A., Schäfer M., Rebek J.
Journal of the American Chemical Society,
2001
39.

Lützen A., Starnes S.D., Rudkevich D.M., Rebek J.
Tetrahedron Letters,
2000
40.

Hof F., Nuckolls C., Craig S.L., Martín T., Rebek J.
Journal of the American Chemical Society,
2000
41.

Hof F., Nuckolls C., Rebek J.
Journal of the American Chemical Society,
2000
42.

Nuckolls C., Hof F., Martín T., Rebek J.
Journal of the American Chemical Society,
1999
43.

Rudkevich D.M., Rebek J.
Angewandte Chemie International Edition in English,
1997
44.

Chen Y., She N., Wu A.
Journal of Chemical Crystallography,
2011
45.

Tokunaga Y., Rudkevich D.M., Santamaría J., Hilmersson G., Rebek, Jr. J.
Chemistry - A European Journal,
1998
46.

She N., Gao M., Meng X., Yang G., Elemans J.A., Wu A., Isaacs L.
Journal of the American Chemical Society,
2009
47.
Baranov V.V., Barsegyan Y.A., Strelenko Y.A., Karnoukhova V.A., Kravchenko A.N.
Mendeleev Communications,
2020
48.

Johnson D.W., Hof F., Palmer L.C., Martín T., Obst U., Rebek, Jr. J.
Chemical Communications,
2003
49.

Moon K., Chen W., Ren T., Kaifer A.E.
CrystEngComm,
2003
50.

Kölbel M., Menger F.M.
Chemical Communications,
2001
51.
Kravchenko A.N., Chegaev K.Y., Chikunov I.E., Belyakov P.A., Maksareva E.Y., Lyssenko K.A., Lebedev O.V., Makhova N.N.
Mendeleev Communications,
2003
52.
Kostyanovsky R.G., El’natanov Y.I., Krutius O.N., Chervin I.I., Lyssenko K.A.
Mendeleev Communications,
1998
53.
Lyssenko K.A., Golovanov D.G., Kravchenko A.N., Chikunov I.E., Lebedev O.V., Makhova N.N.
Mendeleev Communications,
2004
54.

Kravchenko A.N., Lyssenko K.A., Chikunov I.E., Belyakov P.A., Il’in M.M., Baranov V.V., Nelyubina Y.V., Davankov V.A., Pivina T.S., Makhova N.N., Antipin M.Y.
Russian Chemical Bulletin,
2009
55.

Gazieva G._., Nelyubina Y.V., Kravchenko A.N., Sigachev A.S., Glukhov I.V., Struchkova M.I., Lyssenko K.A., Makhova N.N.
Russian Chemical Bulletin,
2009
56.

Baranov V.V., Galochkin A.A., Nelyubina Y.V., Kravchenko A.N., Makhova N.N.
Synthesis,
2020
57.
Zlokazov M.V., Pivnitsky K.K.
Mendeleev Communications,
2020
58.

Kravchenko A.N., Baranov V.V., Gazieva G.A.
Russian Chemical Reviews,
2018
59.

Anikina L.V., Gazieva G.A., Kravchenko A.N.
Russian Chemical Bulletin,
2020
60.

Makhova N.N., Belen’kii L.I., Gazieva G.A., Dalinger I.L., Konstantinova L.S., Kuznetsov V.V., Kravchenko A.N., Krayushkin M.M., Rakitin O.A., Starosotnikov A.M., Fershtat L.L., Shevelev S.A., Shirinian V.Z., Yarovenko V.N.
Russian Chemical Reviews,
2020
61.
(a) R. Kamburg, US Patent 2008/0227838 A1, 2008
62.
(b) A. N. Kravchenko, V. V. Baranov, L. V. Anikina, Yu. B. Vikharev, I. S. Bushmarinov and Yu. V. Nelyubina, Russ. J. Bioorg. Chem., 2012, 38, 550 (Bioorg. Khim., 2012, 38, 621).
63.
Chikunov I.E., Kravchenko A.N., Belyakov P.A., Lyssenko K.A., Baranov V.V., Lebedev O.V., Makhova N.N.
Mendeleev Communications,
2004
64.
Baranov V.V., Kravchenko A.N., Belyakov P.A., Makhova N.N.
Mendeleev Communications,
2008
65.

Vail S.L., Barker R.H., Mennitt P.G.
Journal of Organic Chemistry,
1965
66.

Grillon E., Gallo R., Pierrot M., Boileau J., Wimmer E.
Tetrahedron Letters,
1988
67.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
68.
10.1016/j.mencom.2022.07.034_b0135
Sheldrick
Acta Crystallogr.,
2015
69.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007