Keywords
1-halo-1-nitroethenes
furo[32-c]chromenes
furo[32-c]pyrans
heterocyclization
nitroacrylates
Abstract
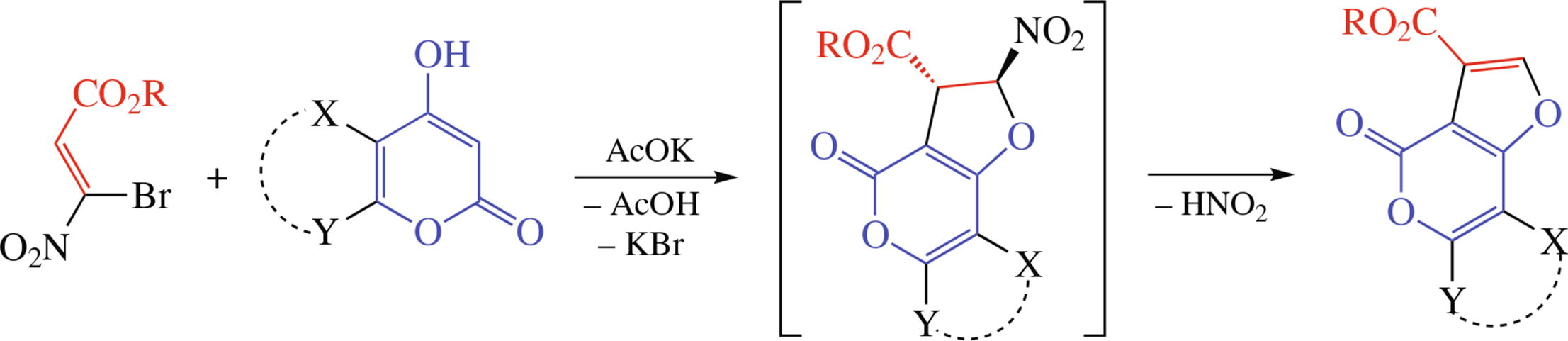
Novel 4-oxo-4H-furo[3,2-c]pyran-3-carboxylates and 4-oxo- 4H-furo[3,2-c]chromene-3-carboxylates were prepared from available alkyl 3-bromo-3-nitroacrylates and 4-hydroxy-6- methyl-2H-pyran-2-one or 4-hydroxycoumarin, respectively. Their structures were confirmed by NMR and X-ray data.
References
1.

Traven V.
Molecules,
2004
2.

Melough M.M., Cho E., Chun O.K.
Food and Chemical Toxicology,
2018
3.

Thorat S.S., Kontham R.
Organic Chemistry Frontiers,
2021
4.

Santana L., Uriarte E., Roleira F., Milhazes N., Borges F.
Current Medicinal Chemistry,
2004
5.
![Furo[3,2-c]coumarins carrying carbon substituents at C-2 and/or C-3. Isolation, biological activity, synthesis and reaction mechanisms](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Cortés I., Cala L.J., Bracca A.B., Kaufman T.S.
RSC Advances,
2020
6.
10.1016/j.mencom.2022.07.009_b0030
Rani
Beilstein Arch.,
2021
7.
![Furo[3,2-c]coumarin-derived Fe3+ Selective Fluorescence Sensor: Synthesis, Fluorescence Study and Application to Water Analysis](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sarih N.M., Ciupa A., Moss S., Myers P., Slater A.G., Abdullah Z., Tajuddin H.A., Maher S.
Scientific Reports,
2020
8.
![Synthesis of novel push-pull fluorescent dyes – 7-(diethylamino)furo[3,2-c]coumarin and 7-(diethylamino)thieno[3,2-c]coumarin derivatives](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Akchurin I.O., Yakhutina A.I., Bochkov A.Y., Solovjova N.P., Traven V.F.
Heterocyclic Communications,
2018
9.
![Synthesis and Photooxygenation of Furo[3,2-c]coumarin Derivatives as Antibacterial and DNA Intercalating Agent](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Al-Sehemi A.G., El-Gogary S.R.
Chinese Journal of Chemistry,
2012
10.
![Synthesis and antimicrobial activity of some new 4-hetarylpyrazole and furo[2,3-c]pyrazole derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bondock S., Khalifa W., Fadda A.A.
European Journal of Medicinal Chemistry,
2011
11.
![Microwave-Assisted Synthesis and Antifungal Activities of Polysubstituted Furo[3,2-c]chromen-4-ones and 7,8,9,10-Tetrahydro-6H-benzofuro[3,2-c]chromen-6-ones](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Zhang R., Xu Z., Yin W., Liu P., Zhang W.
Synthetic Communications,
2014
12.

Ghosh M., Hajra A.
European Journal of Organic Chemistry,
2015
13.
10.1016/j.mencom.2022.07.009_b0065
Singh
RSC Adv.,
2015
14.

Dou X., Zhong F., Lu Y.
Chemistry - A European Journal,
2012
15.
Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
Mendeleev Communications,
2019
16.
![Optimization of the Synthesis of Benzo[b]furan-3-carboxylates Based on Alkyl 3-Bromo-3-nitroacrylates](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Pelipko V.V., Baichurin R.I., Kondrashov E.V., Makarenko S.V.
Russian Journal of General Chemistry,
2021
17.

Makarenko S.V., Pelipko V.V., Pavchenko M.M., Baichurin R.I.
Russian Journal of General Chemistry,
2019
18.

Baichurin R.I., Lyssenko K.A., Kovalenko K.S., Makarenko S.V.
Russian Chemical Bulletin,
2020
19.

Pelipko V.V., Gomonov K.A., Litvinov I.A., Baichurin R.I., Makarenko S.V.
Russian Chemical Bulletin,
2022
20.

Kuritsyna M.A., Pelipko V.V., Kataeva O.N., Baichurin R.I., Sadikov K.D., Smirnov A.S., Makarenko S.V.
Russian Chemical Bulletin,
2021
21.
10.1016/j.mencom.2022.07.009_b0105
Lee
Org. Lett.,
1906
22.
I. Koca, I. Yildirim and E. S¸ahin, Helv. Chim. Acta, 2010, 93, 1336.
23.
N. Yu. Lisovenko, A. V. Dryahlov and M. V. Dmitriev, Russ. J. Org. Chem., 2016, 52, 1183 (Zh. Org. Khim., 2016, 52, 1190).
24.
![Nouvelle méthode d'accès aux benzo[h]furo[3,2-c]quinoléines, analogues isostériques des benzo[c]phénanthridines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Duval O., Mavoungou Gomès L.
Journal of Heterocyclic Chemistry,
1991
25.

Haasnoot C.A., de Leeuw F.A., Altona C.
Tetrahedron,
1980
26.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007