Keywords
1,3-selenazolines
acetonitrile
alkenes
iodine assistance
organoselenium compounds
selenium
solvent interception
Abstract
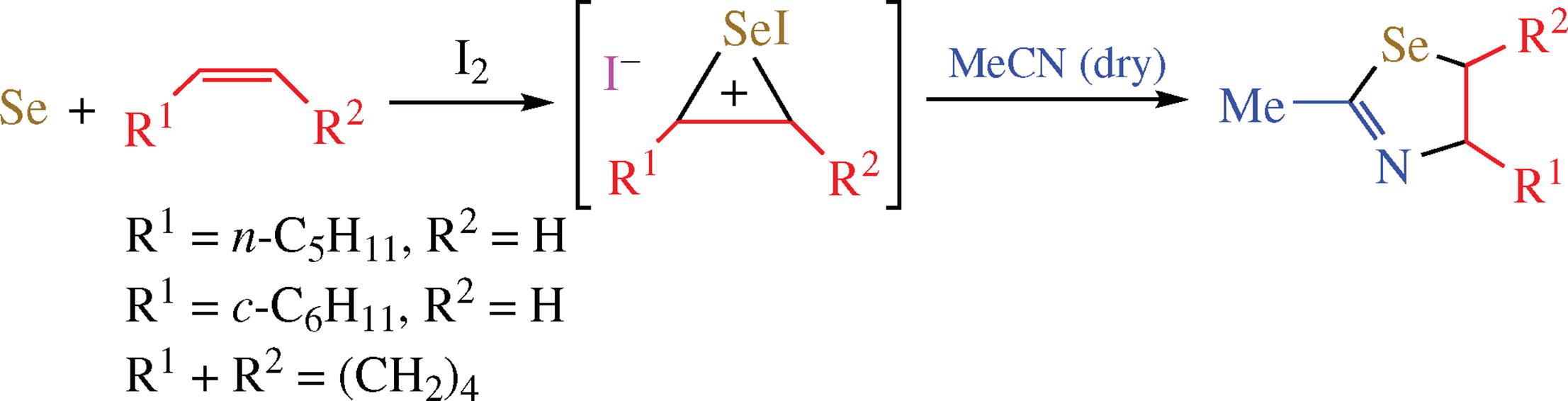
A new method for assembling 1,3-selenazolines by the iodine- mediated reaction of the simplest building blocks such as elemental selenium, alkenes and acetonitrile has been discovered. A proposed mechanism includes the addition of the intermediate selenium iodides to alkene with subsequent solvent interception by the formed seleniranium ion.
References
1.

Mugesh G., du Mont W., Sies H.
Chemical Reviews,
2001
2.

Qiao J., Liu Y., Du Y.
Tetrahedron,
2018
3.

Langer P., Geisler K., Pfeiffer W., Künzler A., Below H., Bulka E.
Synthesis,
2004
4.

Koketsu M., Ando H., Sasaki T., Ishihara H.
Journal of Heterocyclic Chemistry,
2007
5.

Shibahara F., Fukunaga T., Kubota S., Yoshida A., Murai T.
Organic Letters,
2018
6.
Kurkutov E.O., Borodina T.N.
Mendeleev Communications,
2020
7.

Gopal M., Milne J.
Inorganic Chemistry,
1992
8.

Klapoetke T., Passmore J.
Accounts of Chemical Research,
1989
9.

Toma H.E., Takasugi M.S.
Journal of Solution Chemistry,
1983
10.

Minegishi S., Kobayashi S., Mayr H.
Journal of the American Chemical Society,
2004
11.

Astakhova V.V., Moskalik M.Y., Shainyan B.A.
Organic and Biomolecular Chemistry,
2019
12.

Moskalik M.Y., Shainyan B.A., Ushakov I.A., Sterkhova I.V., Astakhova V.V.
Tetrahedron,
2020