Keywords
3,7-diazabicyclo[3.3.1]nonane
allosteric modulators
AMPA receptors
bispidines
heterocyclization
Abstract
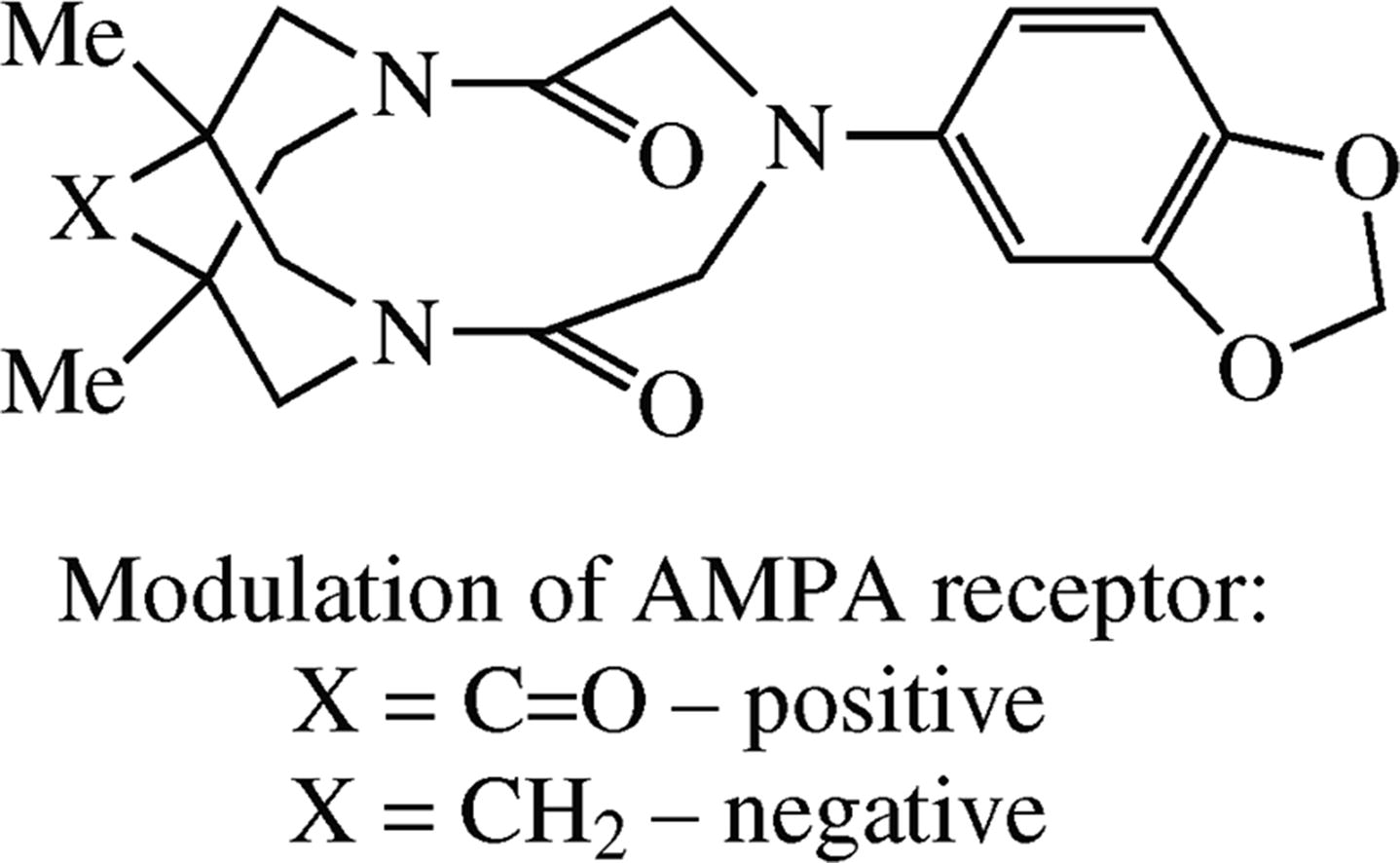
Two new potent AMPA receptor allosteric modulators, 6-(1,3-benzodioxol-5-yl)-1,11-dimethyl-3,6,9-triazatri-cyclo[7.3.1.13,11]tetradecane-4,8,12-trione and -4,8-dione were synthesized from 1,3-benzodioxol-5-amine and the corresponding 3,7-dichloroacetyl-3,7-diazabicyclo[3.3.1]-nonanes. In a wide concentration range (10−12–10−7 m), the 12-oxo derivative acts as a positive modulator causing the potentiation of the kainate-induced AMPA receptor currents with maximum potentiation at 1 nm (62%) while its analogue without a ketone group has significant (up to 40%) negative modulator effect. Their tentative mechanisms of action were analyzed by means of molecular modelling
References
1.

Traynelis S.F., Wollmuth L.P., McBain C.J., Menniti F.S., Vance K.M., Ogden K.K., Hansen K.B., Yuan H., Myers S.J., Dingledine R.
Pharmacological Reviews,
2010
2.

Robbins T.W., Murphy E.R.
Trends in Pharmacological Sciences,
2006
3.

Lynch G.
Current Opinion in Pharmacology,
2004
4.

Lee K., Goodman L., Fourie C., Schenk S., Leitch B., Montgomery J.M.
Advances in Protein Chemistry and Structural Biology,
2016
5.

Radin D.P., Zhong S., Purcell R., Lippa A.
Biomedicine and Pharmacotherapy,
2016
6.

Mendez-David I., Guilloux J., Papp M., Tritschler L., Mocaer E., Gardier A.M., Bretin S., David D.J.
Frontiers in Pharmacology,
2017
7.

Calabrese F., Savino E., Mocaer E., Bretin S., Racagni G., Riva M.A.
Pharmacological Research,
2017
8.

Radin D.P., Johnson S., Purcell R., Lippa A.S.
Biomedicine and Pharmacotherapy,
2018
9.
10.1016/j.mencom.2022.05.023_b0045
Mozafari
Iran J. Basic Med. Sci.,
2018
10.

Gordillo-Salas M., Pascual-Antón R., Ren J., Greer J., Adell A.
Molecular Neurobiology,
2020
11.

Lauterborn J.C., Palmer L.C., Jia Y., Pham D.T., Hou B., Wang W., Trieu B.H., Cox C.D., Kantorovich S., Gall C.M., Lynch G.
Journal of Neuroscience,
2016
12.

Brogi S., Campiani G., Brindisi M., Butini S.
ACS Medicinal Chemistry Letters,
2019
13.

Partin K.M.
Current Opinion in Pharmacology,
2015
14.

Pirotte B., Francotte P., Goffin E., de Tullio P.
Expert Opinion on Therapeutic Patents,
2013
15.

Ward S.E., Pennicott L.E., Beswick P.
Future Medicinal Chemistry,
2015
16.

Reuillon T., E. Ward S., Beswick P.
Current Topics in Medicinal Chemistry,
2016
17.

Hanada T.
Expert Opinion on Drug Discovery,
2014
18.

Potschka H., Trinka E.
Epilepsia,
2018
19.

Stenum-Berg C., Musgaard M., Chavez-Abiega S., Thisted C.L., Barrella L., Biggin P.C., Kristensen A.S.
Molecular Pharmacology,
2019
20.

Narangoda C., Sakipov S.N., Kurnikova M.G.
ACS Chemical Neuroscience,
2019
21.

Karlov D.S., Lavrov M.I., Palyulin V.A., Zefirov N.S.
Journal of Biomolecular Structure and Dynamics,
2017
22.

Guseynov A.D., Pisarev S.A., Shulga D.A., Palyulin V.A., Fedorov M.V., Karlov D.S.
Journal of Molecular Modeling,
2019
23.

Karlov D.S., Lavrov M.I., Palyulin V.A., Zefirov N.S.
Russian Chemical Bulletin,
2016
24.
Radchenko E.V., Karlov D.S., Lavrov M.I., Palyulin V.A.
Mendeleev Communications,
2017
25.

Lavrov M.I., Grigor’ev V.V., Bachurin S.O., Palyulin V.A., Zefirov N.S.
Doklady Biochemistry and Biophysics,
2015
26.
Lavrov M.I., Veremeeva P.N., Karlov D.S., Zamoyski V.L., Grigoriev V.V., Palyulin V.A.
Mendeleev Communications,
2019
27.
Lavrov M.I., Karlov D.S., Palyulin V.A., Grigoriev V.V., Zamoyski V.L., Brkich G.E., Pyatigorskaya N.V., Zapolskiy M.E.
Mendeleev Communications,
2018
28.
![Novel Positive Allosteric Modulators of AMPA Receptors Based on 3,7-Diazabicyclo[3.3.1]nonane Scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Lavrov M.I., Karlov D.S., Voronina T.A., Grigoriev V.V., Ustyugov A.A., Bachurin S.O., Palyulin V.A.
Molecular Neurobiology,
2019
29.
Lavrov M.I., Stroganov O.V., Zamoyski V.L., Grigoriev V.V., Zapolskiy M.E., Sysolyatin S.V., Malykhin V.V., Surmachev V.N., Palyulin V.A.
Mendeleev Communications,
2020
30.
Temnyakova N.S., Vasilenko D.A., Lavrov M.I., Karlov D.S., Grishin Y.K., Zamoyski V.L., Grigoriev V.V., Averina E.B., Palyulin V.A.
Mendeleev Communications,
2021
31.
Temnyakova N.S., Vasilenko D.A., Lavrov M.I., Karlov D.S., Grishin Y.K., Zamoyski V.L., Grigoriev V.V., Averina E.B., Palyulin V.A.
Mendeleev Communications,
2021
32.
![Synthesis of 3,7-diacyl-1,5-dimethyl-3,7-diazabicyclo[3.3.1]nonane derivatives as promising lipid bilayer modifiers](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Veremeeva P.N., Grishina I.V., Zaborova O.V., Averin A.D., Palyulin V.A.
Tetrahedron,
2019
33.
10.1016/j.mencom.2022.05.023_b0165
Chiavarelli
J. Org. Chem.,
1969
34.

Trott O., Olson A.J.
Journal of Computational Chemistry,
2009
35.

Vanommeslaeghe K., Hatcher E., Acharya C., Kundu S., Zhong S., Shim J., Darian E., Guvench O., Lopes P., Vorobyov I., Mackerell A.D.
Journal of Computational Chemistry,
2009
36.

Huang J., MacKerell A.D.
Journal of Computational Chemistry,
2013
37.

Abraham M.J., Murtola T., Schulz R., Páll S., Smith J.C., Hess B., Lindahl E.
SoftwareX,
2015
38.

Drapier T., Geubelle P., Bouckaert C., Nielsen L., Laulumaa S., Goffin E., Dilly S., Francotte P., Hanson J., Pochet L., Kastrup J.S., Pirotte B.
Journal of Medicinal Chemistry,
2018
39.

Valdés-Tresanco M.S., Valdés-Tresanco M.E., Valiente P.A., Moreno E.
Journal of Chemical Theory and Computation,
2021
40.

Miller B.R., McGee T.D., Swails J.M., Homeyer N., Gohlke H., Roitberg A.E.
Journal of Chemical Theory and Computation,
2012
41.

Yelshanskaya M., Singh A., Sampson J., Narangoda C., Kurnikova M., Sobolevsky A.
Neuron,
2016