Keywords
Abstract
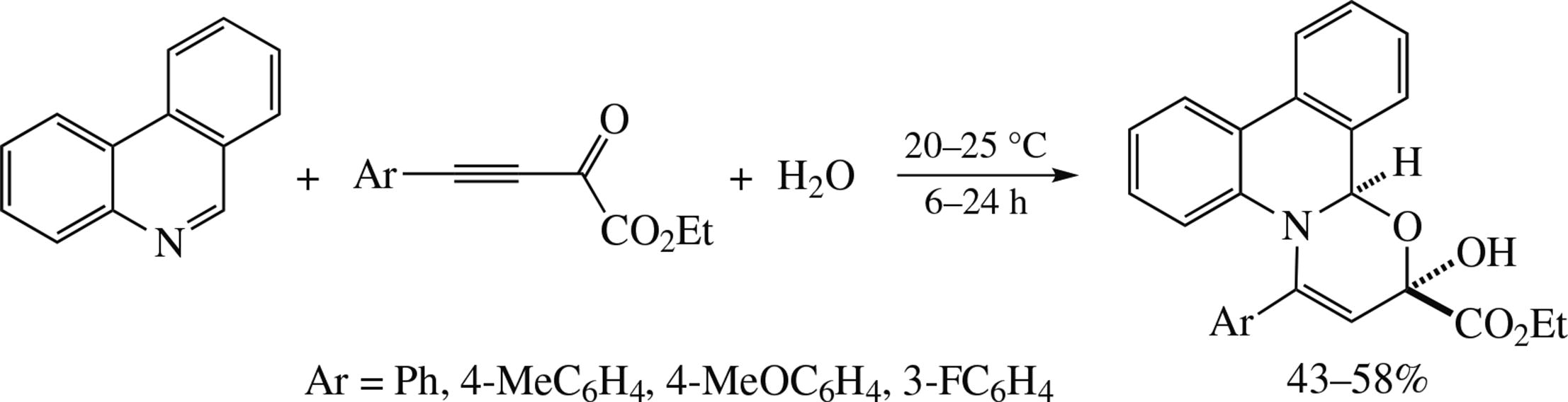
Phenanthridine is readily annulated at room temperature with dihydro-1,3-oxazine ring under the action of oxalylacetylene ethyl ester to diastereoselectively deliver (R*,R*)-2-hydroxy-4-aryl-2H,13bH-[1,3]oxazino[3,2-f]- phenanthridines in up to 58% yield. According to quantum chemical calculations, among the possible transformations of the 1,3(4)-dipole phenanthridine/oxalylacetylene inter- mediate, protonated with water, the preferred route leading to the target product is attack of the hydroxyl anion at the carbonyl moiety, followed by closing of the oxazine ring. The calculated stability of oxazinophenanthridine with respect to the oxazinoquinoline derivative in terms of ΔG, and the higher C6–O bond order in the former are confirmed by experimental results of differences between the reactions of phenanthridine and quinoline with aryloxalylacetylenes in water.
References

![Syntheses, Biological and Material Significance of Dihydro[1,3]oxazine Derivatives: An Overview](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)






![Phenanthridine synthesis via [2+2+2] cyclotrimerization reactions](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
![A Two-Step Synthesis of Cytostatically Active Benzo[c]phenanthridine Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)



![Synthesis of Fused [1,3]Oxazines from Ethyl Trifluoroacetate, Activated Acetylenes and N-Heterocycles](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)


![Uniquely functionalized tetrahydropyrido[2,1-b][1,3]oxazines: Diastereoselective 1:2 assembly from pyridines with oxalylacetylenes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)