Keywords
benzothiadiazole
branched side chains
conjugated polymer
organic solar cells
thiophene
Abstract
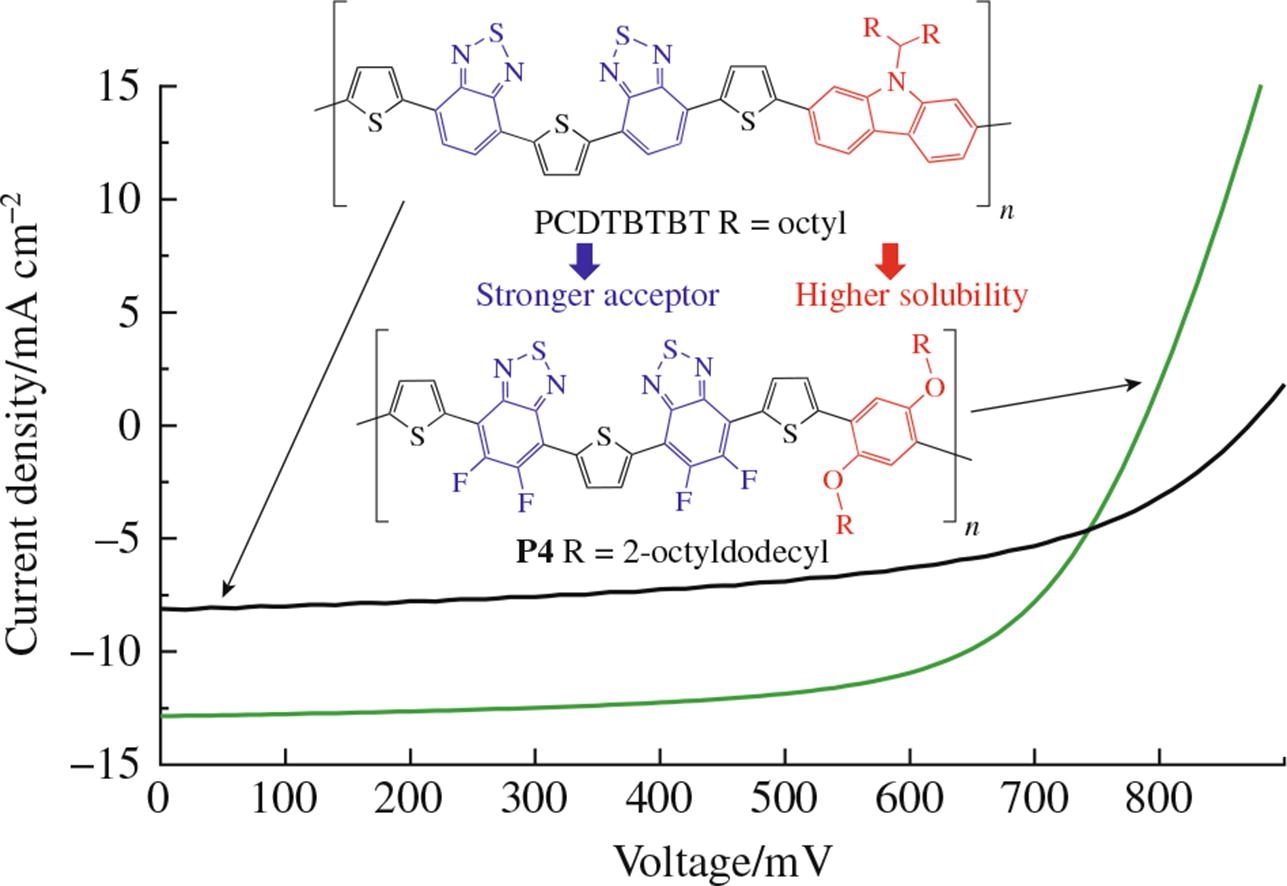
Novel fluorene- and phenylene-based conjugated polymers with the TBTBT molecular framework consisting of the thiophene (T) and benzothiadiazole (B) building blocks have been synthesized and investigated. It has been demonstrated that the variation of X building blocks with branched side chains in (X-TBTBT)n-type structures, as well as the introduction of fluorine into the main chain, strongly affects the optical, electronic and physicochemical properties of the obtained polymers. The phenylene-based polymer with a fluorine- loaded TBTBT block achieves a power conversion efficiency of 7% in organic solar cells, which can be further improved by optimizing the active layer morphology.
References
1.

Xu Y., Yuan J., Zhou S., Seifrid M., Ying L., Li B., Huang F., Bazan G.C., Ma W.
Advanced Functional Materials,
2019
2.
New benzodithiophene-pyrrolopyrroledione-thienopyrazine random terpolymers for organic photovoltaics
Keshtov M.L., Kuklin S.A., Konstantinov I.O., Zou Y., Sharma G.D.
Mendeleev Communications,
2021
3.

Han D., Khan Y., Ting J., King S.M., Yaacobi‐Gross N., Humphries M.J., Newsome C.J., Arias A.C.
Advanced Materials,
2017
4.

Xu X., Zhou X., Zhou K., Xia Y., Ma W., Inganäs O.
Advanced Functional Materials,
2018
5.

Higashihara T.
Polymer Journal,
2021
6.

Xia Y., Xu X., Aguirre L.E., Inganäs O.
Journal of Materials Chemistry A,
2018
7.

Chang J.S., Facchetti A.F., Reuss R.
IEEE Journal on Emerging and Selected Topics in Circuits and Systems,
2017
8.

Adegoke O.O., Jung I.H., Orr M., Yu L., Goodson T.
Journal of the American Chemical Society,
2015
9.

Dou J., Zheng Y., Lei T., Zhang S., Wang Z., Zhang W., Wang J., Pei J.
Advanced Functional Materials,
2014
10.
10.1016/j.mencom.2022.07.031_b0050
Akkuratov
Macromolecules,
2013
11.
Akkuratov A.V., Susarova D.K., Mukhacheva O.A., Troshin P.A.
Mendeleev Communications,
2016
12.
10.1016/j.mencom.2022.07.031_b0060
Scharber
Adv. Mater.,
1994
13.

Kuznetsov I.E., Kuznetsov P.M., Ustinova M.I., Zakirov K.E., Troshin P.A., Akkuratov A.V.
Physica Status Solidi (A) Applications and Materials Science,
2021
14.
Kuznetsov I.E., Kuznetsov P.M., Maskaev A.V., Akkuratov A.V., Troshin P.A.
Mendeleev Communications,
2020
15.

Akkuratov A.V., Prudnov F.A., Inasaridze L.N., Troshin P.A.
Tetrahedron Letters,
2017
16.

Makuła P., Pacia M., Macyk W.
Journal of Physical Chemistry Letters,
2018
17.

Liu Z., Hu Y., Li P., Wen J., He J., Gao X.
Journal of Materials Chemistry C,
2020
18.

Hashemi D., Ma X., Ansari R., Kim J., Kieffer J.
Physical Chemistry Chemical Physics,
2019
19.

Nguyen T.L., Choi H., Ko S.-., Uddin M.A., Walker B., Yum S., Jeong J.-., Yun M.H., Shin T.J., Hwang S., Kim J.Y., Woo H.Y.
Energy and Environmental Science,
2014
20.

Lee C., Lai Y., Hsu J., Huang P., Cheng Y.
Chemical Science,
2017
21.

Ko S., Hoang Q.V., Song C.E., Uddin M.A., Lim E., Park S.Y., Lee B.H., Song S., Moon S., Hwang S., Morin P., Leclerc M., Su G.M., Chabinyc M.L., Woo H.Y., et. al.
Energy and Environmental Science,
2017
22.

Cardona C.M., Li W., Kaifer A.E., Stockdale D., Bazan G.C.
Advanced Materials,
2011
23.

Leclerc N., Chávez P., Ibraikulov O., Heiser T., Lévêque P.
Polymers,
2016
24.

Hummelen J.C., Knight B.W., LePeq F., Wudl F., Yao J., Wilkins C.L.
Journal of Organic Chemistry,
1995
25.
![Efficient Methano[70]fullerene/MDMO‐PPV Bulk Heterojunction Photovoltaic Cells](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Wienk M.M., Kroon J.M., Verhees W.J., Knol J., Hummelen J.C., van Hal P.A., Janssen R.A.
Angewandte Chemie,
2003
26.

Brabec C.J., Cravino A., Meissner D., Sariciftci N.S., Fromherz T., Rispens M.T., Sanchez L., Hummelen J.C.
Advanced Functional Materials,
2001
27.

Xin Y., Zeng G., OuYang J., Zhao X., Yang X.
Journal of Materials Chemistry C,
2019
28.

Umeyama T., Watanabe Y., Douvogianni E., Imahori H.
Journal of Physical Chemistry C,
2013
29.
10.1016/j.mencom.2022.07.031_b0145
Markov
Phys. Rev. B: Condens. Matter Mater. Phys.,
2005
30.

Markov D.E., Amsterdam E., Blom P.W., Sieval A.B., Hummelen J.C.
Journal of Physical Chemistry A,
2005