Keywords
aza-Diels–Alder reaction
ipso-substitution
124-triazines
2'-bipyridines
amino-2
enamines
one-pot synthesis
solvent-free reaction
Abstract
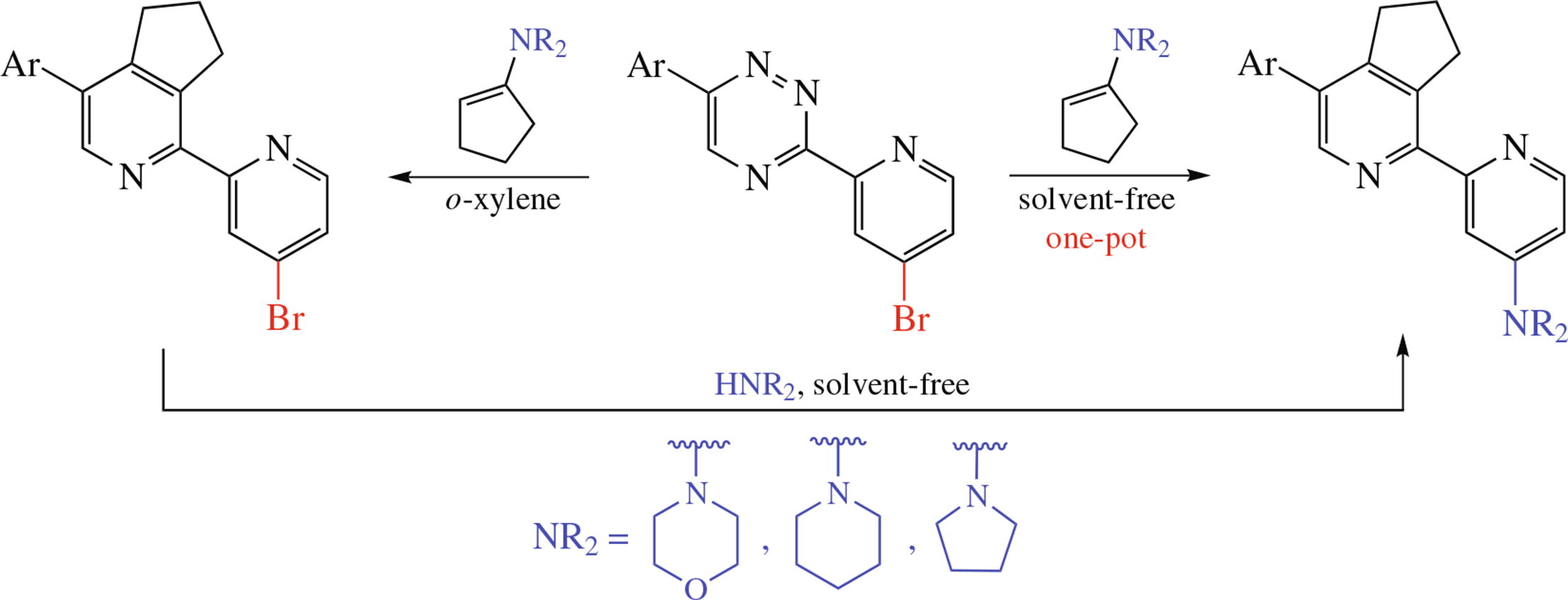
One-pot synthesis of cyclopentane-fused 5'-aryl-4- cycloalkylamino-2,2'-bipyridines based on the neat (200 °C) reaction of 3-(4-bromopyridin-2-yl)-1,2,4-triazines with enamines is reported. In the course of the transformation, consecutive aza-Diels–Alder reaction and nucleophilic substitution of bromine atom under the action of the liberating amine occur. The possibility of the solvent- and catalyst-free replacement of 4-positioned bromine atom in 2,2'-bipyridines by amino moieties was demonstrated.
References
1.

Prokhorov A.M., Kozhevnikov D.N.
Chemistry of Heterocyclic Compounds,
2012
2.

Boger D.L., Panek J.S.
Journal of Organic Chemistry,
1981
3.

Kozhevnikov D.N., Kozhevnikov V.N., Prokhorov A.M., Ustinova M.M., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G., König B.
Tetrahedron Letters,
2006
4.

Moseev T.D., Varaksin M.V., Gorlov D.A., Nikiforov E.A., Kopchuk D.S., Starnovskaya E.S., Khasanov A.F., Zyryanov G.V., Charushin V.N., Chupakhin O.N.
Journal of Fluorine Chemistry,
2019
5.

Kovalev I.S., Savchuk M.I., Kopchuk D.S., Zyryanov G.V., Pospelova T.A., Rusinov V.L., Chupakhin O.N.
Russian Journal of Organic Chemistry,
2019
6.
Fatykhov R.F., Savchuk M.I., Starnovskaya E.S., Bobkina M.V., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Khalymbadzha I.A., Chupakhin O.N., Charushin V.N., Kartsev V.G.
Mendeleev Communications,
2019
7.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
8.

Taylor R., Catozzi N., Bromley W., Wasnaire P., Gibson M.
Synlett,
2007
9.
(b) S. V. Shorshnev, S. E. Esipov, A. I. Chernyshev, A. F. Pozharskii, V. V. Kuz'menko and A. V. Gulevskaya, Chem. Heterocycl. Compd., 1990, 26, 191 (Khim. Geterotsikl. Soedin., 1990, 224)
10.
![Cycloaddition Reactions with Azabenzenes, XVIII. Synthesis of [2]Pyrindines](/storage/images/resized/SovkqI6gX6to8o5XBnS9RNxEOYVicn7hatkMzrvl_small_thumb.webp)
Neunhoeffer H., Neuhoeffer H., Philipp B., Schildhauer B., Eckrich R., Krichbaum U.
Heterocycles,
1993
11.

Gonsalves A.M., Pinho e Melo T.M., Gilchrist T.L.
Tetrahedron,
1992
12.

Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
13.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Gundala S., Krinochkin A.P., Slepukhin P.А., Zyryanov G.V., Venkatapuram P., Chupakhin O.N., Charushin V.N.
Chemistry of Heterocyclic Compounds,
2019
14.

Diring S., Retailleau P., Ziessel R.
Journal of Organic Chemistry,
2007
15.

Wang S., Guo W., Wen L., Li M.
RSC Advances,
2014
16.
Zyryanov G.V., Kopchuk D.S., Kovalev I.S., Santra S., Rahman M., Khasanov A.F., Krinochkin A.P., Taniya O.S., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2020
17.
Grin M.A., Suvorov N.V., Mironov A.F.
Mendeleev Communications,
2020
18.
2-Aminooxazoles as novel dienophiles in the inverse demand Diels–Alder reaction with 1,2,4-triazines
Krinochkin A.P., Mallikarjuna Reddy G., Kopchuk D.S., Slepukhin P.A., Shtaitz Y.K., Khalymbadzha I.A., Kovalev I.S., Kim G.A., Ganebnykh I.N., Zyryanov G.V., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2021
19.

Kozhevnikov V.N., Kozhevnikov D.N., Shabunina O.V., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2005
20.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
21.

Dey B.B.
Journal of the Chemical Society Transactions,
1914
22.

Kozhevnikov V.N., Ustinova M.M., Slepukhin P.A., Santoro A., Bruce D.W., Kozhevnikov D.N.
Tetrahedron Letters,
2008
23.

Starnovskaya E.S., Kopchuk D.S., Khasanov A.F., Tanya O.S., Santra S., Giri K., Rahman M., Kovalev I.S., Zyryanov G.V., Majee A., Charushin V.N.
Dyes and Pigments,
2019
24.

Shen Q., Ogata T., Hartwig J.F.
Journal of the American Chemical Society,
2008
25.
(b) J. Bach Tana, D. Perez Crespo, O. Llera Soldevila, C. Esteve Trias and L. Taboada Martinez, Patent WO 2015/86693 A1, 2015
26.

Ji J., Li T., Bunnelle W.H.
Organic Letters,
2003
27.

Verdelet T., Mercey G., Correa N., Jean L., Renard P.
Tetrahedron,
2011
28.

Kalkman E.D., Mormino M.G., Hartwig J.F.
Journal of the American Chemical Society,
2019
29.

Kohl T., Hornung C., Tsanaktsidis J.
Molecules,
2015
30.

Fallahpour R.
Synthesis,
2008
31.
10.1016/j.mencom.2022.07.007_b0100
Whittle
J. Chem. Soc., Dalton Trans.,
1996
32.
J. R. Allen, K. Biswas, G.-Q. Cao, J. E. Golden, S. Mercede, T. Peterkin, A. Reed and C. M. Tegley, Patent WO 2008/76425 A1, 2008.
33.

Heald R., Bowman K.K., Bryan M.C., Burdick D., Chan B., Chan E., Chen Y., Clausen S., Dominguez-Fernandez B., Eigenbrot C., Elliott R., Hanan E.J., Jackson P., Knight J., La H., et. al.
Journal of Medicinal Chemistry,
2015
34.
J. Rheinheimer, F. Schieweck, T. Grote, C. Blettner, A. Schwogler, M. Gewehr, W. Grammenos, U. Hunger, B. Muller, P. Schafer, J.-B. Speakman, M. Scherer, S. Strathmann, U. Schofl and R. Stierl, Patent WO 2006/000358 A1, 2006.
35.

Kireev D., Chrétien J., Raevsky O.
European Journal of Medicinal Chemistry,
1995
36.

Zhang F., Bhat S., Gabelli S.B., Chen X., Miller M.S., Nacev B.A., Cheng Y.L., Meyers D.J., Tenney K., Shim J.S., Crews P., Amzel L.M., Ma D., Liu J.O.
Journal of Medicinal Chemistry,
2013
37.

Mazuryk O., Łomzik M., Martineau D., Beley M., Brindell M., Stochel G., Gros P.C.
Inorganica Chimica Acta,
2016
38.

Nazeeruddin M.K., Wegh R.T., Zhou Z., Klein C., Wang Q., De Angelis F., Fantacci S., Grätzel M.
Inorganic Chemistry,
2006
39.

Nagata K., Otsuji N., Akagi S., Fujii S., Kitamura N., Yoshimura T.
Inorganic Chemistry,
2020