Keywords
N-hetaryl quinone imines
DFT calculations
imidazo[2,1-b][1,3,4]thiadiazoles
imidazo[2,1-b]thiazoles
recyclization
Abstract
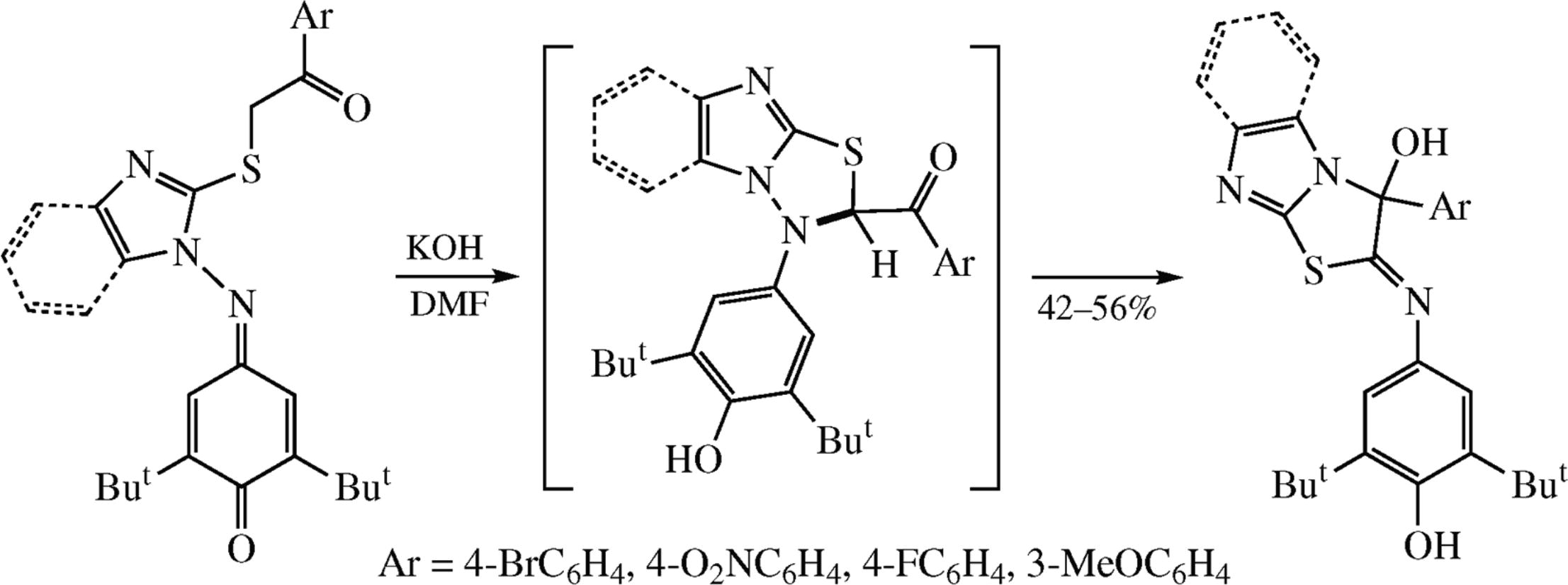
2,6-Di-tert-butyl-4-{[2-(2-aryl-2-oxoethylthio)-1H-imidazol-1-yl]imino}cyclohexa-2,5-dien-1-ones under the action of bases give products of the 2,3-dihydroimidazo[2,1-b]thiazol- 3-ol series by subsequent recyclization reaction of the intermediate imidazo[2,1-b][1,3,4]thiadiazoles. The structure of imidazo[2,1-b]thiazol-3-ol is supported by the X-ray diffraction. The features of the cyclization processes of quinone imine derivatives were stimulated by DFT calculations using the wB97XD/6-311++G** method.
References
1.
![Synthesis and Anticancer Activity of Thiadiazole Containing Thiourea, Benzothiazole and Imidazo[2,1-b][1,3,4]thiadiazole Scaffolds](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Avvaru S.P., Noolvi M.N., More U.A., Chakraborty S., Dash A., Aminabhavi T.M., Narayan K.P., Sutariya V.
Medicinal Chemistry,
2020
2.

Kamal A., Kashi Reddy M., Viswanath A.
Expert Opinion on Drug Discovery,
2013
3.
![Synthesis and diuretic activity of imidazo[2,1-b]thiazole acetohydrazones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Andreani A., Rambaldi M., Mascellani G., Rugarli P.
European Journal of Medicinal Chemistry,
1987
4.
(a) A. K. Gadad, M. N. Maleshappa and P. V. Karpoormath, Bioorg. Med. Chem., 2004, 12, 5651
5.
(b) M. A. Syed, Y. R. P. Reddy and K. B. Chandrasekhar, J. Appl. Pharm. Sci., 2018, 8, 21.
6.
![Synthesis and anti-inflammatory evaluation of methylene bridged benzofuranyl imidazo[2,1-b][1,3,4]thiadiazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Jadhav V.B., Kulkarni M.V., Rasal V.P., Biradar S.S., Vinay M.D.
European Journal of Medicinal Chemistry,
2008
7.
(a) Y. Sun, B.-Q. Fu and M. W. Ding, Phosphorus, Sulfur Silicon Relat. Elem., 2006, 181, 1437
8.
![Novel imidazo[2,1-b]-1,3,4-thiadiazoles as promising antifungal agents against clinical isolate of Cryptococcus neoformans](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Alwan W.S., Karpoormath R., Palkar M.B., Patel H.M., Rane R.A., Shaikh M.S., Kajee A., Mlisana K.P.
European Journal of Medicinal Chemistry,
2015
9.
(a) K. Manjunatha, B. Poojary, V. Kumar, P. L. Lobo, J. Fernandes and C. Chandrashekhar, Pharma Chem., 2015, 7, 207
10.

Ramamurthy S., Jayachandran E.
Asian Journal of Chemistry,
2017
11.
(c) A. K. Gadad, C. S. Mahajanshetti, S. Nimbalkar and A. Raichurkar, Eur. J. Med. Chem., 2000, 35, 853.
12.
![Biological activities of imidazo[2,1-b][1,3,4]thiadiazole derivatives: A review](/storage/images/resized/9z7b0TQJNgUxZHYXLrTmbnjJn9y5iq56Wxtqb1Lv_small_thumb.webp)
Bhongade B.A., Talath S., Gadad R.A., Gadad A.K.
Journal of Saudi Chemical Society,
2016
13.
![Chemistry of imidazo[2,1-b][1,3,4]thiadiazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Khazi I.A., Gadad A.K., Lamani R.S., Bhongade B.A.
Tetrahedron,
2011
14.

Fascio M.L., Errea M.I., D'Accorso N.B.
European Journal of Medicinal Chemistry,
2015
15.

Saliyeva L.N., Diachenko I.V., Vas’kevich R.I., Slyvka N.Y., Vovk M.V.
Chemistry of Heterocyclic Compounds,
2020
16.
(a) A. A. Kolodina, N. I. Gaponenko and A. V. Lesin, Russ. Chem. Bull., 2008, 57, 1273 (Izv. Akad. Nauk, Ser. Khim., 2008, 1249)
17.
Kolodina A.A., Lesin A.V., Nelyubina Y.V.
Mendeleev Communications,
2008
18.
(c) A. A. Kolodina, A. A. Tsaturyan, M. S. Galkina, I. G. Borodkina, E. V. Vetrova, O. P. Demidov, A. G. Berezhnaya and A. V. Metelitsa, ChemisrySelect, 2020, 5, 3586.
19.
(a) A. A. Kolodina, N. I. Gaponenko and A. V. Lesin, Chem. Heterocycl. Compd., 2007, 43, 1202 (Khim. Geterotsikl. Soedin., 2007, 1415)
20.
(b) N. I. Gaponenko, A. A. Kolodina, A. V. Lesin, S. V. Kurbatov, Z. A. Starikova and Yu. V. Nelyubina, Russ. Chem. Bull., 2010, 59, 838 (Izv. Akad. Nauk, Ser. Khim., 2010, 821)
21.

Gaponenko N.I., Kolodina A.A., Lesin A.V., Kurbatov S.V.
Russian Chemical Bulletin,
2012
22.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
23.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
24.
Guseinov F.I., Pistsov M.F., Malinnikov V.M., Lavrova O.M., Movsumzade E.M., Kustov L.M.
Mendeleev Communications,
2020
25.
A. A. Kolodina and A. V. Lesin, Russ. J. Org. Chem., 2009, 45, 139 (Zh. Org. Khim., 2009, 45, 142).