Keywords
s-triazine
123-triazoles
ABB' monomers
alkynes
azides
calculated Gibbs free energy
hyperbranched polymers
M06-2X/6-311++G(d p)
Abstract
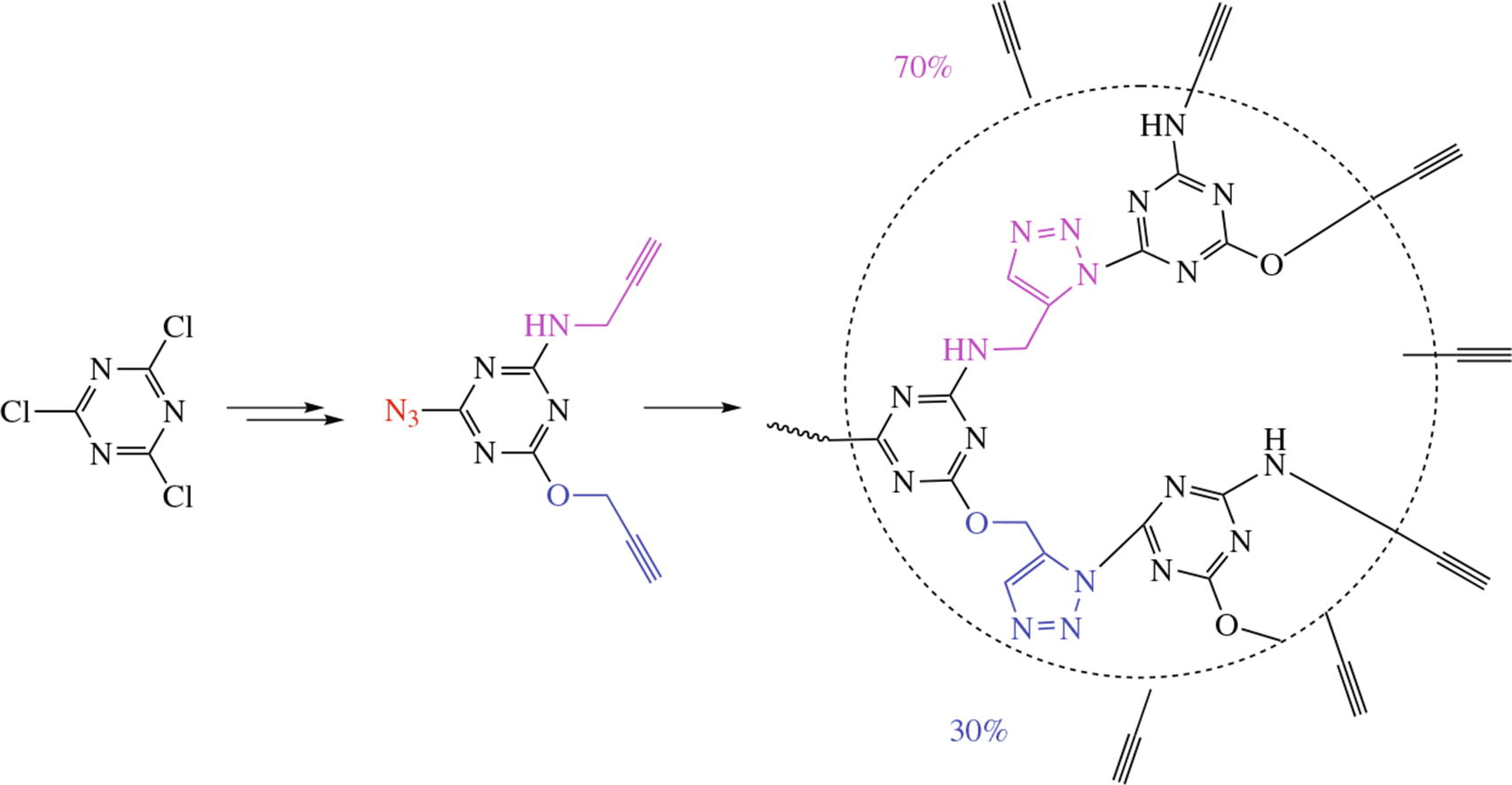
An original synthesis of a new non-symmetric energetic ABB' type monomer, 2-azido-4-propargylamino-6- propargyloxy-s-triazine, with a total yield of 40% involves the sequential introduction of propargylamino, propargyloxy and azido groups into s-triazine. DFT investigation of azide- Cl alkyne cycloaddition mechanism at M06-2X/6-311++G(d,p) level of theory for this monomer predicts that the regioselectivity of polycycloaddition reaction should increase with the number of propargylamino groups in the monomer structure due to the stabilization of the transition state, leading to 1,5-triazole regioisomer.
References
1.
Sheremetev A.B.
Mendeleev Communications,
2020
2.

Pant C., Wagh R., Nair J., Gore G., Thekkekara M., Venugopalan S.
Propellants, Explosives, Pyrotechnics,
2007
3.
Tarasov A.E., Perepelitsina E.O., Romanova L.B., Darovskikh A.V., Smirnov V.S., Badamshina E.R., Mikhailov Y.M.
Mendeleev Communications,
2022
4.

Sagot E., Le Roux A., Soulivet C., Pasquinet E., Poullain D., Girard E., Palmas P.
Tetrahedron,
2007
5.

He G., Li X., Bai L., Meng L., Dai Y., Sun Y., Zeng C., Yang Z., Yang G.
Composites Part B: Engineering,
2020
6.

Zakharov V.V., Chukanov N.V., Larikova T.S., Shilov G.V., Korepin A.G., Pivkina A.N., Monogarov K.A., Korsunskiy B.L., Korchagin D.V., Aldoshin S.M.
Russian Chemical Bulletin,
2020
7.

Zang X., Liu H., Li Q., Li Z., Li Z.
Polymer Chemistry,
2020
8.
10.1016/j.mencom.2022.07.012_b0040
Pacini
J. Polym. Sci.,
2014
9.

Rezania H.
Designed Monomers and Polymers,
2020
10.

Jeong H.J., Kim B.K.
Reactive and Functional Polymers,
2017
11.

Karpov S.V., Perepelitsina E.O., Malkov G.V.
Polymer Science - Series B,
2014
12.
![Synthesis and Characterization of the Nitrogen-Rich Hyperbranched Polymers - Poly([1,2,3]-Triazole-[1,3,5]-Triazine)s](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Malkov G., Shastin A., Estrin Y., Badamshina E., Mikhailov Y.
Propellants, Explosives, Pyrotechnics,
2008
13.

Patil A.J., Muthusamy E., Mann S.
Angewandte Chemie - International Edition,
2004
14.

Li J., Wang S., Liao L., Ma Q., Zhang Z., Fan G.
New Journal of Chemistry,
2019
15.

Shastin A.V., Petrov A.O., Malkov G.V., Gavrishova T.N.
Chemistry of Heterocyclic Compounds,
2021
16.
Karpov S.V., Petrov A.O., Malkov G.V., Badamshina E.R.
Mendeleev Communications,
2022
17.

Petrov A.O., Malkov G.V., Karpov S.V., Shastin A.V., Bakeshko A.V.
Key Engineering Materials,
2019
18.
19.

Karpov S.V., Iakunkov A.G., Malkov G.V.
Key Engineering Materials,
2019
20.

Green S.P., Payne A.D., Wheelhouse K.M., Hallett J.P., Miller P.W., Bull J.A.
Journal of Organic Chemistry,
2019
21.

Jadhav H., Annapure U.
Journal of Chemical Sciences,
2021
22.

Chen D., Yang H., Yi Z., Xiong H., Zhang L., Zhu S., Cheng G.
Angewandte Chemie - International Edition,
2018
23.

Cabral B.J., Costa M.L.
Journal of Molecular Structure THEOCHEM,
1995
24.

Karpov S.V., Lodygina V.P., Malkov G.V.
Journal of Applied Spectroscopy,
2014
25.

Zhao Y., Truhlar D.G.
Journal of Chemical Theory and Computation,
2008
26.

Cohen A.J., Mori-Sánchez P., Yang W.
Chemical Reviews,
2011
27.

Peng C., Bernhard Schlegel H.
Israel Journal of Chemistry,
1993
28.

Peng C., Ayala P.Y., Schlegel H.B., Frisch M.J.
Journal of Computational Chemistry,
1996