Keywords
catalysis
Cyclopentadienyl ligands
diazo compounds
insertion
rhodium complexes
Abstract
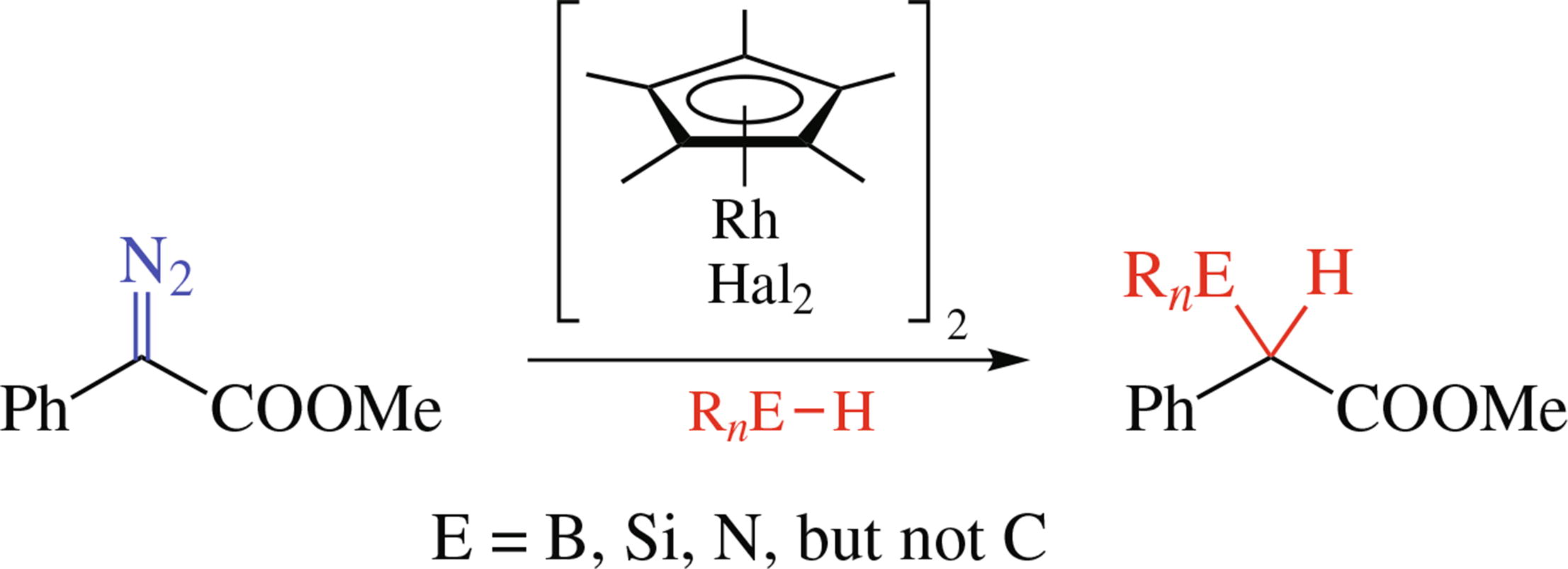
Rhodium(III) complexes catalyze the insertion of carbenoids generated from diazo compounds into E−H bonds (E = B, Si, N, but not C), although less efficiently than classical rhodium(II) carboxylates, despite formally higher oxidation state of the metal.
References
1.

Menchikov L.G., Shulishov E.V., Tomilov Y.V.
Russian Chemical Reviews,
2021
2.

Gillingham D., Fei N.
Chemical Society Reviews,
2013
3.

Yang Z., Stivanin M.L., Jurberg I.D., Koenigs R.M.
Chemical Society Reviews,
2020
4.

Eremeyeva M., Zhukovsky D., Dar’in D., Krasavin M.
Beilstein Journal of Organic Chemistry,
2020
5.

Krasavin M., Zhukovsky D., Solovyev I., Barkhatova D., Dar'in D., Frank D., Martinelli G., Weizel L., Proschak A., Rotter M., Kramer J.S., Brunst S., Wichelhaus T.A., Proschak E.
ChemMedChem,
2021
6.
Shvydkiy N.V., Perekalin D.S.
Mendeleev Communications,
2021
7.
10.1016/j.mencom.2022.07.017_b0035
Solovyov
Eur. J. Org. Chem.,
2019
8.
Vorob’eva D.V., Titanyuk I.D., Beletskaya I.P., Osipov S.N.
Mendeleev Communications,
2005
9.

Loskutova N.L., Shvydkiy N.V., Nelyubina Y.V., Perekalin D.S.
Journal of Organometallic Chemistry,
2018
10.
10.1016/j.mencom.2022.07.017_b0050
Cailler
Dalton Trans.,
2023
11.
Zhmurov P.A., Dar’in D.V., Bakulina O.Y., Krasavin M.
Mendeleev Communications,
2020
12.

Kolos A.V., Nelyubina Y.V., Sundararaju B., Perekalin D.S.
Organometallics,
2021
13.

Davies H.M., Liao K.
Nature Reviews Chemistry,
2019
14.

Chen D., Zhang X., Qi W., Xu B., Xu M.
Journal of the American Chemical Society,
2015
15.

Ankudinov N.M., Chusov D.A., Nelyubina Y.V., Perekalin D.S.
Angewandte Chemie - International Edition,
2021
16.

Chen D., Zhu D., Xu M.
Journal of the American Chemical Society,
2016
17.

Zhu D., Xia H., Liu J., Chung L.W., Xu M.
Journal of the American Chemical Society,
2021
18.
10.1016/j.mencom.2022.07.017_b0090
Liu
Chin. J. Chem.,
1911
19.

Kumar S., Nunewar S., Oluguttula S., Nanduri S., Kanchupalli V.
Organic and Biomolecular Chemistry,
2021
20.

Osipov S., Vorobyeva D.
Synthesis,
2017
21.

Werlé C., Goddard R., Philipps P., Farès C., Fürstner A.
Journal of the American Chemical Society,
2016
22.
10.1016/j.mencom.2022.07.017_b0110
Tindall
J. Am. Chem. Soc.,
1884
23.

Trifonova E.A., Ankudinov N.M., Mikhaylov A.A., Chusov D.A., Nelyubina Y.V., Perekalin D.S.
Angewandte Chemie - International Edition,
2018
24.

Trifonova E.A., Ankudinov N.M., Kozlov M.V., Sharipov M.Y., Nelyubina Y.V., Perekalin D.S.
Chemistry - A European Journal,
2018
25.

Esteruelas M.A., Fernández-Alvarez F.J., López A.M., Oñate E., Ruiz-Sánchez P.
Organometallics,
2006
26.

Fernandez M.J., Maitlis P.M.
Organometallics,
1983
27.
![Arene complexes [(η-C5H5)M(η-C6R6)]2+ (M = Rh, Ir)](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Loginov D.A., Vinogradov M.M., Starikova Z.A., Petrovskii P.V., Kudinov A.R.
Russian Chemical Bulletin,
2004
28.

Doyle M.P., Duffy R., Ratnikov M., Zhou L.
Chemical Reviews,
2009