Keywords
anti-boat and gauche-boat conformers
diazabicyclo[3.1.0]hexanes
diaziridines
enthalpy of formation
gas electron diffraction structure
synthesis
Abstract
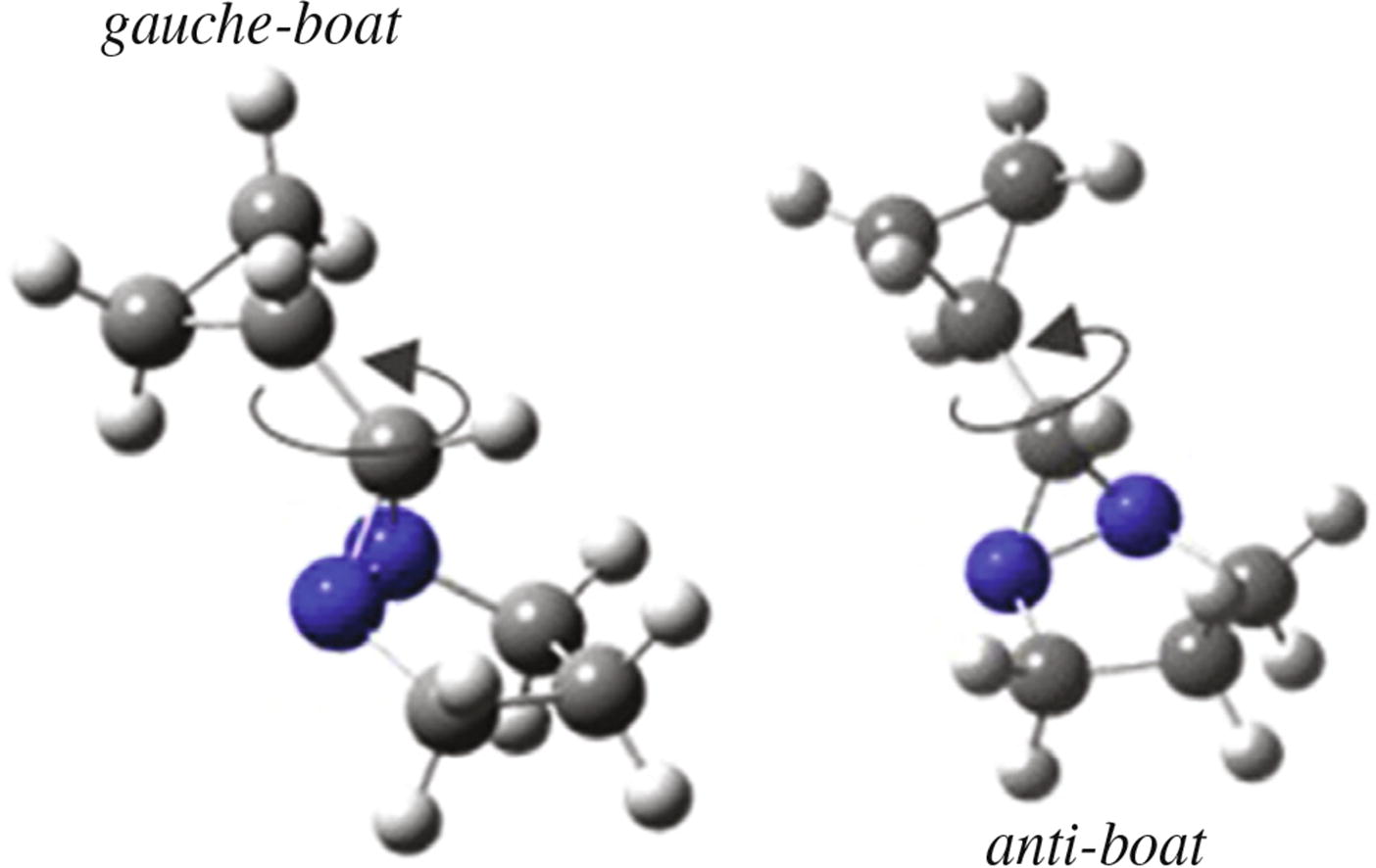
The molecular structure and conformational composition of 6-cyclopropyl-1,5-diazabicyclo[3.1.0]hexane were determined by gas phase electron diffraction and quantum chemical calculations. The gas phase electron diffraction data were well reproduced for the mixture of two conformers with anti-boat and gauche-boat mutual ring orientation having 15 and 85% relative abundance, respectively. The standard enthalpy of formation of substance under study was calculated using atomization reactions, yielding value of 307.9 ± 3.3 kJ mol-1 in gas phase.
References
1.
![1,5- Diazabicyclo[3.1.0]hexanes and 1,6-diazabicyclo[4.1.0]heptanes: A new method for the synthesis, quantum-chemical calculations, and X-ray diffraction study](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kuznetsov V.V.
Russian Chemical Bulletin,
2003
2.
![Molecular structure of 1,5-diazabicyclo[3.1.0]hexane as determined by gas electron diffraction and quantum-chemical calculations](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Vishnevskiy Y.V., Vogt N., Vogt J., Rykov A.N., Kuznetsov V.V., Makhova N.N., Vilkov L.V.
Journal of Physical Chemistry A,
2008
3.
![Conformational and Bonding Properties of 3,3-Dimethyl- and 6,6-Dimethyl-1,5-diazabicyclo[3.1.0]hexane: A Case Study Employing the Monte Carlo Method in Gas Electron Diffraction](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Vishnevskiy Y.V., Schwabedissen J., Rykov A.N., Kuznetsov V.V., Makhova N.N.
Journal of Physical Chemistry A,
2015
4.

Kuznetsov V.V., Marochkin I.I., Goloveshkin A.S., Makhova N.N., Shishkov I.F.
Structural Chemistry,
2017
5.

Altova E.P., Kuznetsov V.V., Marochkin I.I., Rykov A.N., Makhova N.N., Shishkov I.F.
Structural Chemistry,
2018
6.

Marochkin I.I., Kuznetsov V.V., Rykov A.N., Makhova N.N., Shishkov I.F.
Structural Chemistry,
2018
7.

Khaikin L.S., Kochikov I.V., Rykov A.N., Grikina O.E., Ageev G.G., Shishkov I.F., Kuznetsov V.V., Makhova N.N.
Physical Chemistry Chemical Physics,
2019
8.
![Equilibrium molecular structure and spectra of 6-methyl-1,5-diazabicyclo[3.1.0]hexane: Joint analysis of gas phase electron diffraction, quantum chemistry, and spectroscopic data](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Khaikin L.S., Ageev G.G., Rykov A.N., Grikina O.E., Shishkov I.F., Kochikov I.V., Kuznetsov V.V., Makhova N.N., Bukalov S.S., Leites L.A.
Physical Chemistry Chemical Physics,
2020
9.
![Equilibrium Molecular Structure of 3,3,6-trimethyl-1,5-diazabicyclo[3.1.0]hexane: the joint analysis of the gas-phase electron diffraction data and quantum chemical simulations](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ageev G.G., Rykov A.N., Grikina O.E., Shishkov I.F., Kochikov I.V., Kuznetsov V.V., Makhova N.N., Bukalov S.S.
Structural Chemistry,
2021
10.

Paget C.J., Davis C.S.
Journal of Medicinal Chemistry,
1964
11.

Baichurina A.Z., Semina I.I., Garaev R.S.
Bulletin of Experimental Biology and Medicine,
1996
12.
N. N. Makhova, V. Y. Petukhova, A. V. Shevtsov, V. V. Novakovskiy and V. V. Kuznetsov, Patent WO 2013/111117 A2, 2013.
13.

Mannschreck A., Radeglia R., Gründemann E., Ohme R.
European Journal of Inorganic Chemistry,
1967
14.

Shustov G.V., Kadorkina G.K., Varlamov S.V., Kachanov A.V., Kostyanovskii R.G., Rauk A.
Journal of the American Chemical Society,
1992
15.

16.

Chagarovskiy A.O., Vasin V.S., Kuznetsov V.V., Ivanova O.A., Rybakov V.B., Shumsky A.N., Makhova N.N., Trushkov I.V.
Angewandte Chemie - International Edition,
2018
17.

Hu H., Xu J., Wang F., Dong S., Liu X., Feng X.
Organic Letters,
2019
18.

Ravindra S., Irfana Jesin C.P., Shabashini A., Nandi G.C.
Advanced Synthesis and Catalysis,
2021
19.
![Sc(OTf)3-Catalyzed Formal [3 + 3] Cycloaddition Reaction of Diaziridines and Quinones for the Synthesis of Benzo[e][1,3,4]oxadiazines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cortes Vazquez J., Davis J., Nesterov V.N., Wang H., Luo W.
Organic Letters,
2021
20.
![Synthesis and Ignition Properties Research of 1,5-Diazabicyclo[3.1.0]Hexane Type Compounds as Potential Green Hypergolic Propellants](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zhang X., Shen L., Luo Y., Jiang R., Sun H., Liu J., Fang T., Fan H., Liu Z.
Industrial & Engineering Chemistry Research,
2017
21.

Borretto E., Lazzarato L., Spallotta F., Cencioni C., D’Alessandra Y., Gaetano C., Fruttero R., Gasco A.
ACS Medicinal Chemistry Letters,
2013
22.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
23.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
24.

Fershtat L.L., Epishina M.A., Kulikov A.S., Struchkova M.I., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
25.
Fershtat L.L., Epishina M.A., Kulikov A.S., Makhova N.N.
Mendeleev Communications,
2015
26.

Auclair A.L., Martel J.C., Assié M.B., Bardin L., Heusler P., Cussac D., Marien M., Newman-Tancredi A., O'Connor J.A., Depoortère R.
Neuropharmacology,
2013
27.
![Enantiopure Cyclopropane-Bearing Pyridyldiazabicyclo[3.3.0]octanes as Selective α4β2-nAChR Ligands](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Onajole O.K., Eaton J.B., Lukas R.J., Brunner D., Thiede L., Caldarone B.J., Kozikowski A.P.
ACS Medicinal Chemistry Letters,
2014
28.

Talele T.T.
Journal of Medicinal Chemistry,
2016
29.

Lutfy K., Cowan A.
Current Neuropharmacology,
2004
30.

Domagala J.M.
Journal of Antimicrobial Chemotherapy,
1994
31.

YOSHIDA M., EZAKI M., HASHIMOTO M., YAMASHITA M., SHIGEMATSU N., OKUHARA M., KOHSAKA M., HORIKOSHI K.
Journal of Antibiotics,
1990
32.

Tomilov Y.V., Menchikov L.G., Novikov R.A., Ivanova O.A., Trushkov I.V.
Russian Chemical Reviews,
2018
33.
10.1016/j.mencom.2022.07.015_b0115
Frisch
Gaussian 09,
2009
34.

Møller C., Plesset M.S.
Physical Review A,
1934
35.

Becke A.D.
Journal of Chemical Physics,
1993
36.

Lee C., Yang W., Parr R.G.
Physical Review B,
1988
37.

Dunning T.H.
Journal of Chemical Physics,
1989
38.

Sipachev V.A.
Journal of Molecular Structure,
2004
39.
Y. V. Vishnevskiy, UNEX version 1.6, http://unexprog.org, 2022.