Keywords
carene
clays
halloysite
heterogeneous catalysis
montmorillonite K10
pyrano[4,3-b]thieno[3,2-g]benzofuran
terpenoids
thiophene-2-carbaldehyde
Abstract
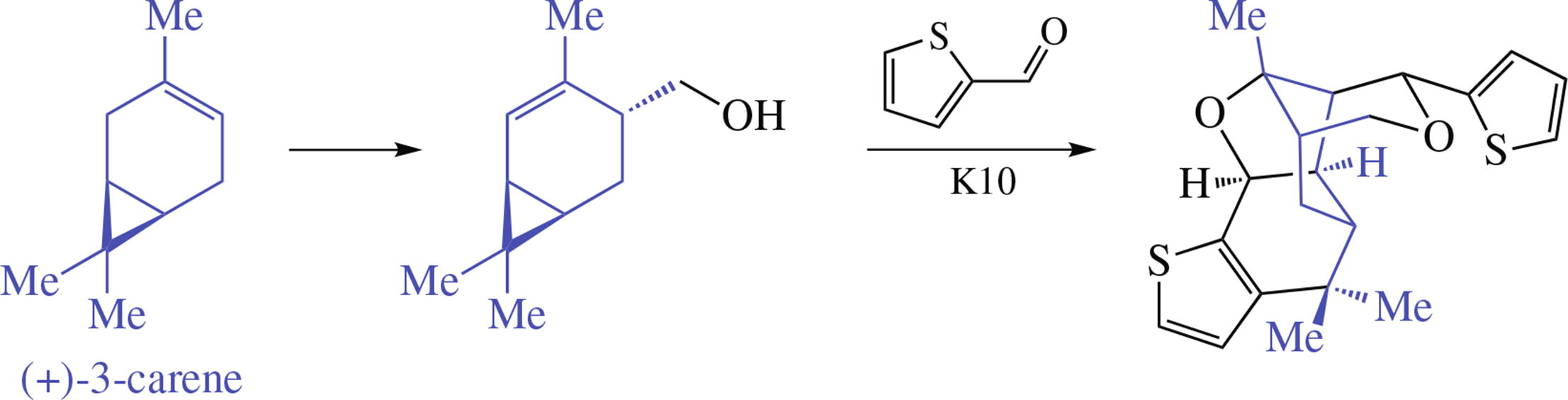
Reaction of trans-4-hydroxymethyl-2-carene with thiophene- 2-carbaldehyde under heterogeneous catalysis by K10 clay affordsubstitutedmethanopyrano[4,3-b]thieno[3,2-g]- benzofuran, a complex 5-membered chiral cage heterocyclic compound containing two thiophene moieties. Its structure was determined by 1D and 2D NMR experiments (HSQC, HMBC, COSY, NOESY) and HRMS.
References
1.

Sidorenko A.Y., Kurban Y.M., Il'ina I.V., Li-Zhulanov N.S., Korchagina D.V., Ardashov O.V., Wärnå J., Volcho K.P., Salakhutdinov N.F., Murzin D.Y., Agabekov V.E.
Applied Catalysis A: General,
2021
2.

Sidorenko A.Y., Kravtsova A.V., Mäki-Arvela P., Aho A., Sandberg T., Il'ina I.V., Li-Zhulanov N.S., Korchagina D.V., Volcho K.P., Salakhutdinov N.F., Murzin D.Y., Agabekov V.E.
Molecular Catalysis,
2020
3.

Salakhutdinov N.F., Volcho K.P., Yarovaya O.I.
Pure and Applied Chemistry,
2017
4.

Yarovaya O.I., Salakhutdinov N.F.
Russian Chemical Reviews,
2021
5.
10.1016/j.mencom.2022.07.005_b0025
de Alvarenga
Crit. Rev. Food Sci. Nutr.,
2021
6.

Minh Le T., Szakonyi Z.
Chemical Record,
2021
7.
10.1016/j.mencom.2022.07.005_b0035
Kozioł
Med. Chem.,
2014
8.

Barreto R., Albuquerque-Júnior R., Araújo A., Almeida J., Santos M., Barreto A., DeSantana J., Siqueira-Lima P., Quintans J., Quintans-Júnior L.
Molecules,
2014
9.
10.1016/j.mencom.2022.07.005_b0045
Dragomanova
Scr. Sci. Pharm.,
2018
10.

Marchese A., Orhan I.E., Daglia M., Barbieri R., Di Lorenzo A., Nabavi S.F., Gortzi O., Izadi M., Nabavi S.M.
Food Chemistry,
2016
11.

Jyoti, Dheer D., Singh D., Kumar G., Karnatak M., Chandra S., Prakash Verma V., Shankar R.
Current Bioactive Compounds,
2018
12.

Wojtunik‐Kulesza K.A., Kasprzak K., Oniszczuk T., Oniszczuk A.
Chemistry and Biodiversity,
2019
13.

Yuan P., Gerlinger C.K., Herberger J., Gaich T.
Journal of the American Chemical Society,
2021
14.

Jackson R.K., Wood J.L.
Organic Letters,
2021
15.

Nikitina L.E., Startseva V.A., Artemova N.P., Dorofeeva L.Y., Kuznetsov I.V., Lisovskaya S.A., Glushko N.P., Kutyreva M.P.
Pharmaceutical Chemistry Journal,
2012
16.

Nikitina L.E., Startseva V.A., Dorofeeva L.Y., Artemova N.P., Kuznetsov I.V., Lisovskaya S.A., Glushko N.P.
Chemistry of Natural Compounds,
2010
17.

Banina O.A., Sudarikov D.V., Slepukhin P.A., Frolova L.L., Kuchin A.V.
Chemistry of Natural Compounds,
2016
18.

Yakovleva M.P., Denisova K.S., Mingaleeva G.R., Myasoedova Y.V., Garifullina L.R., Ishmuratova N.M., Ishmuratov G.Y.
Chemistry of Natural Compounds,
2020
19.

Myasoedova Y.V., Nurieva E.R., Garifullina L.R., Ishmuratov G.Y.
Russian Journal of Organic Chemistry,
2020
20.

Macaev F.Z., Malkov A.V.
Tetrahedron,
2006
21.

Curlat S.N., Barba A.N., Boldescu V.V., Panekok K., Macaev F.Z.
Chemistry of Natural Compounds,
2019
22.
Ohloff G., Farnow H., Philipp W.
1958
23.

Sidorenko A.Y., Kurban Y.M., Kravtsova A.V., Il'ina I.V., Li-Zhulanov N.S., Korchagina D.V., Sánchez-Velandia J.E., Aho A., Volcho K.P., Salakhutdinov N.F., Murzin D.Y., Agabekov V.E.
Applied Catalysis A: General,
2022
24.

Patrusheva O.S., Volcho K.P., Salakhutdinov N.F.
Russian Chemical Reviews,
2018
25.

Salakhutdinov N.F., Volcho K.P., Il'ina I.V., Korchagina D.V., Tatarova L.E., Barkhash V.A.
Tetrahedron,
1998
26.
![The short way to chiral compounds with hexahydrofluoreno[9,1-bc]furan framework: synthesis and cytotoxic activity.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kurbakova S.Y., Il‘ina I.V., Mikhalchenko O.S., Pokrovsky M.A., Korchagina D.V., Volcho K.P., Pokrovsky A.G., Salakhutdinov N.F.
Bioorganic and Medicinal Chemistry,
2015
27.

Komadel P.
Applied Clay Science,
2016
28.

Il'ina I.V., Volcho K.P., Korchagina D.V., Salnikov G.E., Genaev A.M., Karpova E.V., Salakhutdinov N.F.
Helvetica Chimica Acta,
2010