Keywords
3,5-dinitrosalicylic acid
molecular structure
organoantimony compounds
pentaphenylantimony
photocatalytic properties
X-ray analysis
Abstract
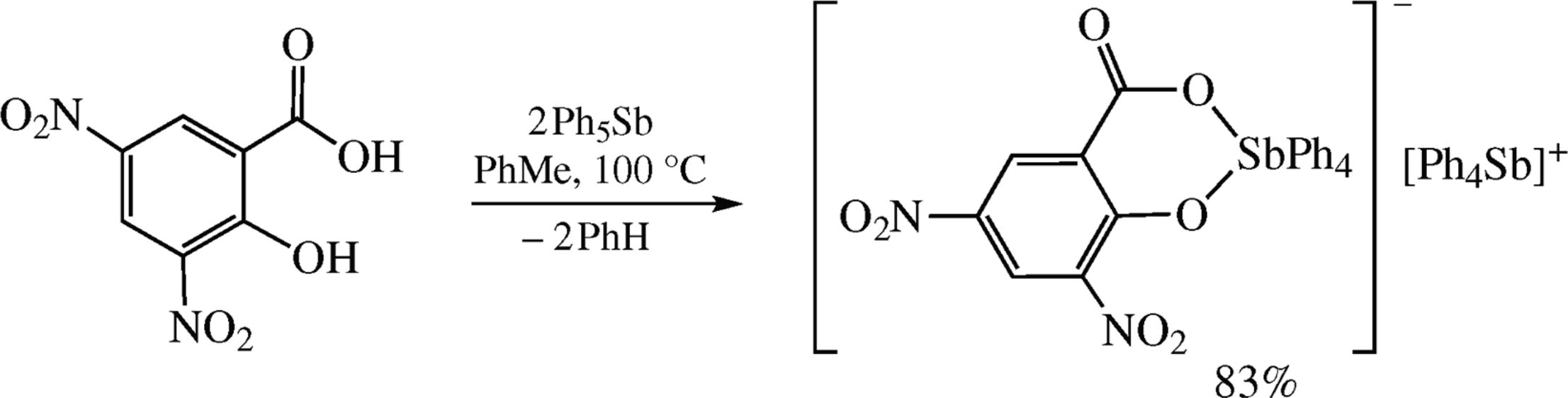
Reaction between pentaphenylantimony and 3,5-dinitro-salicylic acid involves both hydroxy and carboxy groups to form the complex containing an anion with the six-membered metallocycle and Ph4Sb+ counter-cation. The molecular structure of the reaction product has been established by X-ray analysis. The photocatalytic properties have been studied by the methylene blue photodegradation
References
1.

Gushchin A.V., Maleeva A.I., Kipelkin E.V., Tumanyan A.S., Andreev P.V., Ovsetsina T.I., Somov N.V.
Russian Journal of General Chemistry,
2021
2.

Yang M., Hirai M., Gabbaï F.P.
Dalton Transactions,
2019
3.

Zhang X., Cui L., Zhang X., Jin F., Fan Y.
Journal of Molecular Structure,
2017
4.

Yang M., Gabbaï F.P.
Inorganic Chemistry,
2017
5.

Ali M.I., Rauf M.K., Badshah A., Kumar I., Forsyth C.M., Junk P.C., Kedzierski L., Andrews P.C.
Dalton Transactions,
2013
6.
10.1016/j.mencom.2022.05.028_b0030
Mushtaq
Appl. Organomet. Chem.,
2016
7.

Duffin R.N., Blair V.L., Kedzierski L., Andrews P.C.
Dalton Transactions,
2018
8.

Polychronis N.M., Banti C.N., Raptopoulou C.P., Psycharis V., Kourkoumelis N., Hadjikakou S.K.
Inorganica Chimica Acta,
2019
9.

Geng H., Hong M., Yang Y., Li D., Li X., Liu F., Niu M.
Journal of Coordination Chemistry,
2015
10.

Islam A., Rodrigues B.L., Marzano I.M., Perreira-Maia E.C., Dittz D., Paz Lopes M.T., Ishfaq M., Frézard F., Demicheli C.
European Journal of Medicinal Chemistry,
2016
11.

Hong M., Yin H., Li W., You X.
Inorganic Chemistry Communication,
2011
12.

Jiang J., Yin H., Wang F., Han Z., Wang F., Cheng S., Hong M.
Dalton Transactions,
2013
13.
Sharutin V.V., Sharutina O.K., Gubanova Y.O., Fominykh A.S., Eltsov O.S.
Mendeleev Communications,
2020
14.

Quan L., Yin H., Cui J., Hong M., Cui L., Yang M., Wang D.
Journal of Organometallic Chemistry,
2009
15.
Sharutin V.V., Sharutina O.K., Gubanova Y.O., Bregadze V.I., Glazun S.A., Andreev P.V.
Mendeleev Communications,
2018
16.
10.1016/j.mencom.2022.05.028_b0080
Sharutin
Zh. Obshch. Khim.,
1997
17.

Quan L., Yin H., Cui J., Hong M., Wang D.
Journal of Organometallic Chemistry,
2009
18.
10.1016/j.mencom.2022.05.028_b0090
SMART and SAINT-plus
Versions 5.0, Data Collection and Processing Software for the SMART System,
1998
19.
10.1016/j.mencom.2022.05.028_b0095
SHELXTL/PC
Versions 5.10, An Integrated System for Solving, Refining and Displaying Crystal Structures from Diffraction Data,
1998
20.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009