Keywords
biodegradable polymers
DFT calculations
diamido amines
E-caprolactone
ring-opening polymerization
stannylenes
Abstract
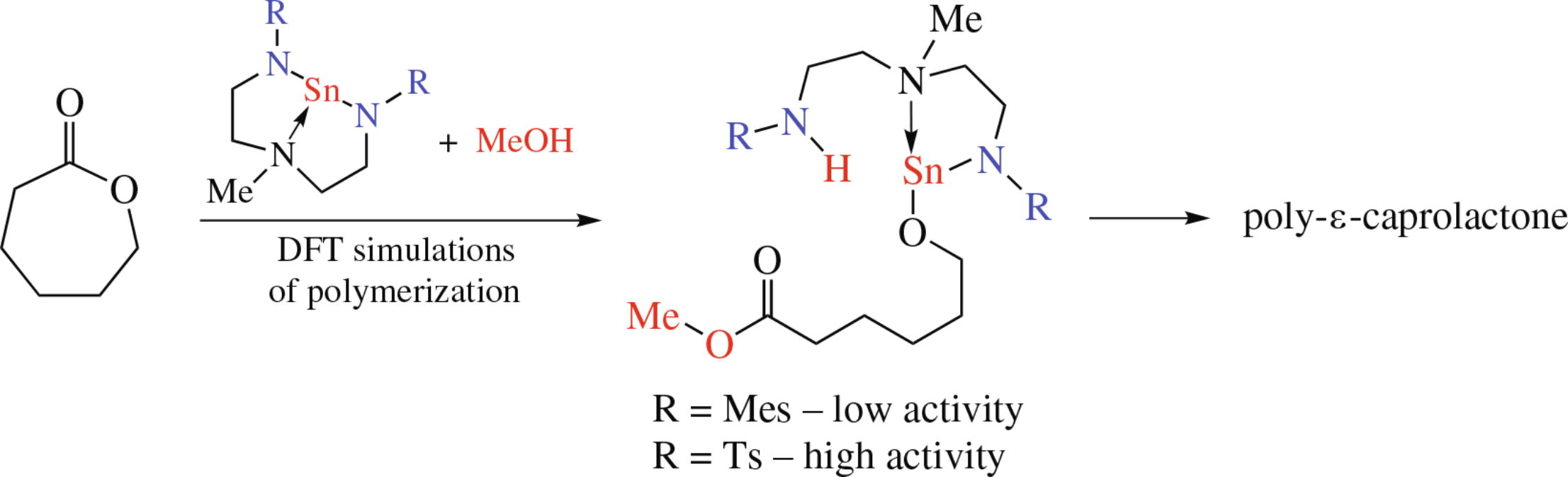
DFT simulations of ring-opening polymerization of ε-caprolactone in the presence of two stannylenes based on bis(2-amidoethyl)amine ligands demonstrated that rate limiting step of the whole process is the nucleophilic attack of a metal initiator with the formation of the tetrahedral carbon from sp2 carbon atom of the carboxy group. The presence of electron-withdrawing groups at the terminal nitrogen atoms of the ligands leads to decrease in the activation energy of the rate limiting step.
References
1.
Biodegradable and Biocompatible Polymer Composites: Processing, Properties and Applications, ed. N. G. Shimpi, Woodhead Publishing, 2017.
2.

Elsawy M.A., Kim K., Park J., Deep A.
Renewable and Sustainable Energy Reviews,
2017
3.

Sarazin Y., Carpentier J.
Chemical Reviews,
2015
4.

Dechy-Cabaret O., Martin-Vaca B., Bourissou D.
Chemical Reviews,
2004
5.

Degée P., Dubois P., Jérǒme R., Jacobsen S., Fritz H.
Macromolecular Symposia,
1999
6.

Nakayama Y., Aihara K., Cai Z., Shiono T., Tsutsumi C.
International Journal of Molecular Sciences,
2017
7.
Nifant’ev I.E., Shlyakhtin A.V., Bagrov V.V., Ezhov R.N., Lozhkin B.A., Churakov A.V., Ivchenko P.V.
Mendeleev Communications,
2018
8.
Lozhkin B.A., Shlyakhtin A.V., Bagrov V.V., Ivchenko P.V., Nifant’ev I.E.
Mendeleev Communications,
2018
9.
Nifant’ev I.E., Kosarev M.A., Shlyakhtin A.V., Tavtorkin A.N., Minyaev M.E., Ivchenko P.V.
Mendeleev Communications,
2020
10.
Kuchuk E.A., Mankaev B.N., Serova V.A., Zaitsev K.V., Churakov A.V., Oprunenko Y.F., Zaitseva G.S., Karlov S.S.
Mendeleev Communications,
2020
11.
![Neodymium monochloride and monoallyl complexes {2-[Ph2P(O)]C6H4NC(But)N(2,6-Me2C6H3)}2NdR (R = Cl, CH2CH=CH2) with the tridentate amidinate ligand in the catalysis of ring-opening polymerization of cyclic esters](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Tolpygin A.O., Linnikova O.A., Kovylina T.A., Cherkasov A.V., Fukin G.K., Trifonov A.A.
Russian Chemical Bulletin,
2020
12.

Bhaw-Luximon A., Jhurry D., Spassky N.
Polymer Bulletin,
2000
13.

Alcazar-Roman L.M., O'Keefe B.J., Hillmyer M.A., Tolman W.B.
Dalton Transactions,
2003
14.

Hormnirun P., Marshall E.L., Gibson V.C., White A.J., Williams D.J.
Journal of the American Chemical Society,
2004
15.

Hormnirun P., Marshall E.L., Gibson V.C., Pugh R.I., White A.J.
Proceedings of the National Academy of Sciences of the United States of America,
2006
16.

Gregson C.K., Blackmore I.J., Gibson V.C., Long N.J., Marshall E.L., White A.J.
Dalton Transactions,
2006
17.
10.1016/j.mencom.2022.07.011_b0085
Du
Macromolecules,
1904
18.

Nomura N., Ishii R., Yamamoto Y., Kondo T.
Chemistry - A European Journal,
2007
19.

Tang Z., Gibson V.C.
European Polymer Journal,
2007
20.

Bouyahyi M., Ajellal N., Kirillov E., Thomas C.M., Carpentier J.
Chemistry - A European Journal,
2011
21.

22.

Mankaev B.N., Zaitsev K.V., Kuchuk E.A., Vershinina M.V., Zaitseva G.S., Egorov M.P., Karlov S.S.
Russian Chemical Bulletin,
2019
23.

Nifant’ev I., Ivchenko P.
Molecules,
2019
24.

Sattayanon C., Sontising W., Jitonnom J., Meepowpan P., Punyodom W., Kungwan N.
Computational and Theoretical Chemistry,
2014
25.

Chen T., Qin Z., Qi Y., Deng T., Ge X., Wang J., Hou X.
Polymer Chemistry,
2011
26.

Ryner M., Stridsberg K., Albertsson A., von Schenck H., Svensson M.
Macromolecules,
2001
27.

Wang L., Kefalidis C.E., Sinbandhit S., Dorcet V., Carpentier J., Maron L., Sarazin Y.
Chemistry - A European Journal,
2013
28.

Sattayanon C., Kungwan N., Punyodom W., Meepowpan P., Jungsuttiwong S.
Journal of Molecular Modeling,
2013
29.

Zabalov M.V., Tiger R.P.
Russian Chemical Bulletin,
2016
30.

Zabalov M.V., Tiger R.P.
Theoretical Chemistry Accounts,
2017