Keywords
N-aminomethylation
acetals
aldehydes
aminals
cage compounds
formaldehyde
hexaazaisowurtzitanes
Abstract
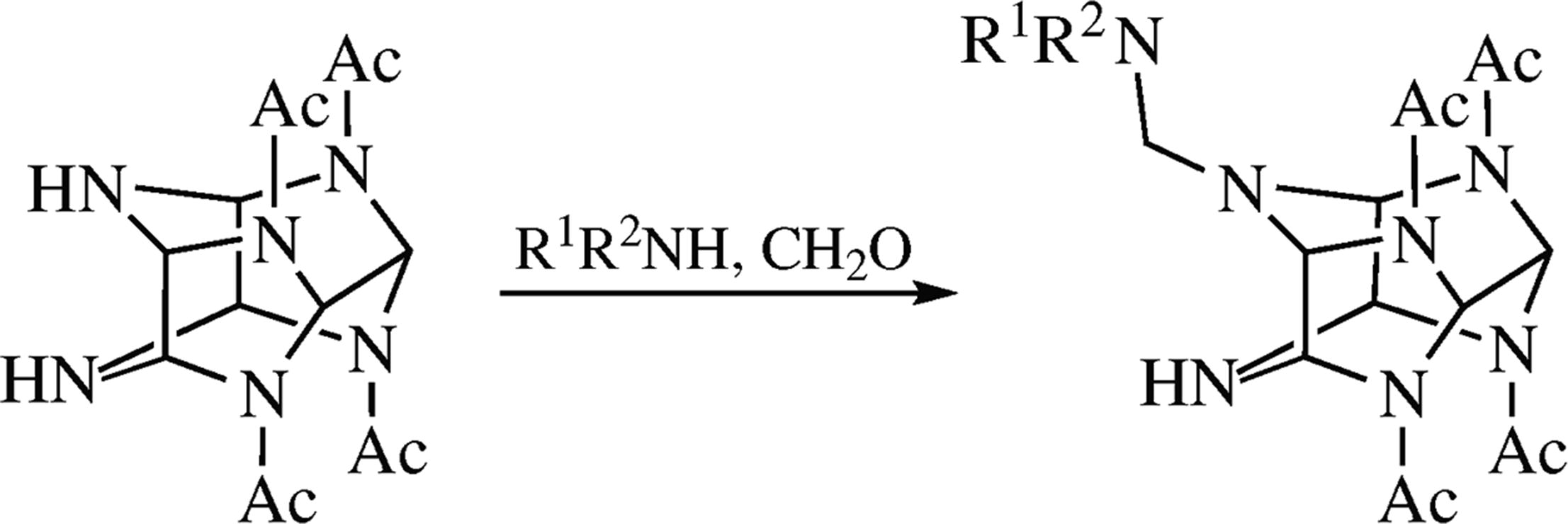
2,6,8,12-Tetraacetyl-2,4,6,8,10,12-hexaazaisowurtzitane was subjected to N-aminomethylation under various conditions, which afforded new derivatives of aminal nature. The highest yields were achieved when the reactions were carried out in a weak-alkali medium.
References
1.

Bellamy A.J.
Tetrahedron,
1995
2.

Sysolyatin S.V., Lobanova A.A., Chernikova Y.T., Sakovich G.V.
Russian Chemical Reviews,
2005
3.
T. Kodama, M. Tojo and M. Ikeda, WO Patent 9623792 A1, 1996.
4.

Latypov N.V., Wellmar U., Goede P., Bellamy A.J.
Organic Process Research and Development,
2000
5.
10.1016/j.mencom.2022.05.018_b0025
Surmachev
Propellants, Explos., Pyrotech.,
1841
6.

Lin G., Tsai H., Tsai Y.
Bioorganic and Medicinal Chemistry Letters,
2003
7.
10.1016/j.mencom.2022.05.018_b0035
Tolstikova
Chem. Sustainable Dev.,
2010
8.
Kulagina D.A., Sysolyatin S.V., Malykhin V.V., Kalashnikov A.I.
Mendeleev Communications,
2016
9.

Qiu W., Chen S., Yu Y.
Chinese Journal of Chemistry,
2010
10.

Chung K., Kil H., Choi I., Chu C., Lee I.
Journal of Heterocyclic Chemistry,
2000
11.
![Synthesis and crystal structure of 3,5,9,11-tetraacetyl-14-oxo-1,3,5,7,9,11-hexaazapentacyclo[5.5.3.02,6.04,10.08,12]pentadecane](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sun C., Zhao X.
Journal of Chemical Crystallography,
2006
12.
10.1016/j.mencom.2022.05.018_b0060
Kulagina
Yuzhno-sibirskii Nauchnyi Vestnik,
2018
13.
Rakhimova E.B., Kirsanov V.Y., Mescheryakova E.S., Ibragimov A.G., Dzhemilev U.M.
Mendeleev Communications,
2020