Keywords
complex formation
merocyanine dyes
NIR-dyes
photochromism
spiropyrans
Abstract
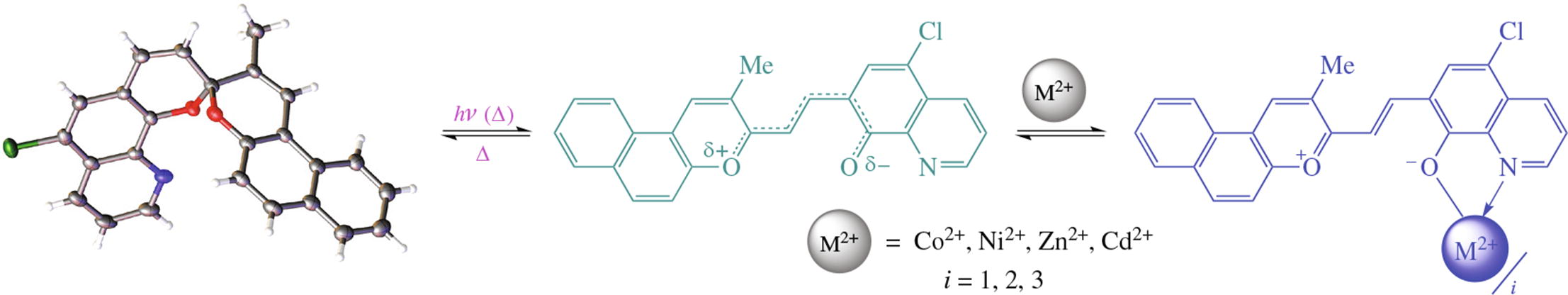
A compound of the new spirobipyran-quinoline series possesses weakly pronounced photochromic properties at room temperature. Its interaction with Co2+, Ni2+, Zn2+ and Cd2+ ions leads to intensely colored complexes absorbing in the λ> 600 nm region, which is important for the study of biological objects. Up to three merocyanine molecules are capable of binding one metal ion, which is not typical for the previously studied indoline analogs.
References
1.

Kortekaas L., Browne W.R.
Chemical Society Reviews,
2019
2.

3.

Tian W., Tian J.
Dyes and Pigments,
2014
4.

Jin Fang Zhi, Baba R., Hashimoto K., Fujishima A.
Journal of Photochemistry and Photobiology A: Chemistry,
1995
5.

6.

Winkler J.D., Bowen C.M., Michelet V.
Journal of the American Chemical Society,
1998
7.

Solov’eva E.V., Voloshin N.A., Chernyshev A.V., Reutova Y.S., Metelitsa A.V.
Doklady Chemistry,
2020
8.

Ali A.A., Kharbash R., Kim Y.
Analytica Chimica Acta,
2020
9.
10.1016/j.mencom.2022.07.032_b0045
Zhang
Inorg. Chem.,
1865
10.

Harada J., Kawazoe Y., Ogawa K.
Chemical Communications,
2010
11.

Ostromisslensky I.
Berichte der deutschen chemischen Gesellschaft,
1911
12.
10.1016/j.mencom.2022.07.032_b0060
Job
Ann. Chim.,
1928
13.
![Photo- and thermochromic cation sensitive spiro[indoline-pyridobenzopyrans]](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Chernyshev A.V., Metelitsa A.V., Gaeva E.B., Voloshin N.A., Borodkin G.S., Minkin V.I.
Journal of Physical Organic Chemistry,
2007
14.

Feuerstein T.J., Müller R., Barner-Kowollik C., Roesky P.W.
Inorganic Chemistry,
2019
15.

Metelitsa A.V., Coudret C., Micheau J.C., Voloshin N.A.
RSC Advances,
2014
16.
CrysAlisPro, Version 171.41.93a, Rigaku Oxford Diffraction, 2015, https://www.rigaku.com/products/crystallography/crysalis.
17.
10.1016/j.mencom.2022.07.032_b0085
Sheldrick
Acta Crystallogr.,
2015
18.
10.1016/j.mencom.2022.07.032_b0090
Sheldrick
Acta Crystallogr.,
2015
19.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009