Keywords
[1,2,5]thiadiazolo[3,4-d]pyridazines
aromatic nucleophilic substitution
donor–acceptor–donor structure
indoles
luminescence
NIR-OLEDs
synthesis
Abstract
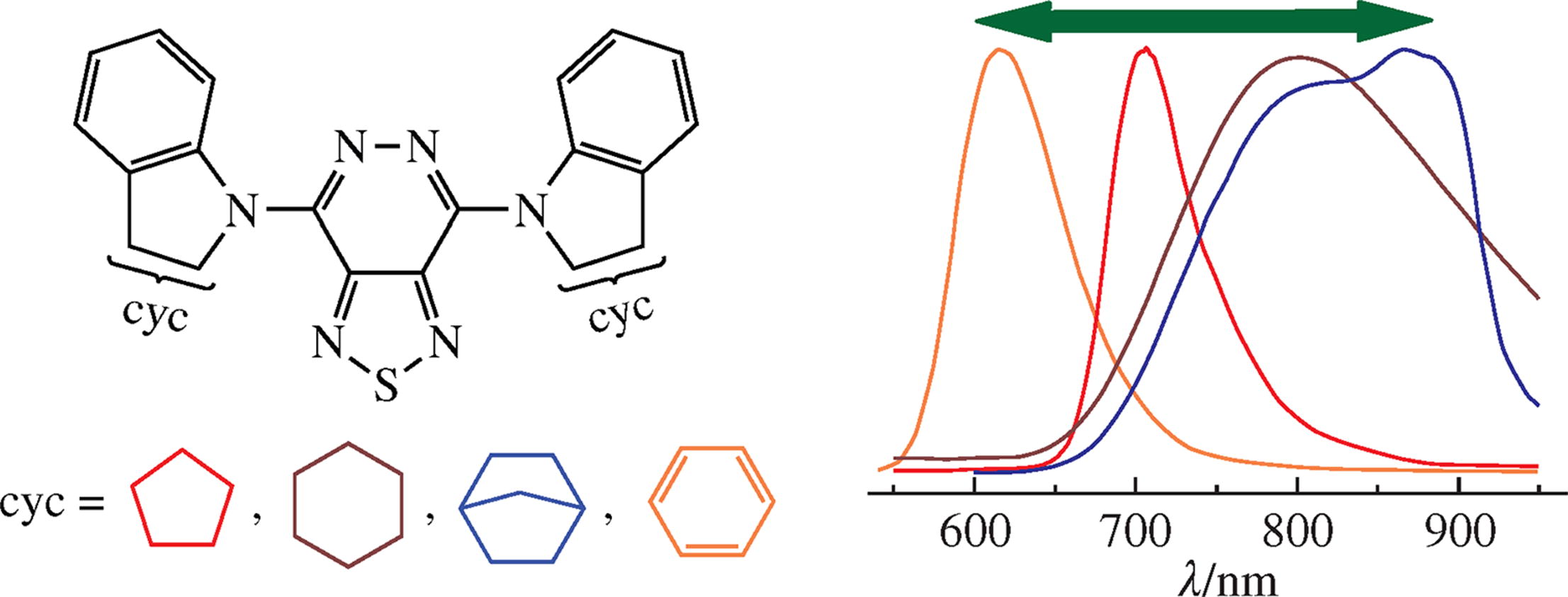
Three [1,2,5]thiadiazolo[3,4-d]pyridazines containing 4,7-positioned indole-type substituents were synthesized from the corresponding 4,7-dibromo precursor employing the SNAr aromatic nucleophilic substitution. Photophysical properties and DFT calculations showed that the D–A–D dyes based on [1,2,5]thiadiazolo[3,4-d]pyridazine core exhibited fluorescence in the near infrared region of the spectrum, which makes them promising for use as an active emitting layer in NIR-OLEDs as well as for other possible applications as an IR luminophore.
References
1.

Knyazeva E.A., Rakitin O.A.
Russian Chemical Reviews,
2016
2.
![Dye-sensitized solar cells: Investigation of D-A-Π-A organic sensitizers based on [1,2,5]selenadiazolo[3,4-c]pyridine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Knyazeva E.A., Wu W., Chmovzh T.N., Robertson N., Woollins J.D., Rakitin O.A.
Solar Energy,
2017
3.
(c) B. A. D. Neto, A. A. M. Lapis, E. N. da Silva, Jr., and J. Dupont, Eur. J. Org. Chem., 2013, 228
4.

Yahya M., Bouziani A., Ocak C., Seferoğlu Z., Sillanpää M.
Dyes and Pigments,
2021
5.

Chaurasia S., Lin J.T.
Chemical Record,
2016
6.
(a) A. V. Akkuratov and P. A. Troshin, Polym. Sci., Ser. B, 2014, 56, 414 (Vysokomol. Soedin., Ser. B, 2014, 56, 371); (b) G. V. Bulavko and A. A. Ishchenko, Russ. Chem. Rev., 2014, 83, 575; (c) T. P. Le, B. H. Smith, Y. Lee, J. H. Litofsky, M. P. Aplan, B. Kuei, C. Zhu, C. Wang, A. Hexemer and E. D. Gomez, Macromolecules, 2020, 53, 1967; (d) W. Xu, Y. Chang, X. Zhu, Z. Wei, X. Zhang, X. Sun, K. Lu and Z. Wei, Chin. Chem. Lett., 2022, 33, 123; (e) A. Wadsworth, Z. Hamid, J. Kosco, N. Gasparini and I. McCulloch, Adv. Mater., 2020, 32, 2001763.
7.

Zhao R., Min Y., Dou C., Lin B., Ma W., Liu J., Wang L.
ACS Applied Polymer Materials,
2019
8.

Leventis A., Chmovzh T.N., Knyazeva E.A., Han Y., Heeney M., Rakitin O.A., Bronstein H.
Polymer Chemistry,
2020
9.

Tang Z., Wei X., Zhang W., Zhou Y., Wei C., Huang J., Chen Z., Wang L., Yu G.
Polymer,
2019
10.

Korshunov V.M., Chmovzh T.N., Golovanov I.S., Knyazeva E.A., Mikhalchenko L.V., Saifutyarov R.S., Avetisov I.C., Woollins J.D., Taydakov I.V., Rakitin O.A.
Dyes and Pigments,
2021
11.

Biswas S., Pramanik A., Ahmed T., Sahoo S.K., Sarkar P.
Chemical Physics Letters,
2016
12.

Chen P., Zhu H., Kong L., Xu X., Tian Y., Yang J.
Dyes and Pigments,
2020
13.
Gribanov P.S., Lypenko D.A., Dmitriev A.V., Pozin S.I., Topchiy M.A., Asachenko A.F., Loginov D.A., Osipov S.N.
Mendeleev Communications,
2021
14.

Rakitin O.A.
Tetrahedron Letters,
2020
15.

Minotto A., Murto P., Genene Z., Zampetti A., Carnicella G., Mammo W., Andersson M.R., Wang E., Cacialli F.
Advanced Materials,
2018
16.
![New Benzo[1,2-d:4,5-d′]bis([1,2,3]thiadiazole) (iso-BBT)-Based Polymers for Application in Transistors and Solar Cells](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Bianchi L., Zhang X., Chen Z., Chen P., Zhou X., Tang Y., Liu B., Guo X., Facchetti A.
Chemistry of Materials,
2019
17.
![Synthesis of the 4,7-Dibromo Derivative of Highly Electron-Deficient [1,2,5]Thiadiazolo[3,4-d]pyridazine and Its Cross-Coupling Reactions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Chmovzh T.N., Knyazeva E.A., Mikhalchenko L.V., Golovanov I.S., Amelichev S.A., Rakitin O.A.
European Journal of Organic Chemistry,
2018
18.
![Reactions of 4,7-dibromo[1,2,5]thiadiazolo[3,4-d]pyridazine with alcohols](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Chmovzh T.N., Knyazeva E.A., Krukovskaya N.V., Rakitin O.A.
Russian Chemical Bulletin,
2020
19.
![Safe synthesis of 4,7-dibromo[1,2,5]thiadiazolo [3,4-d]pyridazine and its SNAr reactions](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Chmovzh T., Knyazeva E., Lyssenko K., Popov V., Rakitin O.
Molecules,
2018