Keywords
boron cage
boron neutron capture therapy
cancer
cholesterol
cobalt bis(dicarbollide)
cytotoxicity
human glioblastoma U-87 MG
lipids
liposomes
Abstract
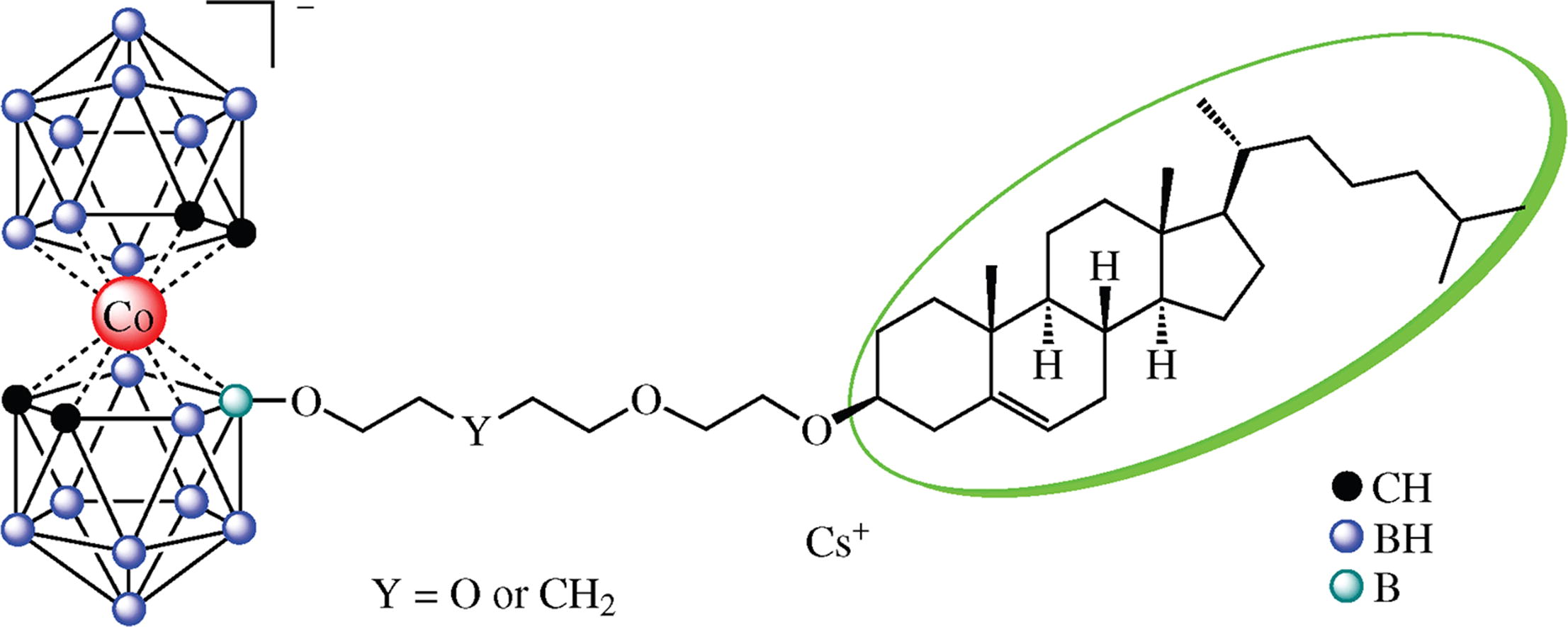
The novel conjugates of cholesterol with cobalt – bis(dicarbollide) were synthesized by the ring-opening reactions of the cyclic oxonium derivatives of [3,3′-Co(C2B9H11)2]– with the OH group of cholesterol 2-hydroxyethyl ether. The compounds obtained were tested for toxicity to glioblastoma U-87 MG cells and human embryo fibroblasts FECH-15 cells
References
1.

Hawthorne M.F., Young D.C., Andrews T.D., Howe D.V., Pilling R.L., Pitts A.D., Reintjes M., Warren L.F., Wegner P.A.
Journal of the American Chemical Society,
1968
2.

Bauduin P., Prevost S., Farràs P., Teixidor F., Diat O., Zemb T.
Angewandte Chemie - International Edition,
2011
3.

Malaspina D.C., Viñas C., Teixidor F., Faraudo J.
Angewandte Chemie - International Edition,
2020
4.

Dash B.P., Satapathy R., Swain B.R., Mahanta C.S., Jena B.B., Hosmane N.S.
Journal of Organometallic Chemistry,
2017
5.
![Metallacarboranes on the Road to Anticancer Therapies: Cellular Uptake, DNA Interaction, and Biological Evaluation of Cobaltabisdicarbollide [COSAN]−](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Fuentes I., García-Mendiola T., Sato S., Pita M., Nakamura H., Lorenzo E., Teixidor F., Marques F., Viñas C.
Chemistry - A European Journal,
2018
6.

Tarrés M., Canetta E., Paul E., Forbes J., Azzouni K., Viñas C., Teixidor F., Harwood A.J.
Scientific Reports,
2015
7.
10.1016/j.mencom.2022.05.021_b0035
Spryshkova
Med. Radiol.,
1981
8.

Verdiá-Báguena C., Alcaraz A., Aguilella V.M., Cioran A.M., Tachikawa S., Nakamura H., Teixidor F., Viñas C.
Chemical Communications,
2014
9.

Tarrés M., Canetta E., Viñas C., Teixidor F., Harwood A.J.
Chemical Communications,
2014
10.

Hawthorne M.F., Maderna A.
Chemical Reviews,
1999
11.

Gozzi M., Schwarze B., Hey-Hawkins E.
Pure and Applied Chemistry,
2019
12.

Sivaev I.B., Bregadze V.V.
European Journal of Inorganic Chemistry,
2009
13.
10.1016/j.mencom.2022.05.021_b0065
Nakamura
Boron Science: New Technologies and Applications,
2012
14.

Viñas C., Teixidor F., Núñez R.
Inorganica Chimica Acta,
2014
15.

Gao S.M., Hosmane N.S.
Russian Chemical Bulletin,
2014
16.

Białek-Pietras M., Olejniczak A.B., Tachikawa S., Nakamura H., Leśnikowski Z.J.
Bioorganic and Medicinal Chemistry,
2013
17.
10.1016/j.mencom.2022.05.021_b0085
Kullberg
Int. J. Oncol.,
2003
18.

Albuquerque H., Santos C., Silva A.
Molecules,
2018
19.

Heber E.M., Hawthorne M.F., Kueffer P.J., Garabalino M.A., Thorp S.I., Pozzi E.C., Hughes A.M., Maitz C.A., Jalisatgi S.S., Nigg D.W., Curotto P., Trivillin V.A., Schwint A.E.
Proceedings of the National Academy of Sciences of the United States of America,
2014
20.

Feakes D.A., Spinler J.K., Harris F.R.
Tetrahedron,
1999
21.

Druzina A.A., Zhidkova O.B., Kosenko I.D.
Russian Chemical Bulletin,
2020
22.

Stogniy M.Y., Erokhina S.A., Druzina A.A., Sivaev I.B., Bregadze V.I.
Journal of Organometallic Chemistry,
2019
23.

Druzina A.A., Stogniy M.Y.
Russian Chemical Bulletin,
2021
24.
Druzina A.A., Kosenko I.D., Zhidkova O.B.
INEOS OPEN,
2020
25.

Boron-Containing Lipids and Liposomes: New Conjugates of Cholesterol with Polyhedral Boron Hydrides.
Bregadze V.I., Sivaev I.B., Dubey R.D., Semioshkin A., Shmal'ko A.V., Kosenko I.D., Lebedeva K.V., Mandal S., Sreejyothi P., Sarkar A., Shen Z., Wu A., Hosmane N.S.
Chemistry - A European Journal,
2020
26.

Dubey R.D., Sarkar A., Shen Z., Bregadze V.I., Sivaev I.B., Druzina A.A., Zhidkova O.B., Shmal'ko A.V., Kosenko I.D., P S., Mandal S., Hosmane N.S.
Journal of Pharmaceutical Sciences,
2021
27.
Druzina A.A., Shmalko A.V., Andreichuk E.P., Zhidkova O.B., Kosenko I.D., Semioshkin A., Sivaev I.B., Mandal S., Shen Z., Bregadze V.I.
Mendeleev Communications,
2019
28.

Druzina A.A., Kosenko I.D., Zhidkova O.B., Ananyev I.V., Timofeev S.V., Bregadze V.I.
European Journal of Inorganic Chemistry,
2020
29.

Druzina A.A., Shmalko A.V., Sivaev I.B., Bregadze V.I.
Russian Chemical Reviews,
2021
30.
![A Cholesterol Containing pH-Sensitive Bistable [2]Rotaxane](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Berg M., Nozinovic S., Engeser M., Lützen A.
European Journal of Organic Chemistry,
2015
31.

Razumov I.A., Zav’yalov E.L., Troitskii S.Y., Romashchenko A.V., Petrovskii D.V., Kuper K.E., Moshkin M.P.
Bulletin of Experimental Biology and Medicine,
2017
32.

Ring C.J.
Journal of General Virology,
2002
33.

Pass H.I., Lavilla C., Canino C., Goparaju C., Preiss J., Noreen S., Blandino G., Cioce M.
Oncotarget,
2016