Keywords
2-hetarylfurans
arylation
benzimidazoles
carboxylate assistance
CH-functionalization
furans
ruthenium complexes
Abstract
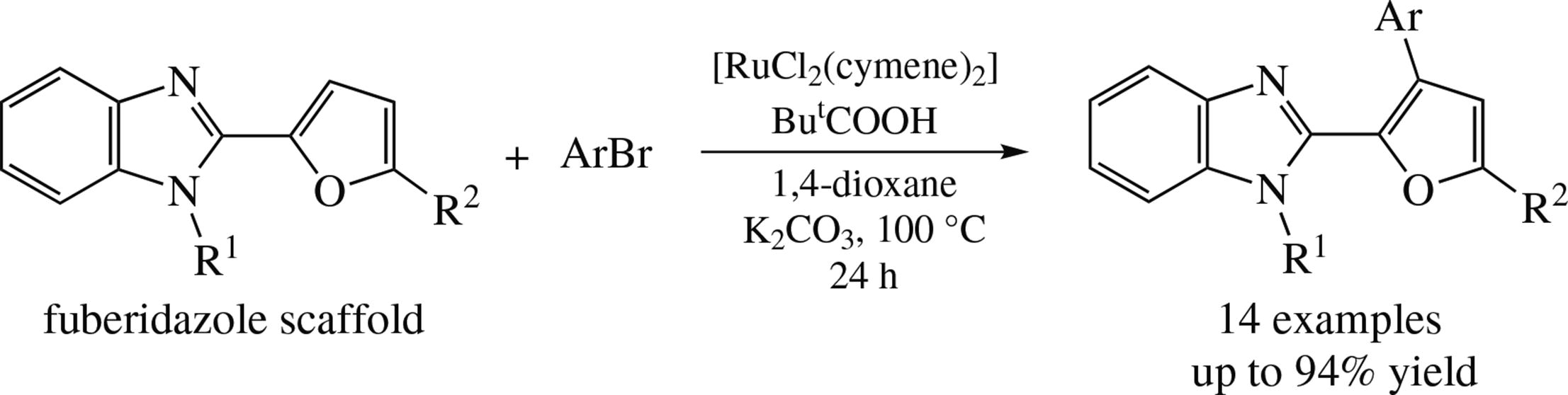
An efficient selective C(3)–H arylation of furan ring in 2-(furan-2-yl)benzimidazoles, derivatives of fuberidazole fungicide, with aryl bromides catalyzed by [RuCl2(cymene)]2/ pivalic acid system has been accomplished. High selectivity of the process may be accounted for by the action of benzimidazol-2-yl substituent as the directing group.
References
1.

Romashov L.V., Kozlov K.S., Skorobogatko M.K., Kostyukovich A.Y., Ananikov V.P.
Chemistry - An Asian Journal,
2021
2.
10.1016/j.mencom.2022.07.018_b0010
Banerjee
Int. J. Rev. Life Sci.,
2012
3.
Verbitskiy E.V., Baskakova S.A., Belyaev D.V., Vakhrusheva D.V., Eremeeva N.I., Rusinov G.L., Charushin V.N.
Mendeleev Communications,
2021
4.

Petrelli F., Ghidini M., Lonati V., Tomasello G., Borgonovo K., Ghilardi M., Cabiddu M., Barni S.
European Journal of Cancer,
2017
5.

LeWitt P.A., Aradi S.D., Hauser R.A., Rascol O.
Parkinsonism and Related Disorders,
2020
6.

Fürstner A.
Angewandte Chemie - International Edition,
2003
7.
10.1016/j.mencom.2022.07.018_b0035
Aftab
Int. J. Environ. Anal. Chem.,
2021
8.

Wen Y., Li J., Yang F., Zhang W., Li W., Liao C., Chen L.
Talanta,
2013
9.

Temirak A., Shaker Y.M., Ragab F.A., Ali M.M., Ali H.I., El Diwani H.I.
European Journal of Medicinal Chemistry,
2014
10.

Abdullaziz M.A., Abdel-Mohsen H.T., El Kerdawy A.M., Ragab F.A., Ali M.M., Abu-bakr S.M., Girgis A.S., El Diwani H.I.
European Journal of Medicinal Chemistry,
2017
11.

Abdel-Mohsen H.T., Abdullaziz M.A., El Kerdawy A.M., Ragab F.A., Flanagan K.J., Mahmoud A.E., Ali M.M., El Diwani H.I., Senge M.O.
Molecules,
2020
12.

Racané L., Zlatar I., Perin N., Cindrić M., Radovanović V., Banjanac M., Shanmugam S., Stojković M.R., Brajša K., Hranjec M.
Molecules,
2021
13.

Peixoto P., Liu Y., Depauw S., Hildebrand M., Boykin D.W., Bailly C., Wilson W.D., David-Cordonnier M.
Nucleic Acids Research,
2008
14.

Dunwell D.W., Evans D., Hicks T.A.
Journal of Medicinal Chemistry,
1975
15.

Kim J., Lee S., Kim S., Jung M., Lee H., Han M.S.
Dyes and Pigments,
2020
16.

Hei J., Zhi Y., Zhen Y., Li J., Zhang F.
Journal of Coordination Chemistry,
2013
17.
![Synthesis, optical and electrochemical properties of 2-[(9H-fluoren-2-yl)aryl]-1H-benz[d]imidazole and 2,7-bis[(1H-benz[d]imidazol-2-yl)aryl]- 9H-fluorene derivatives](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Li Q., Li M., Lin H., Xu P., Gu Z., Cui Y.
Heterocyclic Communications,
2016
18.
![Synthesis and Reactivity of 2-(Furan-2-yl)benzo[1,2-d:4,3-d′]bis[1,3]thiazole](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Aleksandrov A.A., Elchaninov M.M., Tishina D.A., Tarakanova Y.E., Shmanovsky M.L.
Russian Journal of Organic Chemistry,
2021
19.
![2-(Fur-2-yl)thiazolo[4,5-f]quinoline: Synthesis and Electrophilic Substitution Reactions](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Aleksandrov A.A., El’chaninov M.M., Zablotskii D.A.
Russian Journal of General Chemistry,
2019
20.
M. M. El’chaninov, A. M. Simonov and L. Ya. Oleinikova, Chem. Heterocycl. Compd., 1983, 19, 1041 (Khim. Geterotsikl. Soedin., 1983, 19, 1311).
21.

Doucet H., Ben Ammar H., Abdellaoui F., Youssef C., Soulé J.
Synthesis,
2014
22.

Khazipov O.V., Shepelenko K.E., Pasyukov D.V., Chesnokov V.V., Soliev S.B., Chernyshev V.M., Ananikov V.P.
Organic Chemistry Frontiers,
2021
23.

Al Mamari H.H., Grošelj U., Požgan F., Brodnik H.
Journal of Organic Chemistry,
2021
24.

Karlinskii B.Y., Ananikov V.P.
ChemSusChem,
2020
25.

Kucherov F.A., Romashov L.V., Galkin K.I., Ananikov V.P.
ACS Sustainable Chemistry and Engineering,
2018
26.

Karlinskii B.Y., Kostyukovich A.Y., Kucherov F.A., Galkin K.I., Kozlov K.S., Ananikov V.P.
ACS Catalysis,
2020
27.

Nareddy P., Jordan F., Szostak M.
ACS Catalysis,
2017
28.

Gramage-Doria R., Bruneau C.
Coordination Chemistry Reviews,
2021
29.
10.1016/j.mencom.2022.07.018_b0145
Siopa
Eur. J. Org. Chem.,
2018
30.

Nareddy P., Jordan F., Brenner-Moyer S.E., Szostak M.
ACS Catalysis,
2016
31.

Ravasco J.M., Monteiro C.M., Siopa F., Trindade A.F., Oble J., Poli G., Simeonov S.P., Afonso C.A.
ChemSusChem,
2019
32.
10.1016/j.mencom.2022.07.018_b0160
Li
Synlett,
2017
33.

Zha G., Qin H., Kantchev E.A.
RSC Advances,
2016
34.

Ackermann L.
Chemical Reviews,
2011
35.
Shepelenko K.E., Nikolaeva K.A., Shevchenko M.A., Tkachenko Y.N., Minyaev M.E., Chernyshev V.M.
Mendeleev Communications,
2022