Keywords
crystal structure
crystallization
hydrogen bonds
phase behaviour
Polymorphism
π⋯π interaction
Abstract
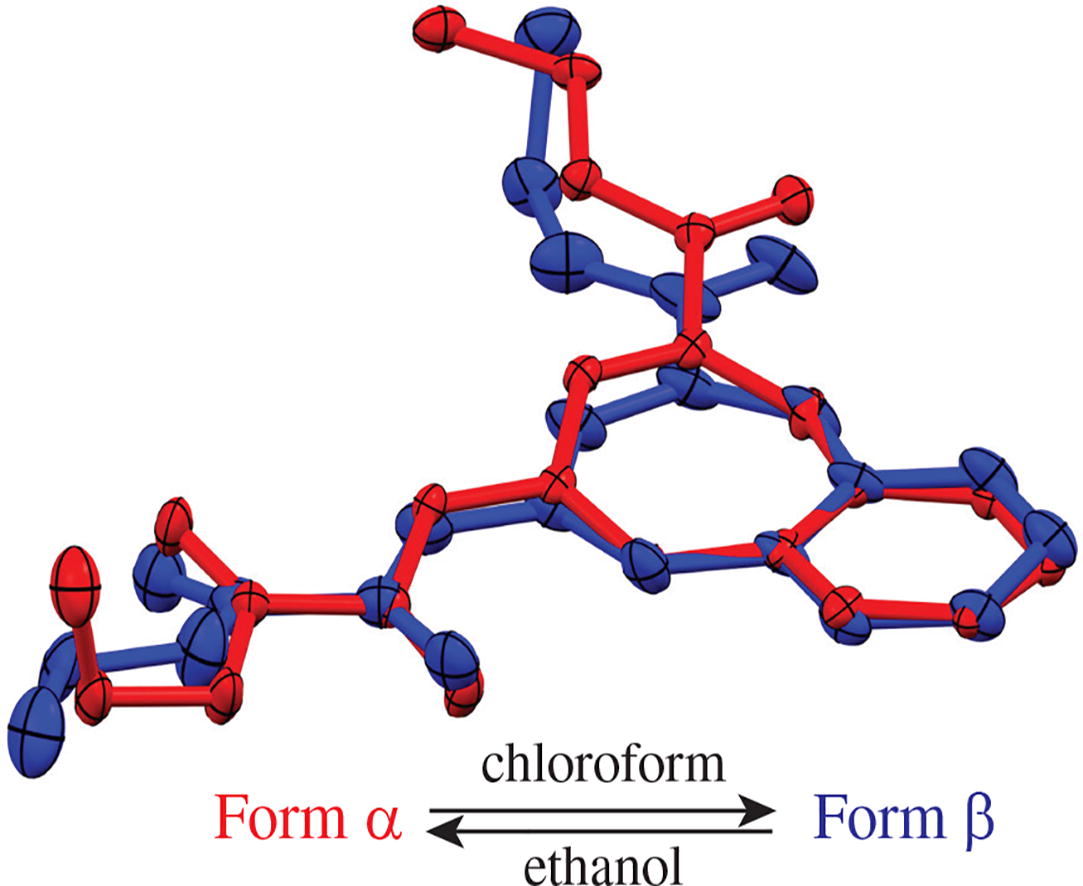
Ethyl 4-[(Z)-3-ethoxy-2,3-dioxopropylidene]-4,5-dihydro-1H-benzo[b][1,4]diazepine-2-carboxylate forms two polymorphic modifications α and β, which differ in molecular conformations, as well as in the dimension of H-bonded supramolecular motifs in crystals that are a 1D chain or a 0D dimer, respectively. The formation of polymorphs can be controlled by choice of the solvent used. Crystallization from a solution in a polar protic solvent, ethanol, results in the thermodynamically favorable α-form, while using a low-polarity solvent, chloroform, and melt crystallization lead to the metastable β-form.
References
1.
2.
Crystallization: Basic Concepts and Industrial Applications, ed. W. Beckmann, Wiley-VCH, Weinheim, 2013.
3.

5.

Kitamura M.
CrystEngComm,
2009
6.

Chen J., Trout B.L.
Journal of Physical Chemistry B,
2008
7.

Kulkarni S.A., McGarrity E.S., Meekes H., ter Horst J.H.
Chemical Communications,
2012
8.

Parveen S., Davey R.J., Dent G., Pritchard R.G.
Chemical Communications,
2005
9.

Gavezzotti A., Filippini G., Kroon J., van Eijck B.P., Klewinghaus P.
Chemistry - A European Journal,
1997
10.

Musumeci D., Hunter C.A., McCabe J.F.
Crystal Growth and Design,
2010
11.

Bobrovs R., Seton L., Actiņš A.
CrystEngComm,
2014
12.

Barbui C., Cipriani A., Patel V., Ayuso-Mateos J.L., van Ommeren M.
British Journal of Psychiatry,
2011
13.
![Condensation of diethyl 2,4,6-Trioxoheptanedioate with 2-(Aryliminomethyl)phenols. A new synthesis of chromeno[4,3-b]pyridines](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Mamedov V.A., Murtazina A.M., Sysoeva L.P., Mironova E.V., Latypov S.K., Balandina A.A., Kadyrova S.F., Gubaidullin A.T., Litvinov I.A.
Russian Journal of Organic Chemistry,
2008
14.
![Fused polycyclic nitrogen-containing heterocycles 24. Three-component condensation of diethyl 2,4,6-trioxoheptanedicarboxylate with salicylaldehydes and ammonium acetate as a new method for the synthesis of 7- and 9-substituted benzo[e]pyrano[4,3-b]pyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Mamedov V.A., Sysoeva L.P., Murtazina A.M., Berdnikov E.A., Gubaidullin A.T., Kadyrova S.F., Mironova E.V., Litvinov I.A.
Russian Chemical Bulletin,
2009
15.
![1,5-Benzodiazepines IX. A new route to substituted 4H-[1,2,4]triazolo[4,3-a][1,5]benzodiazepin-5-amines with analgesic and/or anti-inflammatory activities](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Roma G., Grossi G., Di Braccio M., Ghia M., Mattioli F.
European Journal of Medicinal Chemistry,
1991
16.

Arora N., Dhiman P., Kumar S., Singh G., Monga V.
Bioorganic Chemistry,
2020
17.

An Y., Hao Z., Zhang X., Wang L.
Chemical Biology and Drug Design,
2016
18.

Wang L., Li X., An Y.
Organic and Biomolecular Chemistry,
2015
19.

Fayzullin R.R., Zakharychev D.V., Gubaidullin A.T., Antonovich O.A., Krivolapov D.B., Bredikhina Z.A., Bredikhin A.A.
Crystal Growth and Design,
2016
20.

Lodochnikova O.A., Kosolapova L.S., Saifina A.F., Gubaidullin A.T., Fayzullin R.R., Khamatgalimov A.R., Litvinov I.A., Kurbangalieva A.R.
CrystEngComm,
2017
21.

Lodochnikova O.A., Zaripova A.R., Fayzullin R.R., Samigullina A.I., Vandyukova I.I., Potapova L.N., Kurbangalieva A.R.
CrystEngComm,
2018
22.

Bredikhin A.A., Zakharychev D.V., Bredikhina Z.A., Gubaidullin A.T., Fayzullin R.R.
CrystEngComm,
2012