Keywords
Antibacterial activity
bioavailability
cytotoxicity
glycol carbonates
hydrolysis
modified nucleosides
prodrugs
solubility
Abstract
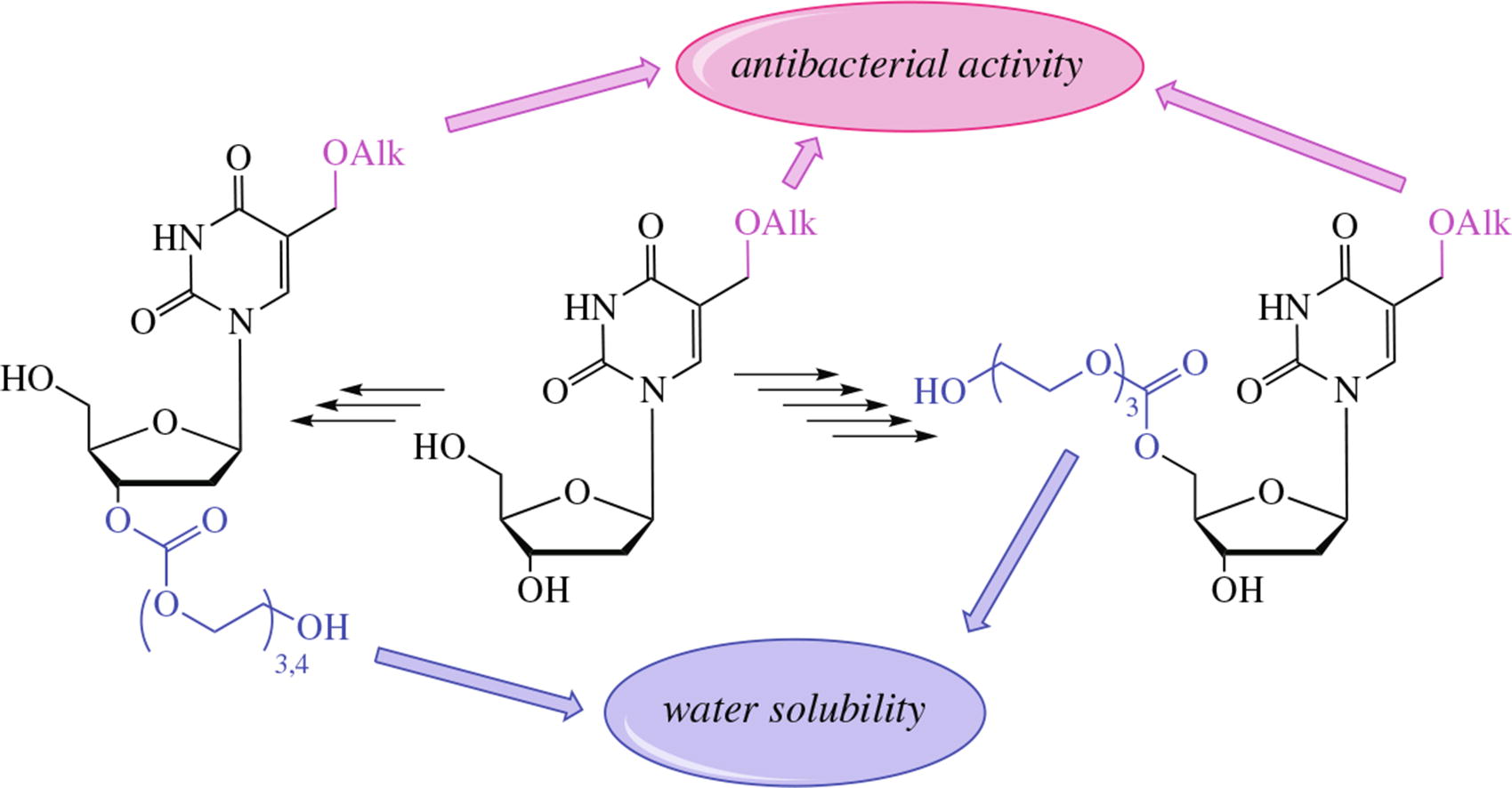
In order to develop a new generation of antibacterial nucleosides, a representative set of novel 3'- and 5'-tri- or tetraethylene glycol prodrug forms of 5-alkyloxymethyl-2'- deoxyuridines was synthesized. These compounds were at least two orders of magnitude more soluble than the parent nucleosides, possessed significant inhibitory activity against a set of bacteria including resistant strains of Staphylococcus aureus and Mycobacterium smegmatis, and showed low cytotoxicity. The obtained data indicate that glycol carbonates are convenient and prospective for usage in prodrugs of nucleoside derivatives with antibacterial activity.
References
1.

Serpi M., Ferrari V., Pertusati F.
Journal of Medicinal Chemistry,
2016
2.

Johar M., Manning T., Kunimoto D.Y., Kumar R.
Bioorganic and Medicinal Chemistry,
2005
3.

Rai D., Johar M., Manning T., Agrawal B., Kunimoto D.Y., Kumar R.
Journal of Medicinal Chemistry,
2005
4.

Shmalenyuk E.R., Kochetkov S.N., Alexandrova L.A.
Russian Chemical Reviews,
2013
5.

Shmalenyuk E.R., Chernousova L.N., Karpenko I.L., Kochetkov S.N., Smirnova T.G., Andreevskaya S.N., Chizhov A.O., Efremenkova O.V., Alexandrova L.A.
Bioorganic and Medicinal Chemistry,
2013
6.

Shmalenyuk E.R., Karpenko I.L., Chernousova L.N., Chizhov A.O., Smirnova T.G., Andreevskaya S.N., Alexandrova L.A.
Russian Chemical Bulletin,
2014
7.

Sanches B.M., Ferreira E.I.
International Journal of Pharmaceutics,
2019
8.

Jornada D., dos Santos Fernandes G., Chiba D., de Melo T., dos Santos J., Chung M.
Molecules,
2015
9.

Huttunen K.M., Raunio H., Rautio J.
Pharmacological Reviews,
2011
10.

Rautio J., Kumpulainen H., Heimbach T., Oliyai R., Oh D., Järvinen T., Savolainen J.
Nature Reviews Drug Discovery,
2008
11.

Bentham Science Publisher B.S.
Current Medicinal Chemistry,
2006
12.

Stella V.J., Nti-Addae K.W.
Advanced Drug Delivery Reviews,
2007
13.

Mori G., Chiarelli L.R., Riccardi G., Pasca M.R.
Drug Discovery Today,
2017
14.

Negrya S.D., Jasko M.V., Solyev P.N., Karpenko I.L., Efremenkova O.V., Vasilyeva B.F., Sumarukova I.G., Kochetkov S.N., Alexandrova L.A.
Journal of Antibiotics,
2020
15.

Negrya S.D., Jasko M.V., Makarov D.A., Solyev P.N., Karpenko I.L., Shevchenko O.V., Chekhov O.V., Glukhova A.A., Vasilyeva B.F., Efimenko T.A., Sumarukova I.G., Efremenkova O.V., Kochetkov S.N., Alexandrova L.A.
Molecular Biology,
2021
16.

Alexandrova L., Zicari S., Matyugina E., Khandazhinskaya A., Smirnova T., Andreevskaya S., Chernousova L., Vanpouille C., Kochetkov S., Margolis L.
Antiviral Research,
2017
17.

De Clercq E.
Journal of Clinical Virology,
2001
18.

Nikš M., Otto M.
Journal of Immunological Methods,
1990
19.
Matyugina E.S., Valuev-Elliston V.T., Chizhov A.O., Kochetkov S.N., Khandazhinskaya A.L.
Mendeleev Communications,
2016
20.
Gaman M.S., Matyugina E.S., Novikov M.S., Babkov D.A., Solyev P.N., Kochetkov S.N., Khandazhinskaya A.L.
Mendeleev Communications,
2017
21.
Paramonova M.P., Ozerov A.A., Chizhov A.O., Snoeck R., Andrei G., Khandazhinskaya A.L., Novikov M.S.
Mendeleev Communications,
2019