Keywords
34-diarylbutadienes
catalytic cyclization
Friedel–Crafts reaction
indenes
one-pot synthesis
sulfones
sulfur dioxide
thermal extrusion
Abstract
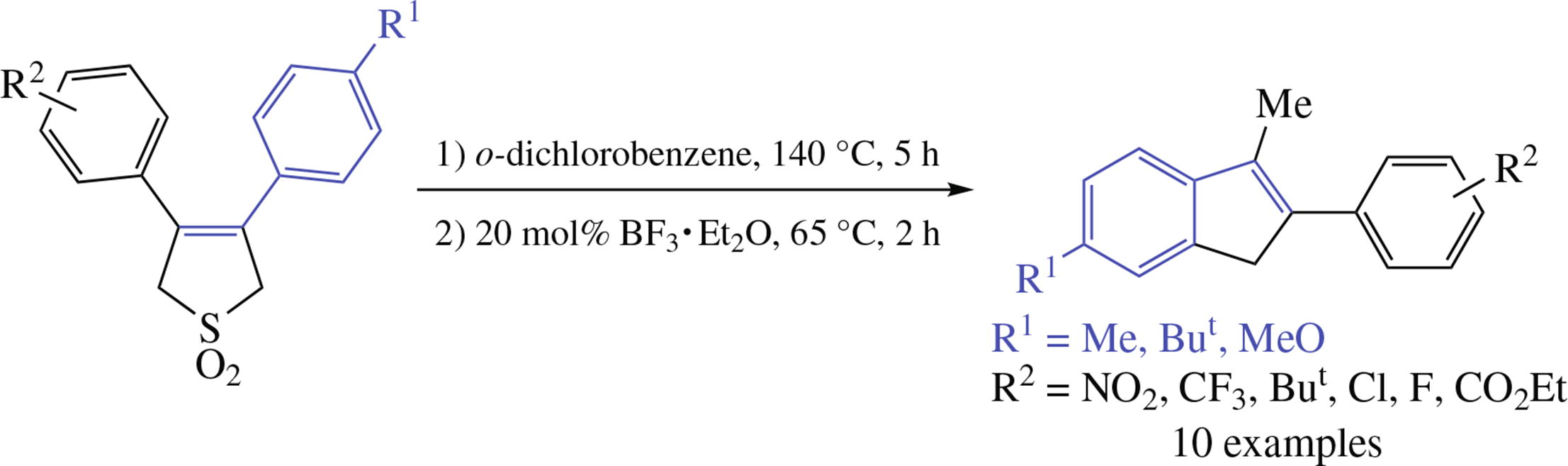
A new one-pot two step synthesis of unsymmetrically substituted indenes from available 3,4-diarylbutadiene sulfones involves SO2 thermal extrusion followed by acid- catalyzed cyclization of the diene formed, the cyclization proceeding selectively at the more electron-rich aryl rings. The procedure is efficient for substrates bearing donor, acceptor, as well as bulky substituents.
References
1.

Maguire A.R., Papot S., Ford A., Touhey S., O'Connor R., Clynes M.
Synlett,
2001
2.

Hagishita S., Yamada M., Shirahase K., Okada T., Murakami Y., Ito Y., Matsuura T., Wada M., Kato T., Ueno M., Chikazawa Y., Yamada K., Ono T., Teshirogi I., Ohtani M., et. al.
Journal of Medicinal Chemistry,
1996
3.

Lee B.H., Choi Y.L., Shin S., Heo J.
Journal of Organic Chemistry,
2011
4.

Majetich G., Shimkus J.M.
Journal of Natural Products,
2010
5.

Senanayake C.H., Roberts F.E., DiMichele L.M., Ryan K.M., Liu J., Fredenburgh L.E., Foster B.S., Douglas A.W., Larsen R.D., Verhoeven T.R., Reider P.J.
Tetrahedron Letters,
1995
6.
10.1016/j.mencom.2022.07.006_b0030
Korte
Synlett,
2004
7.

Li C., Black W.C., Chan C., Ford-Hutchinson A.W., Gauthier J., Gordon R., Guay D., Kargman S., Lau C.K.
Journal of Medicinal Chemistry,
1995
8.

Nord C., Menkis A., Broberg A.
Molecules,
2014
9.

Karaguni I., Glüsenkamp K., Langerak A., Geisen C., Ullrich V., Winde G., Möröy T., Müller O.
Bioorganic and Medicinal Chemistry Letters,
2002
10.

Citta A., Folda A., Bindoli A., Pigeon P., Top S., Vessières A., Salmain M., Jaouen G., Rigobello M.P.
Journal of Medicinal Chemistry,
2014
11.

Differential Response of Estrogen Receptor Subtypes to 1,3-Diarylindene and 2,3-Diarylindene Ligands
Clegg N.J., Paruthiyil S., Leitman D.C., Scanlan T.S.
Journal of Medicinal Chemistry,
2005
12.

Gao H., Katzenellenbogen J.A., Garg R., Hansch C.
Chemical Reviews,
1999
13.

Barberá J., Rakitin O.A., Ros M.B., Torroba T.
Angewandte Chemie - International Edition,
1998
14.
![Synthesis and Characterization of 5,10-Bis(2-thienyl)indeno[2,1-a]indene Derivatives: The First Examples of Conducting Polymers Containing a Rigid Bis(thienyl)butadiene Core](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Yang J., Lakshmikantham M.V., Cava M.P., Lorcy D., Bethelot J.R.
Journal of Organic Chemistry,
2000
15.

Dankova M., Kravchenko R.L., Cole A.P., Waymouth R.M.
Macromolecules,
2002
16.

Alt H.G., Köppl A.
Chemical Reviews,
2000
17.

Rinaldi A., Scarpi D., Occhiato E.G.
European Journal of Organic Chemistry,
2019
18.

Gabriele B., Mancuso R., Veltri L.
Chemistry - A European Journal,
2016
19.

Chakraborty M., Mahesh G., Nakel O.R., Chavda G., Anusha S., Sudhakar G.
ChemistrySelect,
2021
20.

Martynov M.Y., Iakovenko R.O., Kazakova A.N., Boyarskaya I.A., Vasilyev A.V.
Organic and Biomolecular Chemistry,
2017
21.

Holt C., Alachouzos G., Frontier A.J.
Journal of the American Chemical Society,
2019
22.

Kotipalli T., Hou D.
Asian Journal of Organic Chemistry,
2019
23.

Kotipalli T., Hou D.
Organic Letters,
2018
24.

Deng R., Sun L., Li Z.
Organic Letters,
2007
25.

Miyamoto M., Harada Y., Tobisu M., Chatani N.
Organic Letters,
2008
26.

Mahesh S.K., Nanubolu J.B., Sudhakar G.
Journal of Organic Chemistry,
2019
27.

Ma B., Wu Z., Huang B., Liu L., Zhang J.
Chemical Communications,
2016
28.

Preinfalk A., Misale A., Maulide N.
Chemistry - A European Journal,
2016
29.

Niharika P., Satyanarayana G.
ChemistrySelect,
2018
30.

31.

Usanov D.L., Naodovic M., Brasholz M., Yamamoto H.
Helvetica Chimica Acta,
2012
32.

Nifant'ev I.E., Sitnikov A.A., Andriukhova N.V., Laishevtsev I.P., Luzikov Y.N.
Tetrahedron Letters,
2002
33.

Jiang H., He L., Li X., Chen H., Wu W., Fu W.
Chemical Communications,
2013
34.

Eom D., Park S., Park Y., Ryu T., Lee P.H.
Organic Letters,
2012
35.
Rzhevskiy S.A., Bogachev V.N., Minaeva L.I., Sterligov G.K., Nechaev M.S., Topchiy M.A., Asachenko A.F.
Mendeleev Communications,
2021
36.

Shurupova O.V., Rzhevskiy S.A., Minaeva L.I., Topchiy M.A., Asachenko A.F.
RSC Advances,
2022
37.

Chesnokov G.A., Ageshina A.A., Maryanova A.V., Rzhevskiy S.A., Gribanov P.S., Topchiy M.A., Nechaev M.S., Asachenko A.F.
Russian Chemical Bulletin,
2020
38.

Dzhevakov P.B., Topchiy M.A., Ageshina A.A., Minaeva L.I., Rzhevskiy S.A., Nechaev M.S., Osipov S.N., Asachenko A.F.
Russian Chemical Bulletin,
2020
39.
Topchiy M.A., Rzhevskiy S.A., Ageshina A.A., Kirilenko N.Y., Sterligov G.K., Mladentsev D.Y., Paraschuk D.Y., Osipov S.N., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2020
40.
Rzhevskiy S.A., Topchiy M.A., Bogachev V.N., Ageshina A.A., Minaeva L.I., Sterligov G.K., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2021
41.

Rzhevskiy S.A., Philippova A.N., Chesnokov G.A., Ageshina A.A., Minaeva L.I., Topchiy M.A., Nechaev M.S., Asachenko A.F.
Chemical Communications,
2021
42.
Rzhevskiy S.A., Topchiy M.A., Bogachev V.N., Minaeva L.I., Cherkashchenko I.R., Lavrov K.V., Sterligov G.K., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2021
43.
Gribanov P.S., Lypenko D.A., Dmitriev A.V., Pozin S.I., Topchiy M.A., Asachenko A.F., Loginov D.A., Osipov S.N.
Mendeleev Communications,
2021