Keywords
Hypercoordinated carbon
Non-classical structures
organoboron compounds
quantum-chemical calculations
Three- dimensional aromaticity
Abstract
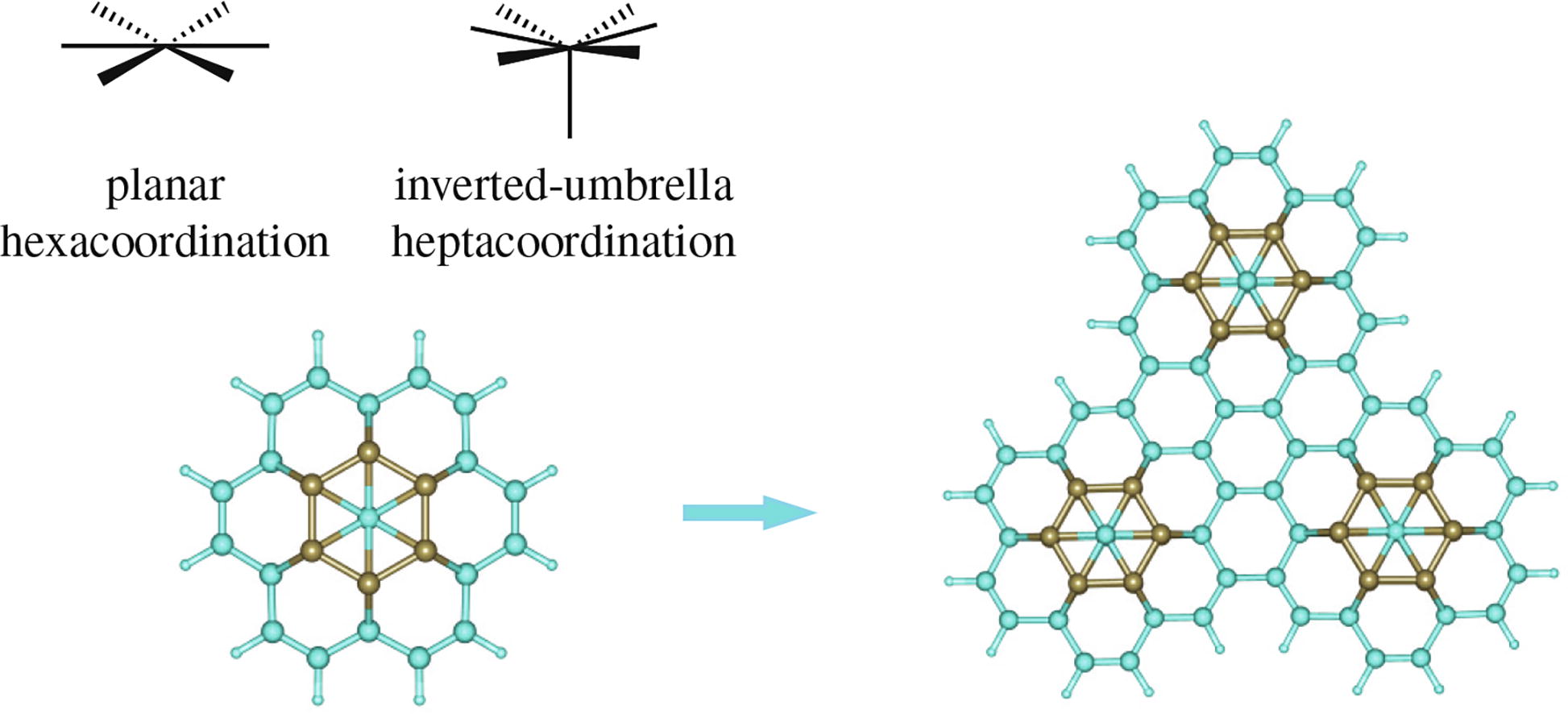
Calculations using the DFT B3LYP/6-311+G(df,2p) and wb97XD/6-311+G(df,2p) methods predict the stability of a new three-dimensional aromatic organoboron species based on the CB6 unit. Derived systems containing carbon atoms in two hypercoordination states (planar hexacoordination and inverted-umbrella heptacoordination) can be used as building blocks for constructing various stable condensed systems with many hypercoordinated carbon centers.
References
1.

Minkin V.I., Minyaev R.M., Hoffmann R.
Russian Chemical Reviews,
2002
2.

Wang Y., Li Y., Chen Z.
Accounts of Chemical Research,
2020
3.

Keese R.
Chemical Reviews,
2006
4.

Yang L., Ganz E., Chen Z., Wang Z., Schleyer P.V.
Angewandte Chemie - International Edition,
2015
5.

Vassilev-Galindo V., Pan S., Donald K.J., Merino G.
Nature Reviews Chemistry,
2018
6.
10.1016/j.mencom.2022.05.002_b0030
von Rague Schleyer
J. Chem. Soc., Chem. Commun.,
1991
7.
10.1016/j.mencom.2022.05.002_b0035
Minyaev
Comprehensive Inorganic Chemistry II: From Elements to Applications,
2013
8.

Gribanova T.N., Minyaev R.M., Minkin V.I.
Russian Journal of General Chemistry,
2008
9.

Wang Z., Schleyer P.V.
Science,
2001
10.
10.1016/j.mencom.2022.05.002_b0050
Exner
Science,
1937
11.

Minyaev R.M., Gribanova T.N., Starikov A.G., Minkin V.I.
Doklady Chemistry,
2002
12.
Minyaev R.M., Gribanova T.N., Starikov A.G., Minkin V.I.
Mendeleev Communications,
2001
13.
![[(?6-B6X)2M] (X=C, N; M=Mn, Fe, Co, Ni): A New Class of Transition-Metal Sandwich-Type Complexes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Li S., Guo J., Miao C., Ren G.
Angewandte Chemie - International Edition,
2005
14.

Luo Q., Zhang X.H., Huang K.L., Liu S.Q., Yu Z.H., Li Q.S.
Journal of Physical Chemistry A,
2007
15.

Wang Y., Li F., Li Y., Chen Z.
Nature Communications,
2016
16.

Yeoh K.H., Chew K.-., Chu Y.Z., Yoon T.L., Rusi, Ong D.S.
Journal of Applied Physics,
2019
17.

Becke A.D.
Journal of Chemical Physics,
1993
18.

Chai J., Head-Gordon M.
Physical Chemistry Chemical Physics,
2008
19.
10.1016/j.mencom.2022.05.002_b0095
Foresman
Exploring Chemistry with Electronic Structure Methods,
1996
20.
10.1016/j.mencom.2022.05.002_b0100
Frisch
Gaussian 16, Revision C.01,
2016
21.

Reed A.E., Curtiss L.A., Weinhold F.
Chemical Reviews,
1988
22.
10.1016/j.mencom.2022.05.002_b0110
Glendening
NBO 6.0,
2013
23.
10.1016/j.mencom.2022.05.002_b0115
Bader
Atoms in Molecules: A Quantum Theory,
1990
24.
10.1016/j.mencom.2022.05.002_b0120
Keith
AIMAll, version 11.06.19,
2011
25.
G.A. Zhurko, ChemCraft software, version 1.8, http://www.chemcraftprog.com.
26.

Hoffmann R., Schleyer P., Schaefer H.
Angewandte Chemie - International Edition,
2008
27.
10.1016/j.mencom.2022.05.002_b0135
Emsley
The Elements,
1991
28.

Lu T., Chen F.
Journal of Physical Chemistry A,
2013
29.

Shaik S., Danovich D., Silvi B., Lauvergnat D.L., Hiberty P.C.
Chemistry - A European Journal,
2005
30.

Shaik S., Danovich D., Wu W., Hiberty P.C.
Nature Chemistry,
2009
31.

Schleyer P.V., Maerker C., Dransfeld A., Jiao H., van Eikema Hommes N.J.
Journal of the American Chemical Society,
1996
32.

Kertesz M.
Chemistry - A European Journal,
2018