Keywords
2H-chromenes
5-diones
chromeno[2,3-c]pyrroles
intramolecular O-H insertion
pyrrolidine-2
rhodium(u) complexes
α-diazo carbonyl compounds
Abstract
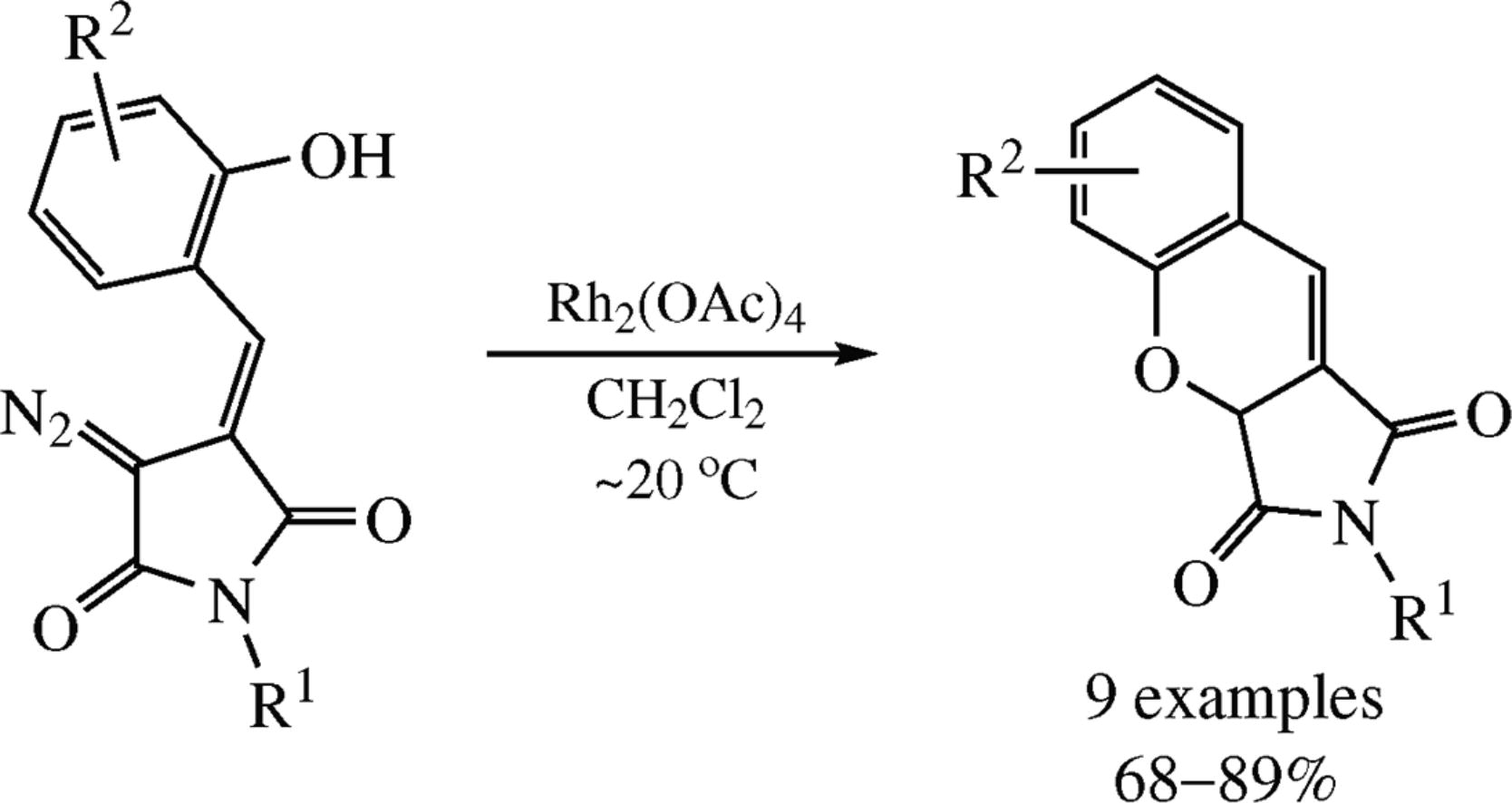
Rhodium(ii) carbenes generated from (E)-3-diazo-4-(2-hydroxybenzylidene)-1-phenylpyrrolidine-2,5-dione under- went a facile transformation into novel succinimide-fused 2H-chromenes, chromeno[2,3-c]pyrrole-1,3(2H,3aH)-diones. The process presumably involves an intramolecular O-H insertion reaction.
References
1.

Raj V., Lee J.
Frontiers in Chemistry,
2020
2.

Cheng J., Ishikawa A., Ono Y., Arrhenius T., Nadzan A.
Bioorganic and Medicinal Chemistry Letters,
2003
3.

Cheenpracha S., Karalai C., Ponglimanont C., Kanjana-Opas A.
Journal of Natural Products,
2009
4.
B. Chen, R. A. Fairhurst, A. Floersheimer, P. Furet, S. Jiang, W. Lu, T. H. Marsilje and A. Vaupel, Patent WO 201164211A1, 2011.
5.

Gómez-Garibay F., Calderón J.S., De La O Arciniega M., Céspedes C.L., Téllez-Valdés O., Taboada J.
Phytochemistry,
1999
6.

Yasunaga T., Kimura T., Naito R., Kontani T., Wanibuchi F., Yamashita H., Nomura T., Tsukamoto S., Yamaguchi T., Mase T.
Journal of Medicinal Chemistry,
1998
7.

Gauthier S., Cloutier J., Dory Y.L., Favre A., Mailhot J., Ouellet C., Schwerdtfeger A., Mérand Y., Martel C., Simard J., Labrie F.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2005
8.

KATIYAR S., ELMETS C., KATIYAR S.
Journal of Nutritional Biochemistry,
2007
9.

Li W., Liang H., Zhang M., Zhang R., Deng Y., Wei Z., Zhang Y., Tang X.
Molecules,
2012
10.

Sawadjoon S., Kittakoop P., Kirtikara K., Vichai V., Tanticharoen M., Thebtaranonth Y.
Journal of Organic Chemistry,
2002
11.

Majumdar N., Paul N.D., Mandal S., de Bruin B., Wulff W.D.
ACS Catalysis,
2015
12.
Chemistry of Heterocyclic Compounds: Chromenes, Chromanones, and Chromones, ed. G. P. Ellis, Wiley, New York, 2009, vol. 31.
13.

Bugaenko D.I., Karchava A.V., Yunusova Z.A., Yurovskaya M.A.
Chemistry of Heterocyclic Compounds,
2019
14.
Mekky A.E., Sanad S.M.
Mendeleev Communications,
2020
15.
![Multicomponent design of chromeno[2,3-b]pyridine systems](/storage/images/resized/9Mus3KG1Tkd7Bwaurt8H3RwWh0CxRlGoO6ng9UK1_small_thumb.webp)
Elinson M.N., Ryzhkova Y.E., Ryzhkov F.V.
Russian Chemical Reviews,
2021
16.

Welsch M.E., Snyder S.A., Stockwell B.R.
Current Opinion in Chemical Biology,
2010
17.
![Spirocyclizations Involving Oxonium Ylides Derived from Cyclic α-Diazocarbonyl Compounds: An Entry into 6-Oxa-2-azaspiro[4.5]decane Scaffold.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Dar’in D., Kantin G., Bakulina O., Inyutina A., Chupakhin E., Krasavin M.
Journal of Organic Chemistry,
2020
18.
Chupakhin E.G., Kantin G.P., Dar’in D.V., Krasavin M.
Mendeleev Communications,
2021
19.

Krasavin M., Dar’in D., Chupakhin E., Gecht M., Ivanov A., Kantin G.
Synthesis,
2020
20.
![Facile entry into the 1H-pyrrolo[3,4-b]indolizine-1,3(2H)-dione scaffold via intramolecular Rh(II) carbene trapping](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chupakhin E., Bakulina O., Dar'in D., Krasavin M.
Tetrahedron Letters,
2021
21.

Dar’in D., Kantin G., Krasavin M.
Chemical Communications,
2019