Keywords
D–π–A structure
dye-sensitized solar cells
metal-free dyes
rhodanine-3-acetic acid
thieno[32-b]indoles
Abstract
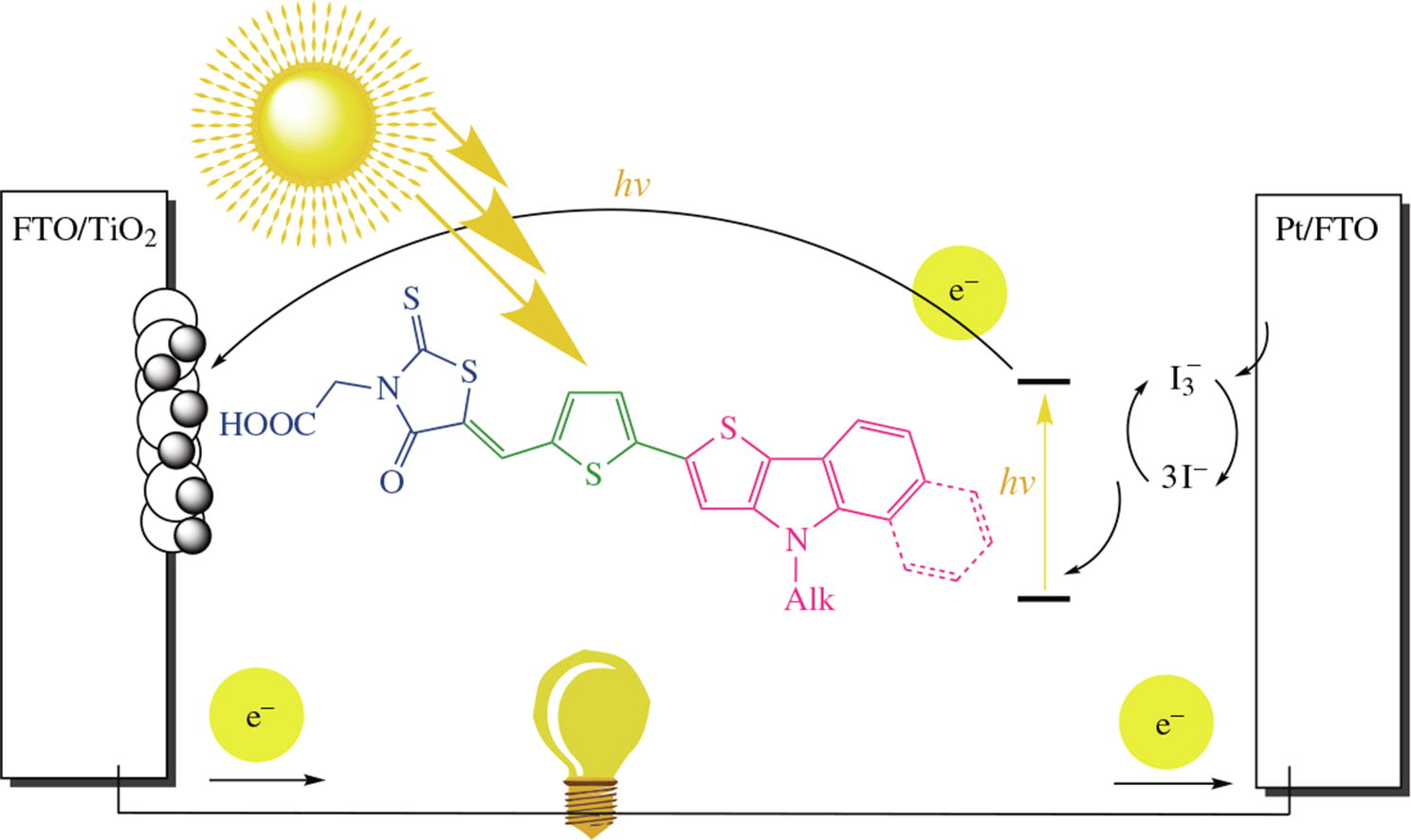
Five new dyes with D–π–A structure bearing 5-(methylene)-rhodanine-3-acetic acid as an acceptor-anchoring part and thieno[3,2-b]indole or benzo[g]thieno[3,2-b]indole as an electron-donating part were synthesized and applied as photosensitizers for dye-sensitized solar cells (DSSCs). In addition, thermal stability, optical and electrochemical properties of these dyes were investigated. The highest PCE value of 1.09% (Jsc = 3.01 mA cm–2, Voc = 0.53 V, FF = 0.69) was achieved for DSSC based on benzo[g]thieno[3,2-b]indole dye under AM 1.5G irradiation.
References
1.
Nam S., Lee K.H., Yu J., Boo J.
Applied Science and Convergence Technology,
2019
2.

Lee C., Lin R.Y., Lin L., Li C., Chu T., Sun S., Lin J.T., Ho K.
RSC Advances,
2015
3.

Dragonetti C., Colombo A.
Molecules,
2021
4.

O'Regan B., Grätzel M.
Nature,
1991
5.

Grätzel M.
Journal of Photochemistry and Photobiology C: Photochemistry Reviews,
2003
6.

Mishra A., Fischer M. ., Bäuerle P.
Angewandte Chemie - International Edition,
2009
7.

Chaurasia S., Lin J.T.
Chemical Record,
2016
8.
Egorov A.E., Kostyukov A.A., Borissevitch I.E., Kuzmin V.A.
Mendeleev Communications,
2021
9.

Murakami T.N., Koumura N.
Advanced Energy Materials,
2018
10.
New benzodithiophene-pyrrolopyrroledione-thienopyrazine random terpolymers for organic photovoltaics
Keshtov M.L., Kuklin S.A., Konstantinov I.O., Zou Y., Sharma G.D.
Mendeleev Communications,
2021
11.
10.1016/j.mencom.2022.07.030_b0055
Ziarani
Metal-Free Synthetic Organic Dyes,
2018
12.
![A new convenient synthetic route towards 2-(hetero)aryl-substituted thieno[3,2-b]indoles using Fischer indolization](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Irgashev R.A., Steparuk A.S., Rusinov G.L.
Organic and Biomolecular Chemistry,
2018
13.
10.1016/j.mencom.2022.07.030_b0065
Irgashev
Tetrahedron Lett.,
2019
14.
![Synthesis and photovoltaic properties of new thieno[3,2-b]indole-based dyes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Steparuk A.S., Irgashev R.A., Rusinov G.L., Krivogina E.V., Lazarenko P.I., Kozyukhin S.A.
Russian Chemical Bulletin,
2019
15.

Zhang L., Cole J.M.
ACS applied materials & interfaces,
2015
16.
![Recent advances in the chemistry of thieno[2,3-b]pyridines 1. Methods of synthesis of thieno[2,3-b]pyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Dotsenko V.V., Buryi D.S., Lukina D.Y., Krivokolysko S.G.
Russian Chemical Bulletin,
2020
17.

Mao M., Zhang X., Fang X., Wu G., Ding Y., Liu X., Dai S., Song Q.
Organic Electronics,
2014
18.

Babu D.D., Gachumale S.R., Anandan S., Adhikari A.V.
Dyes and Pigments,
2015
19.

Babu D.D., Elsherbiny D., Cheema H., El-Shafei A., Adhikari A.V.
Dyes and Pigments,
2016
20.

Fuse S., Sugiyama S., Maitani M.M., Wada Y., Ogomi Y., Hayase S., Katoh R., Kaiho T., Takahashi T.
Chemistry - A European Journal,
2014
21.
![Optical and Photovoltaic Properties of Thieno[3,2-b]thiophene-Based Push–Pull Organic Dyes with Different Anchoring Groups for Dye-Sensitized Solar Cells](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Fernandes S.S., Castro M.C., Pereira A.I., Mendes A., Serpa C., Pina J., Justino L.L., Burrows H.D., Raposo M.M.
ACS Omega,
2017
22.

Patil D.S., Sonigara K.K., Jadhav M.M., Avhad K.C., Sharma S., Soni S.S., Sekar N.
New Journal of Chemistry,
2018
23.

Echeverry-Gonzalez C.A., Ortiz A., Insuasty B.
New Journal of Chemistry,
2019
24.
![Synthesis and characterization of push-pull bithiophene and thieno[3,2-b]thiophene derivatives bearing an ethyne linker as sensitizers for dye-sensitized solar cells](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fernandes S.S., Mesquita I., Andrade L., Mendes A., Justino L.L., Burrows H.D., Raposo M.M.
Organic Electronics,
2017
25.

Elmorsy M.R., Su R., Fadda A.A., Etman H.A., Tawfik E.H., El-Shafei A.
Dyes and Pigments,
2018
26.

Avhad K., Jadhav M., Patil D., Chowdhury T.H., Islam A., Bedja I., Sekar N.
Organic Electronics,
2019
27.

Zhang L., Cole J.M.
Journal of Materials Chemistry A,
2017