Keywords
1,2,4-trioxolanes
1,5-diketones
dispiro compounds
hydrogen peroxide
ozonides
stereoisomerism
tetracyclic compounds
Abstract
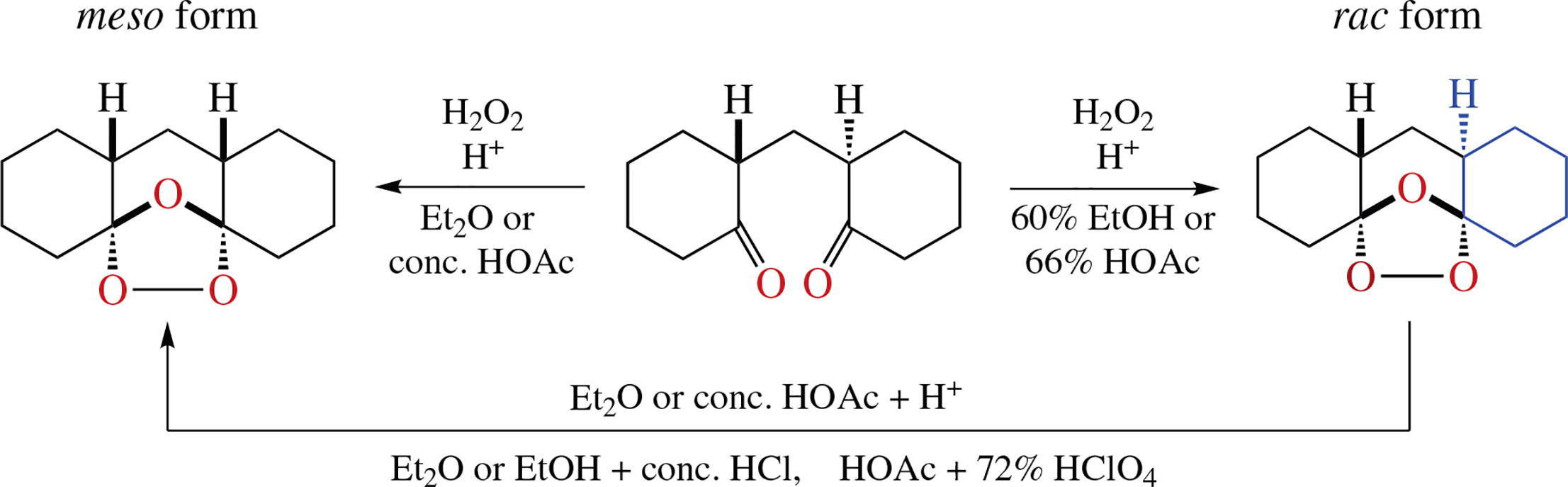
The conditions and reasons for the stereoselective transformation of the rac form of 2,2′-(methylene)-dicyclohexanone into the meso- or rac-diastereomer of tetracyclic dispiro ozonide (1,2,4-trioxolane) in the reaction with 30% aqueous H2O2 in the presence of acid have been determined. A mechanism for the stereoisomerization of ozonides was proposed, and the stereochemistry of diastereomeric ozonides was established by NMR data.
References
1.
J. L. Vennerstrom, Y. Dong, J. Chollet and H. Matile, US Patent 6486199 B1, 2002.
2.

Vennerstrom J.L., Arbe-Barnes S., Brun R., Charman S.A., Chiu F.C., Chollet J., Dong Y., Dorn A., Hunziker D., Matile H., McIntosh K., Padmanilayam M., Santo Tomas J., Scheurer C., Scorneaux B., et. al.
Nature,
2004
3.

Vil’ V., Yaremenko I., Ilovaisky A., Terent’ev A.
Molecules,
2017
4.

Abrams R.P., Carroll W.L., Woerpel K.A.
ACS Chemical Biology,
2016
5.

Küster T., Kriegel N., Stadelmann B., Wang X., Dong Y., Vennerstrom J.L., Keiser J., Hemphill A.
International Journal of Antimicrobial Agents,
2014
6.

Chaudhary S., Sharma V., Jaiswal P.K., Gaikwad A.N., Sinha S.K., Puri S.K., Sharon A., Maulik P.R., Chaturvedi V.
Organic Letters,
2015
7.

Yaremenko I.A., Radulov P.S., Belyakova Y.Y., Demina A.A., Fomenkov D.I., Barsukov D.V., Subbotina I.R., Fleury F., Terent'ev A.O.
Chemistry - A European Journal,
2020
8.

Griesbaum K., Liu X., Dong Y.
Tetrahedron,
1997
9.

Tang Y., Dong Y., Karle J.M., DiTusa C.A., Vennerstrom J.L.
Journal of Organic Chemistry,
2004
10.

Yaremenko I.A., Gomes G.D., Radulov P.S., Belyakova Y.Y., Vilikotskiy A.E., Vil’ V.A., Korlyukov A.A., Nikishin G.I., Alabugin I.V., Terent’ev A.O.
Journal of Organic Chemistry,
2018
11.
T. I. Akimova and O. A. Soldatkina, RU Patent 2578609, 2016.
12.

Akimova T.I., Rybin V.G., Soldatkina O.A.
Russian Journal of Organic Chemistry,
2019
13.
Kondelíková J., Králíček J., Kubánek V.
Collection of Czechoslovak Chemical Communications,
2012
14.

Miura M., Ikegami A., Nojima M., Kusabayashi S., McCullough K.J., Nagase S.
Journal of the American Chemical Society,
1983
15.

Yaremenko I.A., Syroeshkin M.A., Levitsky D.O., Fleury F., Terent’ev A.O.
Medicinal Chemistry Research,
2016
16.
1,5-Dikarbonil’nye soedineniya v organicheskom sinteze (1,5-Dicarbonyl Compounds in Organic Synthesis), eds. V. I. Vysotskii and V. L. Novikov, Dalnevostochnyi Federal’nyi Universitet, Vladivostok, 2014 (in Russian).