Keywords
addition reaction
aldehydes
arylboronic acids
benzylic alcohols
green solvent
Rhodium catalysis
γ-valerolactone
Abstract
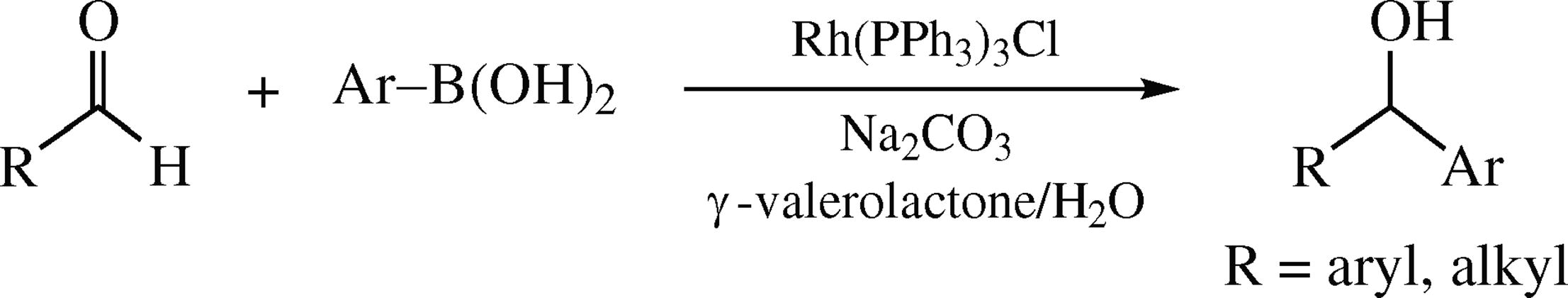
A simple rhodium-catalyzed addition reaction of aldehydes with arylboronic acids in aqueous γ-valerolactone provides the corresponding benzylic alcohols in moderate to good yields. Other organoboron reagents (boronic esters, aryl-trifluoroborates, etc.) also showed good compatibilities, albeit with relatively lower yields.
References
1.
10.1016/j.mencom.2022.05.036_b0005
Schmidt
Chem. Soc. Rev.,
2006
2.

Bolm C., Hildebrand J.P., Muñiz K., Hermanns N.
Angewandte Chemie - International Edition,
2001
3.

Boymond L., Rottländer M., Cahiez G., Knochel P.
Angewandte Chemie - International Edition,
1998
4.

Li C., Meng Y.
Journal of the American Chemical Society,
2000
5.

Wu K., Gau H.
Journal of the American Chemical Society,
2006
6.

Bolm C., Rudolph J.
Journal of the American Chemical Society,
2002
7.

Miyaura N., Suzuki A.
Chemical Reviews,
1995
8.

Sakai M., Ueda M., Miyaura N.
Angewandte Chemie - International Edition,
1998
9.

Ueda M., Miyaura N.
Journal of Organic Chemistry,
2000
10.

Huang R., Shaughnessy K.H.
Chemical Communications,
2005
11.

Pucheault M., Darses S., Genet J.
Chemical Communications,
2005
12.

Moreau C., Hague C., Weller A.S., Frost C.G.
Tetrahedron Letters,
2001
13.

Türkmen H., Çetinkaya B.
Applied Organometallic Chemistry,
2010
14.

Denizaltı S., Türkmen H., Çetinkaya B.
Tetrahedron Letters,
2014
15.

Wang D., Cui S.
Tetrahedron,
2015
16.

Yang J., Chen X., Wang Z.
Tetrahedron Letters,
2015
17.

Nishimura T., Kumamoto H., Nagaosa M., Hayashi T.
Chemical Communications,
2009
18.
![Synthesis of novel macrocyclic planar chiral carbene–Ag complexes derived from [2.2]paracyclophane for Rh-catalyzed asymmetric 1,2-additions of arylboronic acids to aromatic aldehydes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chen J., Yang S., Chen Z., Song C., Ma Y.
Tetrahedron Asymmetry,
2015
19.

Jana R., Tunge J.A.
Journal of Organic Chemistry,
2011
20.

21.

Hu K., Ye P., Zhen Q., Yao X., Xu T., Shao Y., Chen J.
Molecules,
2017
22.

Xing C., Hu Q.
Tetrahedron Letters,
2010
23.

Yamamoto Y., Kurihara K., Miyaura N.
Angewandte Chemie - International Edition,
2009
24.

Zou T., Pi S., Li J.
Organic Letters,
2008
25.

Liao Y., Hu Q.
Journal of Organic Chemistry,
2011
26.

Gandeepan P., Kaplaneris N., Santoro S., Vaccaro L., Ackermann L.
ACS Sustainable Chemistry and Engineering,
2019