Keywords
bicyclic ligand precursor
ferrocene
Palladium complexes
Suzuki reaction
unsymmetrical pincer complexes
Abstract
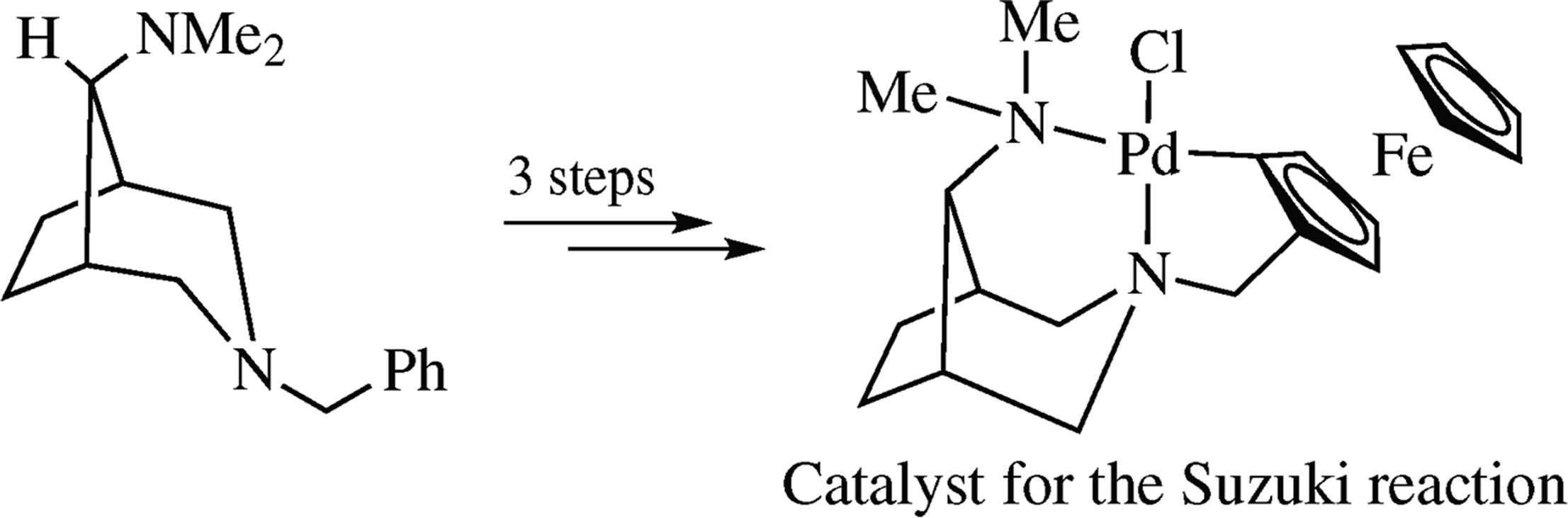
New bicyclic unsymmetrical CNN palladium 5,6-membered pincer complex was synthesized from 3-ferrocenylmethyl-8-dimethylamino-3-azabicyclo[3.2.1]octane by its direct cyclo-palladation with Li2PdCl4 or Na2PdCl4 and sodium acetate in MeOH. The obtained complex exhibited high catalytic activity in the Suzuki cross-coupling between aryl bromides and phenylboronic acid.
References
1.

I M., R S., B I., MT H., E D.
Current Organic Chemistry,
2009
2.

Niu J., Hao X., Gong J., Song M.
Dalton Transactions,
2011
3.

Selander N., Szabó K.J.
Chemical Reviews,
2010
4.

Asay M., Morales-Morales D.
Dalton Transactions,
2015
5.

Liu J., Gong J., Song M.
Organic and Biomolecular Chemistry,
2019
6.
Vasil’ev A.A., Burukin A.S., Zhdankina G.M., Zlotin S.G.
Mendeleev Communications,
2021
7.
![Palladium (II) and platinum (II) compounds containing bi- and terdentate ferrocenyl ligands. X-ray crystal structure of cis-[Pd{(η5-C5H5)Fe{(η5-C5H4)–CHN–CH2–CH2–N(CH3)2]}Cl2]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Caubet A., López C., Bosque R., Solans X., Font-Bardia M.
Journal of Organometallic Chemistry,
1999
8.
10.1016/j.mencom.2022.05.012_b0040
Lopez
J. Organomet. Chem.,
2002
9.

Butler I.R.
Organometallics,
1992
10.

Synthesis, structure and coordination chemistry of mono- and bis-heterocyclic-ferrocenyl derivatives
Isaac C.J., Price C., Horrocks B.R., Houlton A., Elsegood M.R., Clegg W.
Journal of Organometallic Chemistry,
2000
11.

Bulygina L.A., Khrushcheva N.S., Sokolov V.I.
Russian Chemical Bulletin,
2011
12.
![Unsymmetrical pincer CNN palladium complex of 7-ferrocenylmethyl-3-methyl-3,7-diazabicyclo[3.3.1]nonane](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bulygina L.A., Kagramanov N.D., Khrushcheva N.S., Lyssenko K.A., Peregudov A.S., Sokolov V.I.
Journal of Organometallic Chemistry,
2017
13.

Bechara A., Barbosa C.M., Paredes-Gamero E.J., Garcia D.M., Silva L.S., Matsuo A.L., Nascimento F.D., Rodrigues E.G., Caires A.C., Smaili S.S., Bincoletto C.
European Journal of Medicinal Chemistry,
2014
14.

Guillén E., González A., López C., Basu P.K., Ghosh A., Font‐Bardía M., Calvis C., Messeguer R.
European Journal of Inorganic Chemistry,
2015
15.

van Niekerk A., Chellan P., Mapolie S.F.
European Journal of Inorganic Chemistry,
2019
16.

Chellan P., Nasser S., Vivas L., Chibale K., Smith G.S.
Journal of Organometallic Chemistry,
2010
17.

Adams M., Barnard L., de Kock C., Smith P.J., Wiesner L., Chibale K., Smith G.S.
Dalton Transactions,
2016
18.
![5,6-Membered CNN palladium pincer complexes of 3-benzyl-8-dimethylamino -3-azabicyclo[3.2.1]octane and 3-benzyl-9-dimethylamino-3-azabicyclo[3.3.1]nonane](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bulygina L.A., Khrushcheva N.S., Lyssenko K.A., Peregudov A.S.
Journal of Organometallic Chemistry,
2019
19.
10.1016/j.mencom.2022.05.012_b0095
Moyano
Angew. Chem., Int. Ed.,
1865
20.
![Cyclopalladate complex of 3-benzyl-7-methyl-3,7-diazabicyclo[3.3.1]nonane](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bulygina L.A., Khrushcheva N.S., Peregudov A.S., Sokolov V.I.
Russian Chemical Bulletin,
2016
21.

Hao X., Wang Y., Liu J., Wang K., Gong J., Song M.
Journal of Organometallic Chemistry,
2010
22.

Avila-Sorrosa A., Jiménez-Vázquez H.A., Reyes-Arellano A., Pioquinto-Mendoza J.R., Toscano R.A., González-Sebastián L., Morales-Morales D.
Journal of Organometallic Chemistry,
2016
23.

Beletskaya I.P., Cheprakov A.V.
Journal of Organometallic Chemistry,
2004
24.

Sigeev A.S., Peregudov A.S., Cheprakov A.V., Beletskaya I.P.
Advanced Synthesis and Catalysis,
2015
25.

Kletskov A.V., Bumagin N.A., Petkevich S.K., Dikusar E.A., Lyakhov A.S., Ivashkevich L.S., Kolesnik I.A., Potkin V.I.
Inorganic Chemistry,
2020