Keywords
benzyl groups
cage compounds
hexaazaisowurtzitanes
nitrating mixtures
nitration
selective reaction
Abstract
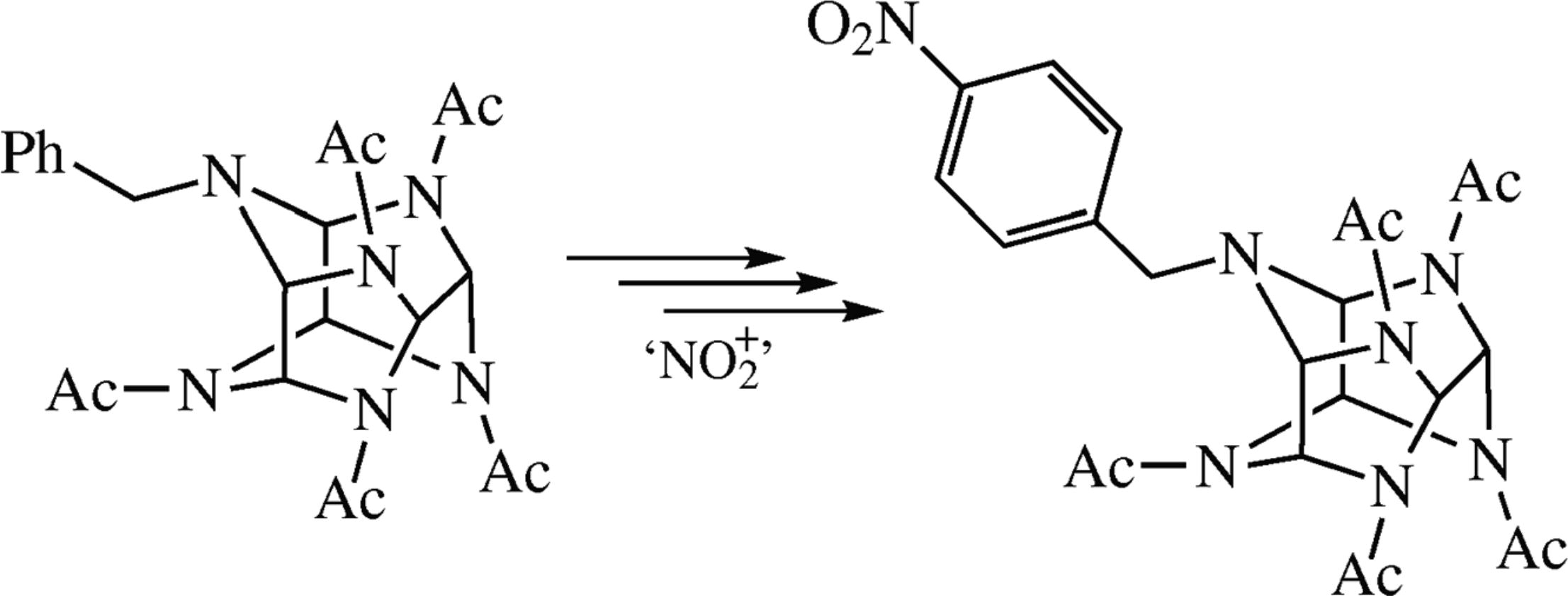
Nitration of N-benzylated 2,4,6,8,10,12-hexaazaisowurtzitane derivatives occurs selectively at para-positions of the benzyl groups. The best reagent for this reaction is ammonium nitrate in sulfuric acid.
References
1.

Bellamy A.J.
Tetrahedron,
1995
2.

Sysolyatin S.V., Lobanova A.A., Chernikova Y.T., Sakovich G.V.
Russian Chemical Reviews,
2005
3.
T. Kodama, M. Tojo and M. Ikeda, WO Patent 9623792 A1, 1996.
4.
A. T. Nielsen, US Patent 569379A, 1997.
5.

Latypov N.V., Wellmar U., Goede P., Bellamy A.J.
Organic Process Research and Development,
2000
6.

Ananikov V.P., Khemchyan L.L., Ivanova Y.V., Bukhtiyarov V.I., Sorokin A.M., Prosvirin I.P., Vatsadze S.Z., Medved'ko A.V., Nuriev V.N., Dilman A.D., Levin V.V., Koptyug I.V., Kovtunov K.V., Zhivonitko V.V., Likholobov V.A., et. al.
Russian Chemical Reviews,
2014
7.
10.1016/j.mencom.2022.05.019_b0035
Surmachev
Propellants, Explos., Pyrotech.,
1841
8.

Lin G., Tsai H., Tsai Y.
Bioorganic and Medicinal Chemistry Letters,
2003
9.

Bardai G., Sunahara G.I., Spear P.A., Martel M., Gong P., Hawari J.
Archives of Environmental Contamination and Toxicology,
2005
10.
T. G. Tolstikova, E. A. Morozova, S. V. Sysolyatin, A. I. Kalashnikov, Yu. I. Zhukova and V. N. Surmachev, Chem. Sustainable Dev., 2010, 18, 511 (Khimiya v Interesakh Ustoichivogo Razvitiya, 2010, 18, 527).
11.
![Nitrolysis of 2,6,8,12-tetraacetyl-4,10-dibenzyl-2,4,6,8,10,12-hexaazatetracyclo[5.5.0.03,11.05,9]dodecane](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kalashnikov A.I., Sysolyatin S.V., Sakovich G.V., Dubkov A.S., Kulagina D.A.
Russian Chemical Bulletin,
2017