Keywords
2,7-naphthyridines
acetylacetone
heterocyclization
hydrazine
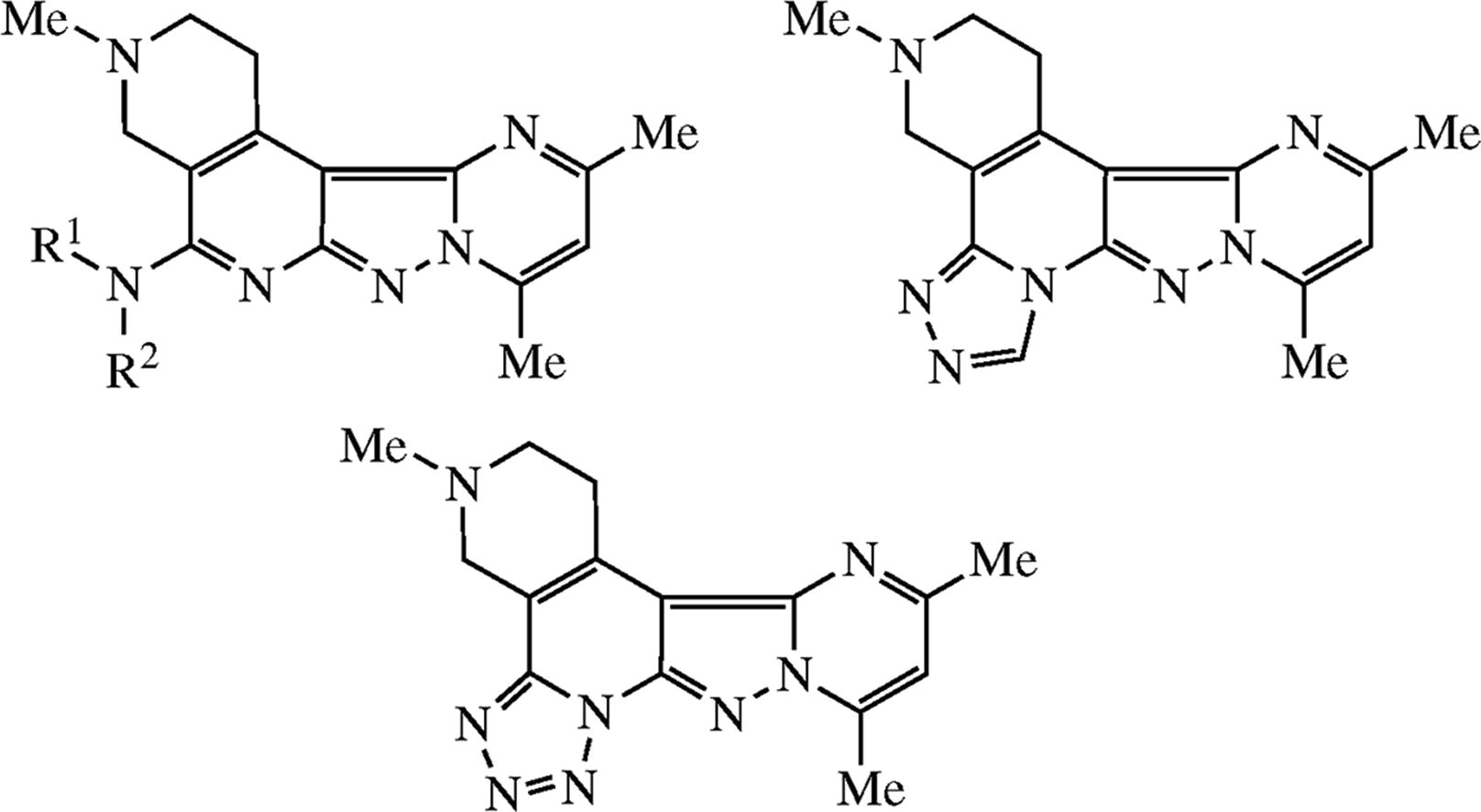
pyrazolo[3,4-c][1,2,4]triazolo[3,4-a]-2,7- naphthyridines
pyrazolo[3,4-c]tetrazolo[5,1-a]-2,7-naphthyridine
pyrimido[1' 2':1,5]pyrazolo[3,4-c]-2,7-naphthyridine
Abstract
Efficient syntheses of new heterocyclic systems comprising pyrimido[1',2':1,5]pyrazolo[3,4-c]-2,7-naphthyridine, pyrazolo[3,4-c][1,2,4]triazolo[3,4-a]-2,7-naphthyridine and pyrazolo[3,4-c]tetrazolo[5,1-a]-2,7-naphthyridine cores were performed in two simple steps. In the first step, pyrazole ring was fused at the 2-chloro-3-cyanopyridine moiety by treatment with hydrazine. In the second step, pyrimidine part was fused at the thus formed 3-aminopyrazole moiety by heterocyclization with acetylacetone.
References
1.
(a) E. G. Brown, Ring Nitrogen and Key Biomolecules: The Biochemistry of N-Heterocycles, Springer, Dordrecht, 1998; (b) S. Hauptmann and A. Speicher, The Chemistry of Heterocycles: Structure, Reactions, Syntheses, and Applications, Wiley-VCH, Weinheim, 2012.
2.
![Synthesis and Anticonvulsant Activity of Pyrazolo[3,4-b]pyrano(thiopyrano)[4,3-d]pyridine and Pyrazolo[3,4-c]isoquinoline Derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Paronikyan E.G., Sirakanyan S.N., Noravyan A.S., Paronikyan R.G., Dzhagatspanyan I.A.
Pharmaceutical Chemistry Journal,
2001
3.
![Synthesis of some novel pyrazolo[3,4-b]pyridine and pyrazolo[3,4-d]pyrimidine derivatives bearing 5,6-diphenyl-1,2,4-triazine moiety as potential antimicrobial agents](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
El-Sayed Ali T.
European Journal of Medicinal Chemistry,
2009
4.
![BAY 41-2272 [5-Cyclopropyl-2-[1-(2-fluoro-benzyl)-1H-pyrazolo[3,4-b]pyridine-3-yl]pyrimidin-4-ylamine]-Induced Dilation in Ovine Pulmonary Artery: Role of Sodium Pump](/storage/images/resized/s6JtNKPXrzklKtbQZ4HrtzuJT0P7eHPX9U8x0nss_small_thumb.webp)
Bawankule D.U., Sathishkumar K., Sardar K.K., Chanda D., Krishna A.V., Prakash V.R., Mishra S.K.
Journal of Pharmacology and Experimental Therapeutics,
2005
5.
![Synthesis and antiviral activity of new 4-(phenylamino)/4-[(methylpyridin-2-yl)amino]-1-phenyl-1H-pyrazolo[3,4-b]pyridine-4-carboxylic acids derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bernardino A.M., de Azevedo A.R., Pinheiro L.C., Borges J.C., Carvalho V.L., Miranda M.D., de Meneses M.D., Nascimento M., Ferreira D., Rebello M.A., Silva V.A., de Frugulhetti I.C.
Medicinal Chemistry Research,
2007
6.

Antileishmanial Pyrazolopyridine Derivatives: Synthesis and Structure−Activity Relationship Analysis
de Mello H., Echevarria A., Bernardino A.M., Canto-Cavalheiro M., Leon L.L.
Journal of Medicinal Chemistry,
2004
7.
![Discovery of Novel Pyrazolo[3,4-b] Pyridine Derivatives with Dual Activities of Vascular Remodeling Inhibition and Vasodilation for the Treatment of Pulmonary Arterial Hypertension](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Hu L., Li L., Chang Q., Fu S., Qin J., Chen Z., Li X., Liu Q., Hu G., Li Q.
Journal of Medicinal Chemistry,
2020
8.

van Linden O.P., Farenc C., Zoutman W.H., Hameetman L., Wijtmans M., Leurs R., Tensen C.P., Siegal G., de Esch I.J.
European Journal of Medicinal Chemistry,
2012
9.
![Synthesis of 3-(1H-benzimidazol-2-yl)-5-isoquinolin-4-ylpyrazolo[1,2-b]pyridine, a potent cyclin dependent kinase 1 (CDK1) inhibitor](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Huang S., Lin R., Yu Y., Lu Y., Connolly P.J., Chiu G., Li S., Emanuel S.L., Middleton S.A.
Bioorganic and Medicinal Chemistry Letters,
2007
10.
![Identification of 1H-pyrazolo[3,4-b]pyridine derivatives as potent ALK-L1196M inhibitors](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Nam Y., Hwang D., Kim N., Seo H., Selim K.B., Sim T.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2019
11.
![Synthesis of novel 1H-Pyrazolo[3,4-b]pyridine derivatives as DYRK 1A/1B inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Park A., Hwang J., Lee J., Heo E.J., Na Y., Kang S., Jeong K., Kim K.Y., Shin S.J., Lee H.
Bioorganic and Medicinal Chemistry Letters,
2021
12.

Ripperger H.
Phytochemistry,
1978
13.
(b) K. P. Tiwari, P. K. Minocha and M. Masood, Pol. J. Chem., 1980, 54, 857
14.

15.

Reimann E., Renz H.
Monatshefte fur Chemie,
1994
16.
(e) E. G. Paronikyan, G. V. Mirzoyan, A. S. Noravyan, E. M. Arzanunts, R. S. Sukasyan, I. S. Sarkisyan, I. M. Nazaryan and I. A. Dzhagatspanyan, Pharm. Chem. J., 1997, 31, 540 [Khim.-Farm. Zh., 1997, 31 (10), 34]
17.
![Synthesis and Anticonvulsant Activity of isothiazolo[5,4-b]pyrano(thiopyrano)[4,3-d]pyridine and Isothiazolo[4,5-b]-2,7-naphthyridine Derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Paronikyan E.G., Noravyan A.S., Dzhagatspanyan I.A., Nazaryan I.M., Paronikyan R.G.
Pharmaceutical Chemistry Journal,
2002
18.
(a) E. G. Paronikyan, S. N. Sirakanyan, A. S. Noravyan, T. O. Asatryan, K. J. Markaryan and R. A. Aleksanyan, Pharm. Chem. J., 1996, 30, 365 [Khim.-Farm. Zh., 1996, 30 (6), 13]
19.
![Synthesis and Neurotropic Activity of Triazolo[3,4-a]-and Triazolo[5,1-a][2,7]Naphthyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sirakanyan S.N., Tonoyants N.A., Noravyan A.S., Dzhagatspanyan I.A., Nazaryan I.M., Akopyan A.G., Paronikyan R.G., Minasyan N.S.
Pharmaceutical Chemistry Journal,
2014
20.
![Synthesis and Neurotropic Activity of New 7-Cyclohexyl-6,7,8,9-Tetrahydro-3H-Pyrazolo[3,4-c]-2,7-Naphthyridine-1,5-Diamines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sirakanyan S.N., Hakobyan E.K., Nikoghosyan A.G., Paronikyan R.G., Dzhagatspanyan I.A., Nazaryan I.M., Akopyan A.G., Hovakimyan A.A.
Pharmaceutical Chemistry Journal,
2018
21.

Sirakanyan S.N., Kartsev V.G., Spinelli D., Geronikaki A., Noravyan A.S., Hovakimyan A.A., Panosyan H.A., Ayvazyan A.G., Tamazyan R.A.
Tetrahedron,
2014
22.
![New heterocyclic systems based on 1-hydrazino-5,6,7,8-tetrahydro[2,7]naphthyridine: 7,8,9,10-tetra-hydro[1,2,4]triazolo[3,4-a]- and 7,8,9,10-tetra-hydro[1,2,4]triazolo[5,1-a][2,7]naphthyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sirakanyan S.N., Avetisyan N.G., Noravyan A.S.
Chemistry of Heterocyclic Compounds,
2012
23.

Sirakanyan S.N., Spinelli D., Geronikaki A., Hovakimyan A.A., Noravyan A.S.
Tetrahedron,
2014